
What is the product of the reaction below?
c
If an aldehyde is oxidized, what type of compound would be produced?
a. a ketone
b. a different aldehyde
c. a carboxylic acid
d. an alcohol
An important reaction in chemistry is oxidation. Which is an oxidation reaction?
a. An alcohol is converted into a ketone.
b. An oxygen is removed from an alcohol.
c. A double bond between an oxygen and a carbon is broken.
d. A carbonyl group is modified to produce an alcohol.
What would be the product if we attempt to oxidize a 3º alcohol?
a. There would be no product, 3º alcohols cannot be oxidized.
b. The product is a ketone.
c. The product is a carboxylic acid.
d. The product is a thiol.
A reaction occurs in an acid solution during which two alcohol molecules react with a ketone. The product will be a(an) ___.
a. ketose
b. acetal
c. alcoholic ketone
d. acidic ketone
Which nucleophile, when reacted with 1-bromopropane (CH3CH2CH2Br), would produce a thiol?
a. OH-
b. SH-
c. I-
d. -OCH3
Aldehydes and ketones both contain a carbonyl carbon atom. They differ in that ___.
a. the carbonyl carbon atom in ketones is attached to two other carbons and in aldehydes, the carbonyl carbon atom is also attached to at least one hydrogen atom.
b. the carbonyl carbon atom in ketones is primary and in aldehydes, the carbonyl is secondary.
c. the carbonyl carbon atom in ketones has a hydrogen attached to it.
d. the carbonyl carbon atom in aldehydes has two carbon atoms attached to it
Each of these is based on the propane molecule. Which is
NOT
capable of
forming hydrogen bonds to another molecule of the same type?
a. 1-propanol
b. 1-propanethiol
c. 2-propanol
d. propanoic acid
The common name for CH3-O-CH3 is ___.
a. methanal
b. dimethanol
c. ethyl ether
d. dimethyl ether
The reaction in which a primary alcohol becomes an aldehyde is an oxidation reaction.
TRUE
Which nucleophile, when reacted with 1-bromopropane (CH3CH2CH2Br), would produce an alcohol?
a. OH-
b. SH-
c. I-
d. -OCH3
A reducing agent found in some biological systems is ___.
a. H2O2
b. acetaldehyde
c. Pt
d. NADH
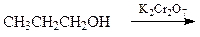
What is the major organic product of the following reaction?
a. CH3CH=CH2
b. CH3CH2CH2Cr
c. CH3CH2CH3
d. CH3CH2CO2H
When alcohols are dehydrated in the presence of H+
and heat, the major product is that formed by removal of
–OH from one carbon atom and removal of –H from the neighboring C atom
that carries more H atoms.Select one:
True
False
What is the IUPAC name of this compound?
a. 4-methy-4-propylbutylaldehyde
b. 4-methylhexanal
c. 4-ethylpentanal
d. 4-ethylpentylaldehyde
Which compound is a hemiacetal?
b.
The oxidation of methanethiol by I2
produces what product?
a.
If formaldehyde reacts with methanol in an acid solution, the product formed is ___.
a. a new aldehyde
b. an alcohol that is very polar
c. a hemiacetal
d. an acid
The compound that is expected to have the highest boiling point is ___.
the alcohol produced from methane.
b. the thiol produced from methane.
c. methane.
d. They all boil at the same temperature since methane is the basis for the compounds.
Benedict’s solution turns a blue color in the presence of glucose.
FALSE
The method to determine the number of stereoisomers of a monosaccharide is to calculate 2n, where n is the total number of carbons in the molecule.
FALSE
Cellulose is composed of glucose molecules connected by a-glycosidic linkages.
FALSE
Both starch and cellulose are polysaccharides of glucose.
TRUE
How many stereoisomers would a carbohydrate with 5 chiral carbons have?
a. 5 stereoisomers
b. 20 stereoisomersc.
c. 32 stereoisomers
d. 60 stereoisomers
Shown below is a cyclic form of D-galactose. This structure is classified as ___.
a. a-D-galactopyranoseb.
b
-D-galactofuranosec.
a-D-galactofuranosed.
b
-D-galactopyranose
Hyaluronic acid solutions serve as lubricants in the fluid of joints and are present in the vitreous humor of the eye. A portion of its structure is shown below.
a. Hyaluronic acid is a homopolysaccharide.
b. Hyaluronic acid is a heteropolysaccharide.
c. Hyaluronic acid is made of D-glucose residues.
d. The monosaccharide residues in hyaluronic acid are of furanose form.
The disaccharides are important in nature; a few examples of disaccharides are
a. D-abqueose, D-fructose, galactose, and glucose.
b. raffinose, stachyose, and verbascose.
c. cellobiose, lactose, maltose, and sucrose.
d. amylopectin, amylose, and starch.
Shown below is a cyclic form of D-Fructose. This structure is classified as ___.
a. a-D-fructopyranoseb.
b-D-fructopyranose
c. b-D-fructofuranose
d. a-D-fructofuranose
A pair of compounds are stereoisomers and they are nonsuperimposable mirror images. This pair of isomers is referred to as a pair of enantiomers.
TRUE
Which of the following is a fruit sugar?
a. D-glucose
b. Galactose
c. Lactose
d. D-Fructose
D-glucose is often called by other names, such as corn sugar and grape sugar. It is also referred to as ___.
a. dextrose
b. fruit sugar
c. blood sugar
d. dextrose and blood sugar
What is the classification of this monosaccharide?
a. aldopentose
b. aldohexose
c. ketopentose
d. ketohexose
Shown below is a cyclic form of D-mannose. This structure is classified as ___.
a. a-D-mannopyranose
b. b-D-mannofuranose
c. a-D-mannofuranosed
d. b-D-mannopyranose
Blood type is determined by oligosaccharides attached to the surface of red blood cells.
TRUE
How would this compound be classified?
a. carboxylic carbohydrate b. hydroxyaldehyde
c. ketotriose
d. aldotriose
Relative sweetness is a measure of the sweetness of any chosen sweetener compared to that of
a. invert sugar.
b. the sweetest carbohydrate, fructose.
c. sucrose.
d. saccharin.
Glycogen and amylopectin are similar in that they are both polymers of glucose. Glycogen is the glucose storage form for ___ and starch is the glucose storage form for ___.
a. plants/plants
b. animals/animals
c. plants/animals
d. animals/plants
Stereoisomers that are not mirror images of one another are called
a. enantiomers
b. anomers
c. diastereomers
d. Fischer projections
The structural formula for erythrose is drawn here. The carbon atoms are numbered from the carbonyl group.
a. This is the only formula for erythrose; there are no stereoisomers.b. This is both a D and an
b. This is both a D and an L structure, as determined by carbons #1 and #2.
c. This is D-erythrose, as determined by carbon #3.
d. This is L-erythrose, as determined by carbon #4.
Glycogen is a polymer of glucose produced by plants for energy storage.
FALSE
Fructose is different from glucose and galactose because
a. fructose is a pentose; the other sugars are hexoses.
b. fructose is a ketose; the other sugars are aldoses.
c. the cyclic form is a square; the other sugars display hexagons.
d. fructose doesn’t have an open form; the other sugars do have an open form.
An aldose is a carbohydrate that contains
a. hydroxy groups on the terminal carbons.
b. water molecules on the alpha carbon.
c. an aldehyde group.
d. monosaccharides.
Which of the following reactant is missing in the reaction below?
A
The organic product expected from the following reaction is:
a
Tertiary alcohols have three carbon atoms attached to carbon that has the OH directly attached.
TRUE
Ethanol is miscible in water, while 1-pentanol has a solubility of just 2.2g/100 mL because solubility in water decreases with increasing alcohol size.
TRUE
Hyaluronic acid solutions serve as lubricants in the fluid of joints and are present in the vitreous humor of the eye. A section of the structure for hyaluronic acid is:
WHICH OF THE FOLLOWING STATEMENTS IS TRUE
a. The alternating pattern of glycosidic bond types are a-(1→3) and a-(1→4).
b. The alternating pattern of glycosidic bond types are - (1→3) and - (1→4).
c. The alternating pattern of glycosidic bond types are - (1→3) and a- (1→4).
d. The alternating pattern of glycosidic bond types are - (1→3) and - (1→3).
What is the classification of this monosaccharide?
a. aldotriose b. ketotriose c. aldotetrose d . ketotetrose
Maltose is a monosaccharide
False
The structure of the compound 2,3,4-trihydroxybutanal is shown below. How many stereoisomers are possible for this molecule?
a. 2 B. 4 c. 8 d. 16
Sucrose is a disaccharide of D-glucose connected to D-fructose by an a,b-(1 →2)- glycosidic bond. Sucrose is not a reducing sugar because
a. it is a disaccharide.
b. it reacts with Benedict's solution. c. it contains both glucose and fructose.
d. it cannot react to produce an aldehyde group.
Which of the following statements is true about aldehydes?
a. Aldehydes have much lower boiling points than alcohols with a similar molecular weight.b. Aldehydes interact with like molecules through hydrogen bonding.c. The carbonyl carbon atom in an aldehyde can never be in position number 1.d. The parent chain of an aldehyde is named by dropping the final “e” from a corresponding alkane and adding “one.”
Which compound type is most susceptible to a nucleophillic substitution reaction?
a. hydrocarbon b. alcohol c. alkyl halide d. acid
The IUPAC name for the organic compound below is
a. 3-Butanol
b. 2-Butanol
c. Ethyl methyl ether
d. 3-butanethiol
What is mutarotation?
a. The conversion of a D-monosaccharide into an L-monosaccharide.
b. The conversion of a-D-galacose into b-D-galactose.
c. The conversion of a pyranose into a furanose.
d. The conversion of an aldose into a ketose.
Which are the most commonly found monosaccharides in nature?
a. those with 4 carbons
b. those with 5-6 carbons
c. those with 7-9 carbons
d. those with 10 or more carbons
The reduction of a ketone produces a 2° alcohol.
TRUE
The structural similarity of acetals and ethers is the presence of ___.
a. C—O—C
b. a carboxylic carbon
c. C—O—H
d. CHO
Oxidation of a secondary alcohol results in the formation of a(n) ___.
a. tertiary alcohol
b. acid
c. aldehyde
d. ketone