Which term describes the degree to which an element attracts electrons?
Electronegativity
Electronegativity is the tendency of an atom to attract electrons toward itself.
Which terms describe two atoms when they form a bond in which electrons are completely transferred from one atom to the other?
Anion and cation.
Each atom will carry a charge from the transfer of electrons.
Which of the following statements is true of the bonds in a water molecule?
Oxygen holds electrons more tightly than hydrogen does, and the net charge is zero
The oxygen and hydrogen atoms in water have partial charges, but the molecule has a net charge of zero.
Which of the following statements is not true of most cellular redox reactions?
A hydrogen atom is transferred to the atom that loses an electron
A hydrogen atom (proton, or H+) is often transferred to the atom that gains an electron.
What kind of bond is formed when lithium and fluorine combine to form lithium fluoride?
Ionic.
The complete transfer of an electron from lithium to fluorine results in a stable compound in which both atoms have full outermost shells.
Gaseous hydrogen burns in the presence of oxygen to form water:
2H2 + O2 → 2H2 O + energy
Which molecule is oxidized and what kind of bond is formed?
Hydrogen, polar.
Hydrogen loses electrons to oxygen, which is more electronegative and thus pulls the electrons closer to itself in the water molecule
Which of the following best describes the main purpose of the combined processes of glycolysis and cellular respiration?
transforming the energy in glucose and related molecules in a chemical form that cells can use for work
The energy made available during cellular respiration is coupled to a production of ATP, the basic energy currency that cells use for work.
In the combined processes of glycolysis and cellular respiration, what is consumed and what is produced?
Glucose is consumed, and carbon dioxide is produced.
The carbon in glucose is oxidized to carbon dioxide during cellular respiration.
Why does the oxidation of organic compounds by molecular oxygen to produce CO2 and water release free energy?
Electrons are being moved from atoms that have a lower affinity for electrons (such as C) to atoms with a higher affinity for electrons (such as O
Which of the following statements describes the results of this reaction?
C6H12O6 + 6 O2 → 6 CO2 + 6 H2O + Energy
C6H12O6 is oxidized and O2 is reduced.
When a glucose molecule loses a hydrogen atom as the result of an oxidation-reduction reaction, the molecule becomes
oxidized.
When a molecule of NAD+ (nicotinamide adenine dinucleotide) gains a hydrogen atom (not a proton), the molecule becomes
reduced.
Where does glycolysis take place in eukaryotic cells?
cytosol
The ATP made during glycolysis is generated by
substrate-level phosphorylation
The oxygen consumed during cellular respiration is involved directly in which process or event?
accepting electrons at the end of the electron transport chain
Why are carbohydrates and fats considered high energy foods?
They have a lot of electrons associated with hydrogen.
Glycolysis
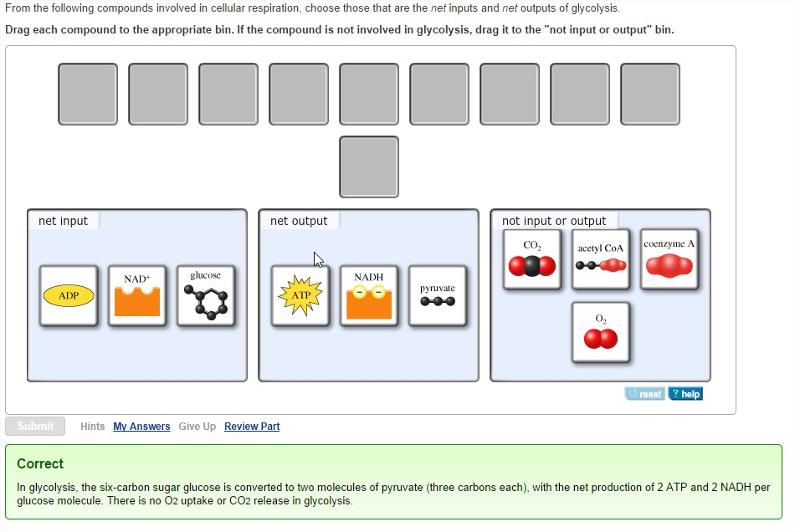
From the following compounds involved in cellular respiration, choose those that are the net inputs and net outputs of glycolysis.
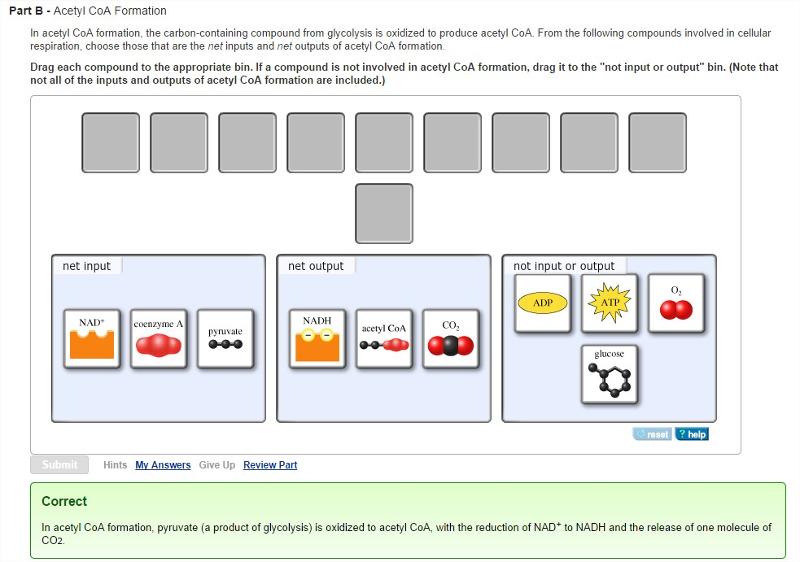
Acetyl CoA Formation
In acetyl CoA formation, the carbon-containing compound from glycolysis is oxidized to produce acetyl CoA. From the following compounds involved in cellular respiration, choose those that are the net inputs and net outputs of acetyl CoA formation.
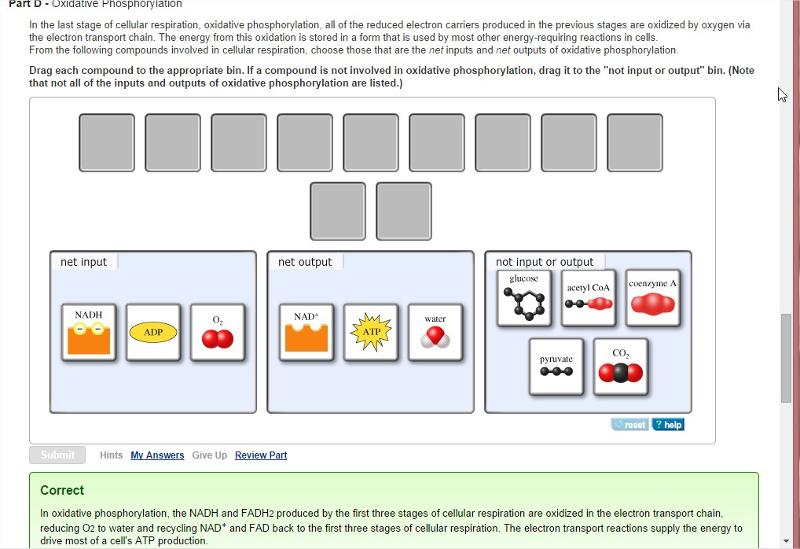
Oxidative Phosphorylation
In the last stage of cellular respiration, oxidative phosphorylation, all of the reduced electron carriers produced in the previous stages are oxidized by oxygen via the electron transport chain. The energy from this oxidation is stored in a form that is used by most other energy-requiring reactions in cells.
From the following compounds involved in cellular respiration, choose those that are the net inputs and net outputs of oxidative phosphorylation.
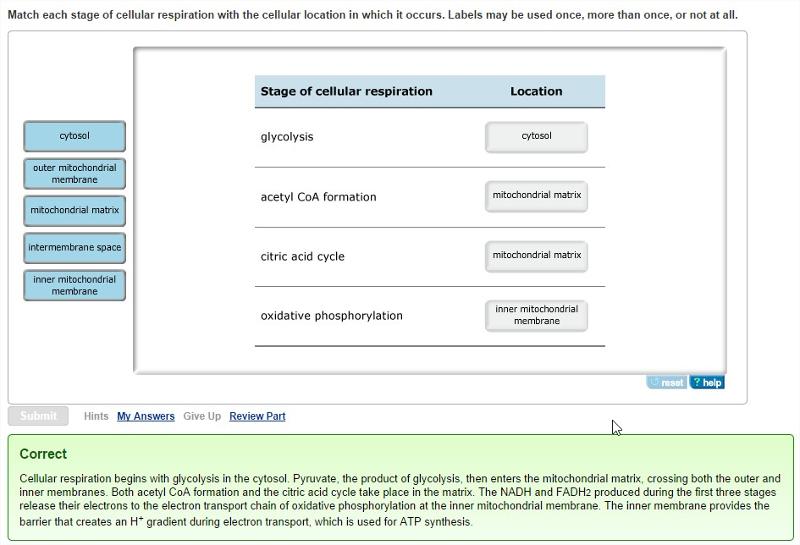
Cellular locations of the four stages of cellular respiration
Each of the four stages of cellular respiration occurs in a specific location inside or outside the mitochondria. These locations permit precise regulation and partitioning of cellular resources to optimize the utilization of cellular energy.
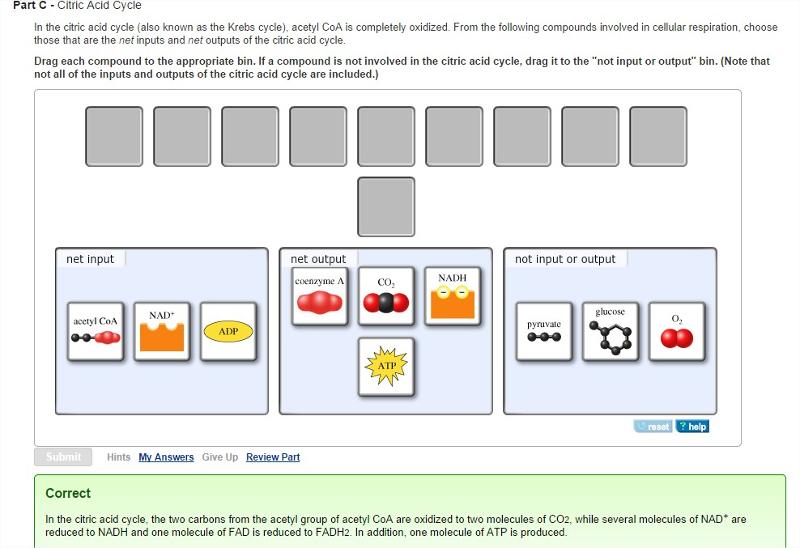
Citric Acid Cycle
In the citric acid cycle (also known as the Krebs cycle), acetyl CoA is completely oxidized. From the following compounds involved in cellular respiration, choose those that are the net inputs and net outputs of the citric acid cycle.
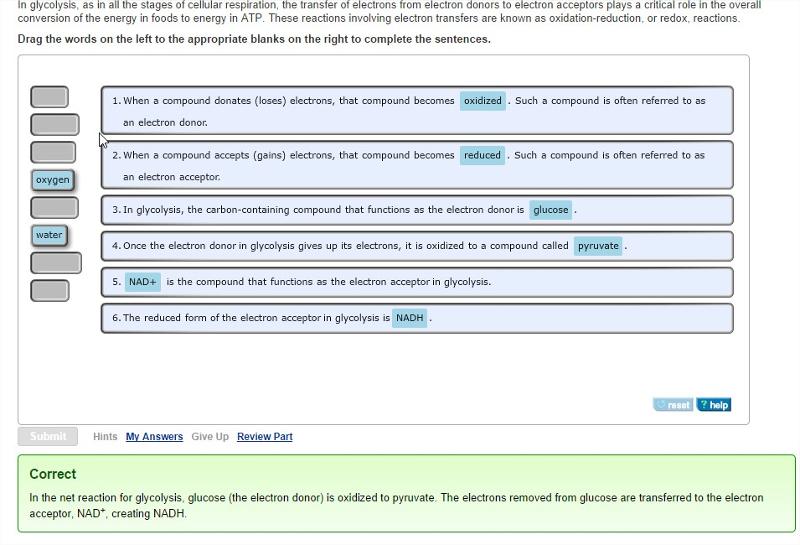
Redox (oxidation-reduction) reactions in glycolysis
In glycolysis, as in all the stages of cellular respiration, the transfer of electrons from electron donors to electron acceptors plays a critical role in the overall conversion of the energy in foods to energy in ATP. These reactions involving electron transfers are known as oxidation-reduction, or redox, reactions.
Energy from glycolysis
Among the products of glycolysis, which compounds contain energy that can be used by other biological reactions?
pyruvate, ATP, and NADH
ATP is the main product of cellular respiration that contains energy that can be used by other cellular processes. Some ATP is made in glycolysis. In addition, the NADH and pyruvate produced in glycolysis are used in subsequent steps of cellular respiration to make even more ATP.
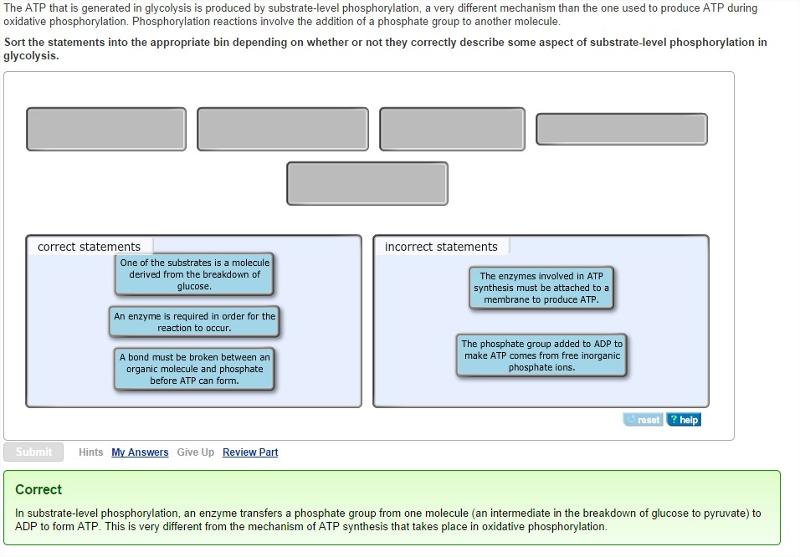
The ATP that is generated in glycolysis is produced by substrate-level phosphorylation, a very different mechanism than the one used to produce ATP during oxidative phosphorylation. Phosphorylation reactions involve the addition of a phosphate group to another molecule.
How many NADH are produced by glycolysis?
2
Two NADH molecules are produced by glycolysis.
In glycolysis, ATP molecules are produced by _____.
substrate-level phosphorylation
A phosphate group is transferred from glyceraldehyde phosphate to ADP
Which of these is NOT a product of glycolysis?
FADH2
FADH2 is a product of the citric acid cycle.
In glycolysis, what starts the process of glucose oxidation?
ATP
Some ATP energy is used to start the process of glucose oxidation.
In glycolysis there is a net gain of _____ ATP
2
It takes 2 ATP to produce 4 ATP
Glycolysis
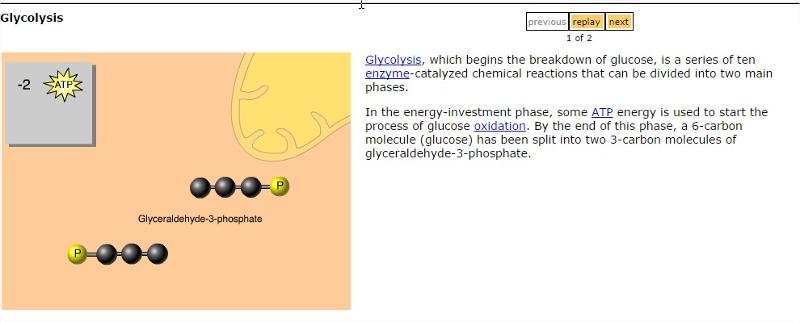
Which of the following describes the process of glycolysis?
It represents the first stage in the chemical oxidation of glucose by a cell.
Catabolism of glucose begins with glycolysis.
Substrate-level phosphorylation accounts for approximately what percentage of the ATP formed by the reactions of glycolysis?
100%
In addition to ATP, what are the end products of glycolysis?
NADH and pyruvate
In glycolysis, for each molecule of glucose oxidized to pyruvate
two molecules of ATP are used and four molecules of ATP are produced.
A molecule that is phosphorylated
has an increased chemical potential energy; it is primed to do cellular work.
Which kind of metabolic poison would most directly interfere with glycolysis?
an agent that closely mimics the structure of glucose but is not metabolized