group of molecular biologists is trying to synthesize a new artificial compound to mimic the effects of a known hormone that influences sexual behavior. They have turned to you for advice. Which of the following compounds is most likely to mimic the effects of the hormone?
A) a compound with the same number of carbon atoms as the hormone
B) a compound with the same molecular mass (measured in daltons) as the hormone
C) a compound with the same three-dimensional shape as part of the hormone
D) a compound with the same number of orbital electrons as the hormone
E) a compound with the same number of hydrogen and nitrogen atoms as the hormone
Answer: C
The complexity and variety of organic molecules is due to
A) the chemical versatility of carbon atoms.
B) the variety of rare elements in organic molecules.
C) the fact that they can be synthesized only in living organisms.
D) their interaction with water.
E) their tremendously large sizes.
Answer: A
Differences among organisms are caused by
A) large differences in elemental composition from organism to organism.
B) differences in the types and relative amounts of organic molecules synthesized by each organism.
C) differences in the elements that bond with carbon in each organism.
D) differences in the sizes of the organic molecules in each organism.
E) differences in inorganic compounds present in each organism
Answer: B
Which of the following statements correctly describes cis-trans isomers?
A) They have variations in arrangement around a double bond.
B) They have an asymmetric carbon that makes them mirror images.
C) They have the same chemical properties.
D) They have different molecular formulas.
E) Their atoms and bonds are arranged in different sequences.
Answer: A
Research indicates that ibuprofen, a drug used to relieve inflammation and pain, is a mixture of two enantiomers; that is, molecules that
A) have identical chemical formulas but differ in the branching of their carbon skeletons.
B) are mirror images of one another.
C) exist in either linear chain or ring forms.
D) differ in the location of their double bonds.
E) differ in the arrangement of atoms around their double bonds.
Answer: B
What determines whether a carbon atom's covalent bonds to other atoms are in a tetrahedral configuration or a planar configuration?
A) the presence or absence of bonds with oxygen atoms
B) the presence or absence of double bonds between the carbon atom and other atoms
C) the polarity of the covalent bonds between carbon and other atoms
D) the presence or absence of bonds with nitrogen atoms
E) the solvent that the organic molecule is dissolved in
Answer: B
Compared to a hydrocarbon chain where all the carbon atoms are linked by single bonds, a hydrocarbon chain with the same number of carbon atoms, but with one or more double bonds, will
A) be more flexible in structure.
B) be more constrained in structure.
C) be more polar.
D) have more hydrogen atoms.
E) have fewer structurally distinct isomers.
Answer: B
Organic molecules with only hydrogens and five carbon atoms can have different structures in all of the following ways except
A) by branching of the carbon skeleton.
B) by varying the number of double bonds between carbon atoms.
C) by varying the position of double bonds between carbon atoms.
D) by forming a ring.
E) by forming enantiomers.
Answer: E
A compound contains hydroxyl groups as its predominant functional group. Which of the following statements is true concerning this compound?
A) It lacks an asymmetric carbon, and it is probably a fat or lipid.
B) It should dissolve in water.
C) It should dissolve in a nonpolar solvent.
D) It won't form hydrogen bonds with water.
E) It is hydrophobic.
Answer: B
Which of the following is a false statement concerning amino groups?
A) They are basic in pH.
B) They are found in amino acids.
C) They contain nitrogen.
D) They are nonpolar.
E) They are components of urea.
Answer: D
Which two functional groups are always found in amino acids?
A) ketone and methyl
B) carbonyl and amino
C) carboxyl and amino
D) amino and sulfhydryl
E) hydroxyl and carboxyl
Answer: C
Amino acids are acids because they always possess which functional group?
A) amino
B) carbonyl
C) carboxyl
D) phosphate
E) hydroxyl
Answer: C
Which functional groups can act as acids?
A) amino and sulfhydryl
B) carbonyl and carboxyl
C) carboxyl and phosphate
D) hydroxyl and aldehyde
E) ketone and amino
Answer: C
Testosterone and estradiol are
A) soluble in water.
B) structural isomers of each other.
C) proteins.
D) lipids.
E) enantiomers of each other.
Answer: B
Testosterone and estradiol are male and female sex hormones, respectively, in many vertebrates. In what way(s) do these molecules differ from each other?
A) Testosterone and estradiol are structural isomers but have the same molecular formula.
B) Testosterone and estradiol are cis-trans isomers but have the same molecular formula.
C) Testosterone and estradiol have different functional groups attached to the same carbon skeleton.
D) Testosterone and estradiol have distinctly different chemical structures, with one including four fused rings of carbon atoms, while the other has three rings.
E) Testosterone and estradiol are enantiomers of the same organic molecule.
Answer: C
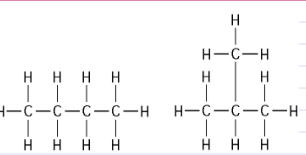
The two molecules shown in the figure above are best described as
A) optical isomers.
B) enantiomers.
C) structural isomers.
D) cis-trans isomers.
E) chain length isomers.
Answer: C
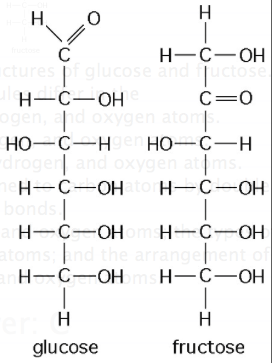
he figure above shows the structures of glucose and fructose. These two molecules differ in the
A) number of carbon, hydrogen, and oxygen atoms.
B) types of carbon, hydrogen, and oxygen atoms.
C) arrangement of carbon, hydrogen, and oxygen atoms.
D) number of oxygen atoms joined to carbon atoms by double covalent bonds.
E) number of carbon, hydrogen, and oxygen atoms; the types of carbon, hydrogen, and oxygen atoms; and the arrangement of carbon, hydrogen, and oxygen atoms.
The figure above shows the structures of glucose and fructose. These two molecules are
A) geometric isotopes.
B) enantiomers.
C) cis-trans isomers.
D) structural isomers.
E) nonisotopic isomers.
Answer: C
Answer: D
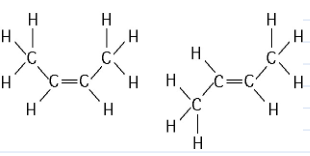
The two molecules shown in the figure above are best described as
A) enantiomers.
B) radioactive isotopes.
C) structural isomers.
D) nonisotopic isomers.
E) cis-trans isomers.
Answer: E
Thalidomide and L-dopa, shown below, are examples of pharmaceutical drugs that occur as enantiomers, or molecules that
A) have identical three-dimensional shapes.
B) are mirror images of one another.
C) are structural isomers.
D) are mirror images of one another and have the same biological activity.
E) are cis-trans isomers.
Answer: B
O=C-O-H
-What is the name of the functional group shown in the figure above?
A) carbonyl
B) ketone
C) aldehyde
D) carboxyl
E) hydroxyl
Carboxyl
A. -OH B. C=O C. O=C-O-H D. -NH2 E. -SH
Which functional group shown above is characteristic of alcohols?
A) A
B) B
C) C
D) D
E) E
Answer: A
A. -OH B. C=O C. O=C-O-H D. -NH2 E. -SH
Which functional group(s) shown above is (are) present in all amino acids?
A) A and B
B) B and D
C) C only
D) D only
E) C and D
Answer: E
A. -OH B. C=O C. O=C-O-H D. -NH2 E. -SH
Which of the groups shown above is a carbonyl functional group?
A) A
B) B
C) C
D) D
E) E
Answer: B
A. -OH B. C=O C. O=C-O-H D. -NH2 E. -SH
Which of the groups shown above is a functional group that helps stabilize proteins by forming covalent cross-links within or between protein molecules?
A) A
B) B
C) C
D) D
E) E
Answer: E
A. -OH B. C=O C. O=C-O-H D. -NH2 E. -SH
Which of the groups above is a carboxyl functional group?
A) A
B) B
C) C
D) D
E) E
Answer: C
A. -OH B. C=O C. O=C-O-H D. -NH2 E. -SH
Which of the groups above is an acidic functional group that can dissociate and release H⁺ into a solution?
A) A
B) B
C) C
D) D
E) E
Answer: C
A. -OH B. C=O C. O=C-O-H D. -NH2 E. -SH
Which of the groups above is a basic functional group that can accept H⁺ and become positively charged?
A) A
B) B
C) C
D) D
E) E
Answer: D
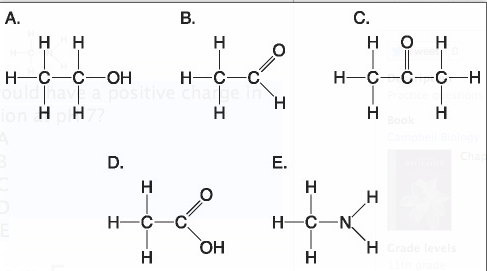
Which molecule shown above would have a positive charge in aqueous solution at pH 7?
A) A
B) B
C) C
D) D
E) E
Answer: E
***Which molecule(s) shown above is (are) ionized in aqueous solution at pH 7?
A) A
B) B and D
C) D and E
D) D
E) E
Answer: A
***Which molecules shown above contain a carbonyl group?
A) A and B
B) B and C
C) B, C, and D
D) D and E
E) E and A
Answer: B
***Which molecule shown above has a carbonyl functional group in the form of a ketone?
A) A
B) B
C) C
D) D
E) E
Answer: C
***Which molecule shown above has a carbonyl functional group in the form of an aldehyde?
A) A
B) B
C) C
D) D
E) E
Answer:B
***Which molecule shown above contains a carboxyl group?
A) A
B) B
C) C
D) D
E) E
Answer: D
Which molecule shown above can increase the concentration of hydrogen ions in a solution and is therefore an organic acid?
A) A
B) B
C) C
D) D
E) E
Answer: D
Humans and mice differ because
A) their cells have different small organic molecules.
B) their cells make different types of large biological molecules.
C) their cells make different types of lipids.
D) their cells have some differences in the sequence of nucleotides in their nucleic acids.
E) their cells make different types of proteins.
Answer: D
Molecules with which functional groups may form polymers via dehydration reactions?
A) hydroxyl groups
B) carbonyl groups
C) carboxyl groups
D) either carbonyl or carboxyl groups
E) either hydroxyl or carboxyl groups
Answer: E
Which of these molecules is not formed by dehydration reactions?
A) fatty acids
B) disaccharides
C) DNA
D) protein
E) amylose
Answer: A
) In animal metabolism, most of the monomers released by digestion of food macromolecules are metabolized to provide energy. Only a small portion of these monomers are used for synthesis of new macromolecules. The net result is that
A) water is generated by animal metabolism.
B) water is consumed by animal metabolism.
C) the water consumed is exactly balanced by the water generated, to maintain homeostasis.
D) water is consumed during homeostasis, but water is generated during periods of growth.
E) water is generated during homeostasis, but water is consumed during periods of growth.
Answer: B
What is the chemical reaction mechanism by which cells make polymers from monomers?
A) phosphodiester linkages
B) hydrolysis
C) dehydration reactions
D) ionic bonding of monomers
E) the formation of disulfide bridges between monomers
Answer: C
Which of the following best summarizes the relationship between dehydration reactions and hydrolysis?
A) Dehydration reactions assemble polymers, and hydrolysis reactions break down polymers.
B) Dehydration reactions eliminate water from lipid membranes, and hydrolysis makes lipid membranes water permeable.
C) Dehydration reactions can occur only after hydrolysis.
D) Hydrolysis creates monomers, and dehydration reactions break down polymers.
E) Dehydration reactions ionize water molecules and add hydroxyl groups to polymers; hydrolysis reactions release hydroxyl groups from polymers.
Answer: A
All of the following contain amino acids except
A) hemoglobin.
B) cholesterol.
C) antibodies.
D) enzymes.
E) insulin.
Answer: B
) The bonding of two amino acid molecules to form a larger molecule requires
A) the release of a water molecule.
B) the release of a carbon dioxide molecule.
C) the addition of a nitrogen atom.
D) the addition of a water molecule.
E) the release of a nitrous oxide molecule.
Answer: A
There are 20 different amino acids. What makes one amino acid different from another?
A) different side chains (R groups) attached to a carboxyl carbon
B) different side chains (R groups) attached to the amino groups
C) different side chains (R groups) attached to an α carbon
D) different structural and optical isomers
E) different asymmetric carbons
Answer: C
Dehydration reactions are used in forming which of the following compounds?
A) triacylglycerides
B) polysaccharides
C) proteins
D) triacylglycerides and proteins only
E) triacylglycerides, polysaccharides, and proteins
Answer: E
What aspects of protein structure are stabilized or assisted by hydrogen bonds?
A) primary structure
B) secondary structure
C) tertiary structure
D) quaternary structure
E) secondary, tertiary, and quaternary structures, but not primary structure
Answer: E
Which bonds are created during the formation of the primary structure of a protein?
A) peptide bonds
B) hydrogen bonds
C) disulfide bonds
D) phosphodiester bonds
E) peptide bonds, hydrogen bonds, and disulfide bonds
Answer: A
What maintains the secondary structure of a protein?
A) peptide bonds
B) hydrogen bonds between the amino group of one peptide bond and the carboxyl group of another peptide bond
C) disulfide bonds
D) hydrophobic interactions
E) hydrogen bonds between the R groups
Answer: B
What type of covalent bond between amino acid side chains (R groups) functions in maintaining a polypeptide's specific three-dimensional shape?
A) ionic bond
B) hydrophobic interaction
C) van der Waals interaction
D) disulfide bond
E) hydrogen bond
Answer: D
Misfolding of polypeptides is a serious problem in cells. Which of the following diseases are associated with an accumulation of misfolded polypeptides?
A) Alzheimer's only
B) Parkinson's only
C) diabetes mellitus only
D) Alzheimer's and Parkinson's only
E) Alzheimer's, Parkinson's, and diabetes mellitus
Answer:D
Changing a single amino acid in a protein consisting of 325 amino acids would
A) alter the primary structure of the protein, but not its tertiary structure or function.
B) cause the tertiary structure of the protein to unfold.
C) always alter the biological activity or function of the protein.
D) always alter the primary structure of the protein and disrupt its biological activity.
E) always alter the primary structure of the protein, sometimes alter the tertiary structure of the protein, and affect its biological activity.
Answer: E
Normal hemoglobin is a tetramer, consisting of two molecules of β hemoglobin and two molecules of α hemoglobin. In sickle-cell disease, as a result of a single amino acid change, the mutant hemoglobin tetramers associate with each other and assemble into large fibers. Based on this information alone, we can conclude that sickle-cell hemoglobin exhibits
A) altered primary structure.
B) altered secondary structure.
C) altered tertiary structure.
D) altered quaternary structure.
E) altered primary structure and altered quaternary structure; the secondary and tertiary structures may or may not be altered.
Answer: E
Which of the following statements about the 5' end of a polynucleotide strand of DNA is correct?
A) The 5' end has a hydroxyl group attached to the number 5 carbon of ribose.
B) The 5' end has a phosphate group attached to the number 5 carbon of ribose.
C) The 5' end has phosphate attached to the number 5 carbon of the nitrogenous base.
D) The 5' end has a carboxyl group attached to the number 5 carbon of ribose.
E) The 5' end is the fifth position on one of the nitrogenous bases.
Answer: B
One of the primary functions of RNA molecules is to
A) transmit genetic information to offspring.
B) function in the synthesis of proteins.
C) make a copy of itself, thus ensuring genetic continuity.
D) act as a pattern or blueprint to form DNA.
E) form the genes of higher organisms.
Answer: B
If ¹⁴C-labeled uridine triphosphate is added to the growth medium of cells, what macromolecules will be labeled?
A) phospholipids
B) DNA
C) RNA
D) both DNA and RNA
E) proteins
Answer: C
Which of the following descriptions best fits the class of molecules known as nucleotides?
A) a nitrogenous base and a phosphate group
B) a nitrogenous base and a pentose sugar
C) a nitrogenous base, a phosphate group, and a pentose sugar
D) a phosphate group and an adenine or uracil
E) a pentose sugar and a purine or pyrimidine
Answer: C
Which of the following are nitrogenous bases of the pyrimidine type?
A) guanine and adenine
B) cytosine and uracil
C) thymine and guanine
D) ribose and deoxyribose
E) adenine and thymine
Answer: B
Which of the following are nitrogenous bases of the purine type?
A) cytosine and guanine
B) guanine and adenine
C) adenine and thymine
D) thymine and uracil
E) uracil and cytosine
Answer: B
The difference between the sugar in DNA and the sugar in RNA is that the sugar in DNA
A) is a six-carbon sugar and the sugar in RNA is a five-carbon sugar.
B) can form a double-stranded molecule.
C) is an aldehyde sugar and the sugar in RNA is a keto sugar.
D) is in the α configuration and the sugar in RNA is in the β configuration.
E) contains one less oxygen atom.
Answer: E
Which of the following statements best summarizes the differences between DNA and RNA?
A) DNA encodes hereditary information, whereas RNA does not.
B) The bases in DNA form base-paired duplexes, whereas the bases in RNA do not.
C) DNA nucleotides contain a different sugar than RNA nucleotides.
D) DNA contains the base uracil, whereas RNA contains the base thymine.
E) DNA encodes hereditary information, whereas RNA does not; the bases in DNA form base-paired duplexes, whereas the bases in RNA do not; and DNA nucleotides contain a different sugar than RNA nucleotides.
Answer: C
If one strand of a DNA molecule has the sequence of bases 5'ATTGCA3', the other complementary strand would have the sequence
A) 5'TAACGT3'.
B) 5'TGCAAT3'.
C) 5'UAACGU3'.
D) 3'UAACGU5'.
E) 5'UGCAAU3'.
Answer: B
What is the structural feature that allows DNA to replicate?
A) sugar-phosphate backbone
B) complementary pairing of the nitrogenous bases
C) disulfide bonding (bridging) of the two helixes
D) twisting of the molecule to form an α helix
E) three-component structure of the nucleotides
Answer: B
Which of the following is an example of hydrolysis?
A) the reaction of two monosaccharides, forming a disaccharide with the release of water
B) the synthesis of two amino acids, forming a peptide with the release of water
C) the reaction of a fat, forming glycerol and fatty acids with the release of water
D) the reaction of a fat, forming glycerol and fatty acids with the consumption of water
E) the synthesis of a nucleotide from a phosphate, a pentose sugar, and a nitrogenous base with the production of a molecule of water
Answer: D
If cells are grown in a medium containing radioactive ³²P-labeled phosphate, which of these molecules will be labeled?
A) phospholipids
B) nucleic acids
C) proteins
D) amylose
E) both phospholipids and nucleic acids
Answer: E
If cells are grown in a medium containing radioactive ¹⁵N, which of these molecules will be labeled?
A) fatty acids only
B) nucleic acids only
C) proteins only
D) amylase only
E) both proteins and nucleic acids
Answer: E
Approximately 32 different monomeric carbohydrate subunits are found in various natural polysaccharides. Proteins are composed of 20 different amino acids. DNA and RNA are each synthesized from four nucleotides.
Among these biological polymers, which has the least structural variety?
A) polysaccharides
B) proteins
C) DNA
D) RNA
Answer: C
Which class of biological polymer has the greatest functional variety?
A) polysaccharides
B) proteins
C) DNA
D) RNA
Answer: B
Which organelle or structure is absent in plant cells?
A) mitochondria
B) Golgi vesicles
C) microtubules
D) centrosomes
E) peroxisomes
Answer: D
Large numbers of ribosomes are present in cells that specialize in producing which of the following molecules?
A) lipids
B) glycogen
C) proteins
D) cellulose
E) nucleic acids
Answer: C
The nuclear lamina is an array of filaments on the inner side of the nuclear membrane. If a method were found that could cause the lamina to fall into disarray, what would you expect to be the most likely consequence?
A) the loss of all nuclear function
B) the inability of the nucleus to divide during cell division
C) a change in the shape of the nucleus
D) failure of chromosomes to carry genetic information
E) inability of the nucleus to keep out destructive chemicals
Answer: C
A cell with a predominance of free ribosomes is most likely
A) producing primarily proteins for secretion.
B) producing primarily cytoplasmic proteins.
C) constructing an extensive cell wall or extracellular matrix.
D) digesting large food particles.
E) enlarging its vacuole.
Answer: B
Which type of organelle or structure is primarily involved in the synthesis of oils, phospholipids, and steroids?
A) ribosome
B) lysosome
C) smooth endoplasmic reticulum
D) mitochondrion
E) contractile vacuole
Answer: C
Which structure is the site of the synthesis of proteins that may be exported from the cell?
A) rough ER
B) lysosomes
C) plasmodesmata
D) Golgi vesicles
E) free cytoplasmic ribosomes
Answer: A
The Golgi apparatus has a polarity or sidedness to its structure and function. Which of the following statements correctly describes this polarity?
A) Transport vesicles fuse with one side of the Golgi and leave from the opposite side.
B) Proteins in the membrane of the Golgi may be sorted and modified as they move from one side of the Golgi to the other.
C) Lipids in the membrane of the Golgi may be sorted and modified as they move from one side of the Golgi to the other.
D) Soluble proteins in the cisternae (interior) of the Golgi may be sorted and modified as they move from one side of the Golgi to the other.
E) All of the above correctly describe polar characteristics of the Golgi function.
Answer: E
The fact that the outer membrane of the nuclear envelope has bound ribosomes allows one to most reliably conclude that
A) at least some of the proteins that function in the nuclear envelope are made by the ribosomes on the nuclear envelope.
B) the nuclear envelope is not part of the endomembrane system.
C) the nuclear envelope is physically separated from the endoplasmic reticulum.
D) small vesicles from the Golgi fuse with the nuclear envelope.
E) nuclear pore complexes contain proteins.
Answer: A
The difference in lipid and protein composition between the membranes of the endomembrane system is largely determined by
A) the physical separation of most membranes from each other.
B) the transportation of membrane lipids among the endomembrane system by small membrane vesicles.
C) the function of the Golgi apparatus in sorting and directing membrane components.
D) the modification of the membrane components once they reach their final destination.
E) the synthesis of different lipids and proteins in each of the organelles of the endomembrane system.
Answer: C
Hydrolytic enzymes must be segregated and packaged to prevent general destruction of cellular components. Which of the following organelles contains these hydrolytic enzymes in animal cells?
A) chloroplast
B) lysosome
C) central vacuole
D) peroxisome
E) glyoxysome
Answer: B
Which of the following statements correctly describes some aspect of protein secretion from prokaryotic cells?
A) Prokaryotes are unlikely to be able to secrete proteins because they lack an endomembrane system.
B) The mechanism of protein secretion in prokaryotes is probably the same as that in eukaryotes.
C) Proteins that are secreted by prokaryotes are synthesized on ribosomes that are bound to the cytoplasmic surface of the plasma membrane.
D) In prokaryotes, the ribosomes that are used for the synthesis of secreted proteins are located outside of the cell.
E) Prokaryotes contain large pores in their plasma membrane that permit the movement of proteins out of the cell.
Answer: C
Tay-Sachs disease is a human genetic abnormality that results in cells accumulating and becoming clogged with very large and complex lipids. Which cellular organelle must be involved in this condition?
A) the endoplasmic reticulum
B) the Golgi apparatus
C) the lysosome
D) mitochondria
E) membrane-bound ribosomes
Answer: C
The liver is involved in detoxification of many poisons and drugs. Which of the following structures is primarily involved in this process and therefore abundant in liver cells?
A) rough ER
B) smooth ER
C) Golgi apparatus
D) nuclear envelope
E) transport vesicles
Answer: B
Which of the following produces and modifies polysaccharides that will be secreted?
A) lysosome
B) vacuole
C) mitochondrion
D) Golgi apparatus
E) peroxisome
Answer: D
Which of the following contains hydrolytic enzymes?
A) lysosome
B) vacuole
C) mitochondrion
D) Golgi apparatus
E) peroxisome
Answer: A
Which organelle often takes up much of the volume of a plant cell?
A) lysosome
B) vacuole
C) mitochondrion
D) Golgi apparatus
E) peroxisome
Answer: B
Which organelle is the primary site of ATP synthesis in eukaryotic cells?
A) lysosome
B) vacuole
C) mitochondrion
D) Golgi apparatus
E) peroxisome
Answer: C
Which plant cell organelle contains its own DNA and ribosomes?
A) glyoxysome
B) vacuole
C) mitochondrion
D) Golgi apparatus
E) peroxisome
Answer: C
The chemical reactions involved in respiration are virtually identical between prokaryotic and eukaryotic cells. In eukaryotic cells, ATP is synthesized primarily on the inner membrane of the mitochondria. In light of the endosymbiont theory for the evolutionary origin of mitochondria, where is most ATP synthesis likely to occur in prokaryotic cells?
A) in the cytoplasm
B) on the inner mitochondrial membrane
C) on the endoplasmic reticulum
D) on the plasma membrane
E) on the inner nuclear envelope
Answer: D
In a liver cell detoxifying alcohol and some other poisons, the enzymes of the peroxisome remove hydrogen from these molecules and
A) combine the hydrogen with water molecules to generate hydrogen peroxide.
B) use the hydrogen to break down hydrogen peroxide.
C) transfer the hydrogen to the mitochondria.
D) transfer the hydrogen to oxygen molecules to generate hydrogen peroxide.
Answer: D
Motor proteins provide for molecular motion in cells by interacting with what types of cellular structures?
A) sites of energy production in cellular respiration
B) membrane proteins
C) ribosomes
D) cytoskeletal structures
E) cellulose fibers in the cell wall
Answer: D
Centrioles, cilia, flagella, and basal bodies have remarkably similar structural elements and arrangements. Which of the following hypotheses is most plausible in light of such structural similarities?
A) Cilia and flagella arise from the centrioles.
B) Loss of basal bodies should lead to loss of all cilia, flagella, and centrioles.
C) Motor proteins such as dynein must have evolved before any of these four kinds of structure.
D) Cilia and flagella coevolved in the same ancestral eukaryotic organism.
E) Natural selection for cell motility repeatedly selected for microtubular arrays in circular patterns in the evolution of each of these structures.
Answer: A
Cytochalasin D is a drug that prevents actin polymerization. A cell treated with cytochalasin D will still be able to
A) perform amoeboid movement.
B) form cleavage furrows.
C) contract muscle fibers.
D) extend pseudopodia.
E) move vesicles around the cell.
Answer: E
When a potassium ion (K+) moves from the soil into the vacuole of a cell on the surface of a root, it must pass through several cellular structures. Which of the following correctly describes the order in which these structures will be encountered by the ion?
A) plasma membrane → primary cell wall → cytoplasm → vacuole
B) secondary cell wall → plasma membrane → primary cell wall → cytoplasm → vacuole
C) primary cell wall → plasma membrane → cytoplasm → vacuole
D) primary cell wall → plasma membrane → lysosome → cytoplasm → vacuole
E) primary cell wall → plasma membrane → cytoplasm → secondary cell wall → vacuole
Answer: C
A mutation that disrupts the ability of an animal cell to add polysaccharide modifications to proteins would most likely cause defects in its
A) nuclear lamina and nuclear matrix.
B) nuclear matrix and extracellular matrix.
C) mitochondria and Golgi apparatus.
D) Golgi apparatus and extracellular matrix.
E) nuclear pores and secretory vesicles.
Answer: D
ECM proteins are made by ribosomes in which part of a eukaryotic cell?
A) mitochondria
B) cytoplasm
C) nuclear envelope
D) Golgi apparatus
E) rough ER
Answer: E
What types of proteins are not synthesized in the rough ER?
A) endoplasmic reticulum proteins
B) extracellular matrix proteins
C) secreted proteins
D) mitochondrial proteins
E) plasma membrane proteins
Answer: D
A biologist ground up some plant leaf cells and then centrifuged the mixture to fractionate the organelles. Organelles in one of the heavier fractions could produce ATP in the light, whereas organelles in the lighter fraction could produce ATP in the dark. The heavier and lighter fractions are most likely to contain, respectively,
A) mitochondria and chloroplasts.
B) chloroplasts and peroxisomes.
C) peroxisomes and chloroplasts.
D) chloroplasts and mitochondria.
E) mitochondria and peroxisomes.
Answer: D
Which structure is not part of the endomembrane system?
A) nuclear envelope
B) chloroplast
C) Golgi apparatus
D) plasma membrane
E) ER
Answer: B
Which structure-function pair is mismatched?
A) nucleolus; production of ribosomal subunits
B) lysosome; intracellular digestion
C) ribosome; protein synthesis
D) Golgi; protein trafficking
E) microtubule; muscle contraction
Answer: E
Cyanide binds with at least one molecule involved in producing ATP. If a cell is exposed to cyanide, most of the cyanide will be found within the
A) mitochondria.
B) ribosomes.
C) peroxisomes.
D) lysosomes.
E) endoplasmic reticulum.
Answer: A
What is the most likely pathway taken by a newly synthesized protein that will be secreted by a cell?
A) ER → Golgi → nucleus
B) Golgi →ER →lysosome
C) nucleus →ER →Golgi
D) ER →Golgi →vesicles that fuse with plasma membrane
E) ER →lysosomes →vesicles that fuse with plasma membrane
Answer: D
Who was/were the first to propose that cell membranes are phospholipid bilayers?
A) H. Davson and J. Danielli
B) I. Langmuir
C) C. Overton
D) S. Singer and G. Nicolson
E) E. Gorter and F. Grendel
Answer: E
Singer and Nicolson's fluid mosaic model of the membrane proposed that
A) membranes are a phospholipid bilayer.
B) membranes are a phospholipid bilayer between two layers of hydrophilic proteins.
C) membranes are a single layer of phospholipids and proteins.
D) membranes consist of protein molecules embedded in a fluid bilayer of phospholipids.
E) membranes consist of a mosaic of polysaccharides and proteins.
Answer: D
The presence of cholesterol in the plasma membranes of some animals
A) enables the membrane to stay fluid more easily when cell temperature drops.
B) enables the animal to remove hydrogen atoms from saturated phospholipids.
C) enables the animal to add hydrogen atoms to unsaturated phospholipids.
D) makes the membrane less flexible, allowing it to sustain greater pressure from within the cell.
E) makes the animal more susceptible to circulatory disorders.
Answer: A
According to the fluid mosaic model of cell membranes, which of the following is a true statement about membrane phospholipids?
A) They can move laterally along the plane of the membrane.
B) They frequently flip-flop from one side of the membrane to the other.
C) They occur in an uninterrupted bilayer, with membrane proteins restricted to the surface of the membrane.
D) They are free to depart from the membrane and dissolve in the surrounding solution.
E) They have hydrophilic tails in the interior of the membrane.
Answer: A
Which of the following is one of the ways that the membranes of winter wheat are able to remain fluid when it is extremely cold?
A) by increasing the percentage of unsaturated phospholipids in the membrane
B) by increasing the percentage of cholesterol molecules in the membrane
C) by decreasing the number of hydrophobic proteins in the membrane
D) by cotransport of glucose and hydrogen
E) by using active transport
Answer: A
In order for a protein to be an integral membrane protein it would have to be
A) hydrophilic.
B) hydrophobic.
C) amphipathic, with at least one hydrophobic region.
D) completely covered with phospholipids.
E) exposed on only one surface of the membrane.
Answer: C
Which of the following is a reasonable explanation for why unsaturated fatty acids help keep any membrane more fluid at lower temperatures?
A) The double bonds form kinks in the fatty acid tails, preventing adjacent lipids from packing tightly.
B) Unsaturated fatty acids have a higher cholesterol content and therefore more cholesterol in membranes.
C) Unsaturated fatty acids are more polar than saturated fatty acids.
D) The double bonds block interaction among the hydrophilic head groups of the lipids.
E) The double bonds result in shorter fatty acid tails and thinner membranes.
Answer: A
Which of the following is true of integral membrane proteins?
A) They lack tertiary structure.
B) They are loosely bound to the surface of the bilayer.
C) They are usually transmembrane proteins.
D) They are not mobile within the bilayer.
E) They serve only a structural role in membranes.
Answer: C
The primary function of polysaccharides attached to the glycoproteins and glycolipids of animal cell membranes is
A) to facilitate diffusion of molecules down their concentration gradients.
B) to actively transport molecules against their concentration gradients.
C) to maintain the integrity of a fluid mosaic membrane.
D) to maintain membrane fluidity at low temperatures.
E) to mediate cell-to-cell recognition.
Answer: E
In a paramecium, cell surface integral membrane proteins are synthesized
A) in the cytoplasm by free ribosomes.
B) by ribosomes in the nucleus.
C) by ribosomes bound to the rough endoplasmic reticulum.
D) by ribosomes in the Golgi vesicles.
E) by ribosomes bound to the inner surface of the plasma membrane.
Answer: C
Which of the following is true of the evolution of cell membranes?
A) Cell membranes have stopped evolving now that they are fluid mosaics.
B) Cell membranes cannot evolve if the membrane proteins do not.
C) The evolution of cell membranes is driven by the evolution of glycoproteins and glycolipids.
D) All components of membranes evolve in response to natural selection.
E) An individual organism selects its preferred type of cell membrane for particular functions.
Answer: D
What kinds of molecules pass through a cell membrane most easily?
A) large and hydrophobic
B) small and hydrophobic
C) large polar
D) ionic
E) monosaccharides such as glucose
Answer: B
Which of the following is a characteristic feature of a carrier protein in a plasma membrane?
A) It is a peripheral membrane protein.
B) It exhibits a specificity for a particular type of molecule.
C) It requires the expenditure of cellular energy to function.
D) It works against diffusion.
E) It has few, if any, hydrophobic amino acids.
Answer: B
Nitrous oxide gas molecules diffusing across a cell's plasma membrane is an example of
A) diffusion across the lipid bilayer.
B) facilitated diffusion.
C) active transport.
D) osmosis.
E) cotransport.
Answer: A
Which of the following statements is correct about diffusion?
A) It is very rapid over long distances.
B) It requires an expenditure of energy by the cell.
C) It is a passive process in which molecules move from a region of higher concentration to a region of lower concentration.
D) It is an active process in which molecules move from a region of lower concentration to one of higher concentration.
E) It requires integral proteins in the cell membrane.
Answer: C
Celery stalks that are immersed in fresh water for several hours become stiff and hard. Similar stalks left in a 0.15 M salt solution become limp and soft. From this we can deduce that the cells of the celery stalks are
A) hypotonic to both fresh water and the salt solution.
B) hypertonic to both fresh water and the salt solution.
C) hypertonic to fresh water but hypotonic to the salt solution.
D) hypotonic to fresh water but hypertonic to the salt solution.
E) isotonic with fresh water but hypotonic to the salt solution.
Answer: C
Mammalian blood contains the equivalent of 0.15 M NaCl. Seawater contains the equivalent of 0.45 M NaCl. What will happen if red blood cells are transferred to seawater?
A) Water will leave the cells, causing them to shrivel and collapse.
B) NaCl will be exported from the red blood cells by facilitated diffusion.
C) The blood cells will take up water, swell, and eventually burst.
D) NaCl will passively diffuse into the red blood cells.
E) The blood cells will expend ATP for active transport of NaCl into the cytoplasm.
Answer: A
Which of the following statements correctly describes the normal tonicity conditions for typical plant and animal cells?
A) The animal cell is in a hypotonic solution, and the plant cell is in an isotonic solution.
B) The animal cell is in an isotonic solution, and the plant cell is in a hypertonic solution.
C) The animal cell is in a hypertonic solution, and the plant cell is in an isotonic solution.
D) The animal cell is in an isotonic solution, and the plant cell is in a hypotonic solution.
E) The animal cell is in a hypertonic solution, and the plant cell is in a hypotonic solution.
Answer: D
When a plant cell, such as one from a peony stem, is submerged in a very hypotonic solution, what is likely to occur?
A) The cell will burst.
B) The cell membrane will lyse.
C) Plasmolysis will shrink the interior.
D) The cell will become flaccid.
E) The cell will become turgid.
Answer: E
Which of the following membrane activities require energy from ATP hydrolysis?
A) facilitated diffusion of chloride ions across the membrane through a chloride channel
B) movement of water into a cell
C) Na⁺ ions moving out of a mammalian cell bathed in physiological saline
D) movement of glucose molecules into a bacterial cell from a medium containing a higher concentration of glucose than inside the cell
E) movement of carbon dioxide out of a paramecium
Answer: C
The phosphate transport system in bacteria imports phosphate into the cell even when the concentration of phosphate outside the cell is much lower than the cytoplasmic phosphate concentration. Phosphate import depends on a pH gradient across the membrane–more acidic outside the cell than inside the cell. Phosphate transport is an example of
A) passive diffusion.
B) facilitated diffusion.
C) active transport.
D) osmosis.
E) cotransport.
Answer: E
Glucose diffuses slowly through artificial phospholipid bilayers. The cells lining the small intestine, however, rapidly move large quantities of glucose from the glucose-rich food into their glucose-poor cytoplasm. Using this information, which transport mechanism is most probably functioning in the intestinal cells?
A) simple diffusion
B) phagocytosis
C) active transport pumps
D) exocytosis
E) facilitated diffusion
Answer: E
In most cells, there are electrochemical gradients of many ions across the plasma membrane even though there are usually only one or two electrogenic pumps present in the membrane. The gradients of the other ions are most likely accounted for by
A) cotransport proteins.
B) ion channels.
C) carrier proteins.
D) passive diffusion across the plasma membrane.
E) cellular metabolic reactions that create or destroy ions.
Answer: A
The sodium-potassium pump is called an electrogenic pump because it
A) pumps equal quantities of Na⁺ and K⁺ across the membrane.
B) pumps hydrogen ions out of the cell.
C) contributes to the membrane potential.
D) ionizes sodium and potassium atoms.
E) is used to drive the transport of other molecules against a concentration gradient.
Answer: C
Which of the following is most likely true of a protein that cotransports glucose and sodium ions into the intestinal cells of an animal?
A) The sodium ions are moving down their electrochemical gradient while glucose is moving up.
B) Glucose entering the cell along its concentration gradient provides energy for uptake of sodium ions against the electrochemical gradient.
C) Sodium ions can move down their electrochemical gradient through the cotransporter whether or not glucose is present outside the cell.
D) The cotransporter can also transport potassium ions.
E) A substance that blocks sodium ions from binding to the cotransport protein will also block the transport of glucose.
Answer: E
The movement of potassium into an animal cell requires
A) low cellular concentrations of sodium.
B) high cellular concentrations of potassium.
C) an energy source such as ATP.
D) a cotransport protein.
E) a potassium channel protein.
Answer: C
Ions diffuse across membranes through specific ion channels
A) down their chemical gradients.
B) down their concentration gradients.
C) down the electrical gradients.
D) down their electrochemical gradients.
E) down the osmotic potential gradients.
Answer: D
Which of the following would increase the electrochemical potential across a membrane?
A) a chloride channel
B) a sucrose-proton cotransporter
C) a proton pump
D) a potassium channel
E) both a proton pump and a potassium channel
Answer: C
The sodium-potassium pump in animal cells requires cytoplasmic ATP to pump ions across the plasma membrane. When the proteins of the pump are first synthesized in the rough ER, what side of the ER membrane will the ATP binding site be on?
A) It will be on the cytoplasmic side of the ER.
B) It will be on the side facing the interior of the ER.
C) It could be facing in either direction because proteins are properly reoriented in the Golgi apparatus.
D) It doesn't matter, because the pump is not active in the ER.
Answer: A
Proton pumps are used in various ways by members of every domain of organisms: Bacteria, Archaea, and Eukarya. What does this most probably mean?
A) Proton pumps must have evolved before any living organisms were present on Earth.
B) Proton gradients across a membrane were used by cells that were the common ancestor of all three domains of life.
C) The high concentration of protons in the ancient atmosphere must have necessitated a pump mechanism.
D) Cells of each domain evolved proton pumps independently when oceans became more acidic.
E) Proton pumps are necessary to all cell membranes.
Answer: B
The difference between pinocytosis and receptor-mediated endocytosis is that
A) pinocytosis brings only water molecules into the cell, but receptor-mediated endocytosis brings in other molecules as well.
B) pinocytosis increases the surface area of the plasma membrane whereas receptor-mediated endocytosis decreases the plasma membrane surface area.
C) pinocytosis is nonselective in the molecules it brings into the cell, whereas receptor-mediated endocytosis offers more selectivity.
D) pinocytosis requires cellular energy, but receptor-mediated endocytosis does not.
E) pinocytosis can concentrate substances from the extracellular fluid, but receptor-mediated endocytosis cannot.
Answer: C
In receptor-mediated endocytosis, receptor molecules initially project to the outside of the cell. Where do they end up after endocytosis?
A) on the outside of vesicles
B) on the inside surface of the cell membrane
C) on the inside surface of the vesicle
D) on the outer surface of the nucleus
E) on the ER
Answer: C
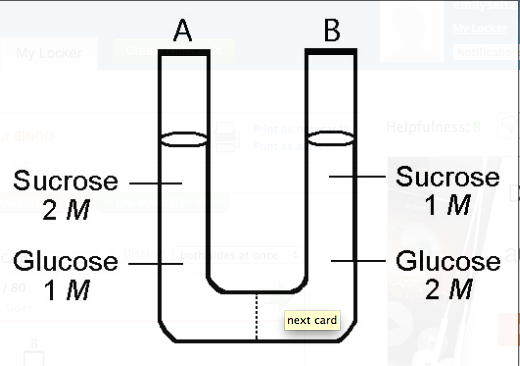
The solutions in the two arms of this U-tube are separated by a membrane that is permeable to water and glucose but not to sucrose. Side A is half-filled with a solution of 2 M sucrose and 1 M glucose. Side B is half-filled with 1 M sucrose and 2 M glucose. Initially, the liquid levels on both sides are equal.
Initially, in terms of tonicity, the solution in side A with respect to that in side B is
A) hypotonic.
B) plasmolyzed.
C) isotonic.
D) saturated.
E) hypertonic.
Answer: C
***After the system reaches equilibrium, what changes are observed?
A) The molarity of sucrose and glucose are equal on both sides.
B) The molarity of glucose is higher in side A than in side B.
C) The water level is higher in side A than in side B.
D) The water level is unchanged.
E) The water level is higher in side B than in side A.
Answer: C
In the small airways of the lung, a thin layer of liquid is needed between the epithelial cells and the mucus layer in order for cilia to beat and move the mucus and trapped particles out of the lung. One hypothesis is that the volume of this airway surface liquid is regulated osmotically by transport of sodium and chloride ions across the epithelial cell membrane. How would the lack of a functional chloride channel in cystic fibrosis patients affect sodium ion transport and the volume of the airway surface liquid?
A) Sodium ion transport will increase; higher osmotic potential will increase airway surface liquid volume.
B) Sodium ion transport will increase; higher osmotic potential will decrease airway surface liquid volume.
C) Sodium ion transport will decrease; lower osmotic potential will decrease airway surface liquid volume.
D) Sodium ion transport will decrease; lower osmotic potential will increase the airway surface liquid volume.
E) Sodium ion transport will be unaffected; lack of chloride transport still reduces osmotic potential and decreases the airway surface liquid volume.
Answer: C
A patient has had a serious accident and lost a lot of blood. In an attempt to replenish body fluids, distilled water–equal to the volume of blood lost–is transferred directly into one of his veins. What will be the most probable result of this transfusion?
A) It will have no unfavorable effect as long as the water is free of viruses and bacteria.
B) The patient's red blood cells will shrivel up because the blood fluid has become hypotonic compared to the cells.
C) The patient's red blood cells will swell because the blood fluid has become hypotonic compared to the cells.
D) The patient's red blood cells will shrivel up because the blood fluid has become hypertonic compared to the cells.
E) The patient's red blood cells will burst because the blood fluid has become hypertonic compared to the cells.
Answer: C
In what way do the membranes of a eukaryotic cell vary?
A) Phospholipids are found only in certain membranes.
B) Certain proteins are unique to each membrane.
C) Only certain membranes of the cell are selectively permeable.
D) Only certain membranes are constructed from amphipathic molecules.
E) Some membranes have hydrophobic surfaces exposed to the cytoplasm, while others have hydrophilic surfaces facing the cytoplasm.
Answer: B
According to the fluid mosaic model of membrane structure, proteins of the membrane are mostly
A) spread in a continuous layer over the inner and outer surfaces of the membrane.
B) confined to the hydrophobic interior of the membrane.
C) embedded in a lipid bilayer.
D) randomly oriented in the membrane, with no fixed inside-outside polarity.
E) free to depart from the fluid membrane and dissolve in the surrounding solution.
Answer: C
Which of the following factors would tend to increase membrane fluidity?
A) a greater proportion of unsaturated phospholipids
B) a greater proportion of saturated phospholipids
C) a lower temperature
D) a relatively high protein content in the membrane
E) a greater proportion of relatively large glycolipids compared with lipids having smaller molecular masses
Answer: A
Which of the following processes includes all others?
A) osmosis
B) diffusion of a solute across a membrane
C) facilitated diffusion
D) passive transport
E) transport of an ion down its electrochemical gradient
Answer: D
When chemical, transport, or mechanical work is done by an organism, what happens to the heat generated?
A) It is used to power yet more cellular work.
B) It is used to store energy as more ATP.
C) It is used to generate ADP from nucleotide precursors.
D) It is lost to the environment.
E) It is transported to specific organs such as the brain.
Answer: D
When ATP releases some energy, it also releases inorganic phosphate. What purpose does this serve (if any) in the cell?
A) The phosphate is released as an excretory waste.
B) The phosphate can only be used to regenerate more ATP.
C) The phosphate can be added to water and excreted as a liquid.
D) The phosphate may be incorporated into any molecule that contains phosphate.
E) It enters the nucleus to affect gene expression.
Answer:D
A number of systems for pumping ions across membranes are powered by ATP. Such ATP-powered pumps are often called ATPases although they don't often hydrolyze ATP unless they are simultaneously transporting ions. Because small increases in calcium ions in the cytosol can trigger a number of different intracellular reactions, cells keep the cytosolic calcium concentration quite low under normal conditions, using ATP-powered calcium pumps. For example, muscle cells transport calcium from the cytosol into the membranous system called the sarcoplasmic reticulum (SR). If a resting muscle cell's cytosol has a free calcium ion concentration of 10⁻⁷ while the concentration in the SR is 10⁻², then how is the ATPase acting?
A) ATPase activity must be powering an inflow of calcium from the outside of the cell into the SR.
B) ATPase activity must be transferring Pi to the SR to enable this to occur.
C) ATPase activity must be pumping calcium from the cytosol to the SR against the concentration gradient.
D) ATPase activity must be opening a channel for the calcium ions to diffuse back into the SR along the concentration gradient.
E) ATPase activity must be routing calcium ions from the SR to the cytosol, and then to the cell's environment.
Answer: C
What is the difference (if any) between the structure of ATP and the structure of the precursor of the A nucleotide in RNA?
A) The sugar molecule is different.
B) The nitrogen-containing base is different.
C) The number of phosphates is three instead of one.
D) The number of phosphates is three instead of two.
E) There is no difference.
Answer: E
The active site of an enzyme is the region that
A) binds allosteric regulators of the enzyme.
B) is involved in the catalytic reaction of the enzyme.
C) binds noncompetitive inhibitors of the enzyme.
D) is inhibited by the presence of a coenzyme or a cofactor.
Answer: B
According to the induced fit hypothesis of enzyme catalysis, which of the following is correct?
A) The binding of the substrate depends on the shape of the active site.
B) Some enzymes change their structure when activators bind to the enzyme.
C) A competitive inhibitor can outcompete the substrate for the active site.
D) The binding of the substrate changes the shape of the enzyme's active site.
E) The active site creates a microenvironment ideal for the reaction.
Answer: D
Mutations that result in single amino acid substitutions in an enzyme
A) can have no effect on the activity or properties of the enzyme.
B) will almost always destroy the activity of the enzyme.
C) will often cause a change in the substrate specificity of the enzyme.
D) may affect the physicochemical properties of the enzyme such as its optimal temperature and pH.
E) may, in rare cases, cause the enzyme to run reactions in reverse.
Answer: D
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following?
A) denaturization of the enzyme
B) allosteric inhibition
C) competitive inhibition
D) saturation of the enzyme activity
E) insufficient cofactors
Answer: C
Zinc, an essential trace element for most organisms, is present in the active site of the enzyme carboxypeptidase. The zinc most likely functions as a(n)
A) competitive inhibitor of the enzyme.
B) noncompetitive inhibitor of the enzyme.
C) allosteric activator of the enzyme.
D) cofactor necessary for enzyme activity.
E) coenzyme derived from a vitamin.
Answer: D
In order to attach a particular amino acid to the tRNA molecule that will transport it, an enzyme, an aminoacyl-tRNA synthetase, is required, along with ATP. Initially, the enzyme has an active site for ATP and another for the amino acid, but it is not able to attach the tRNA. What must occur in order for the final attachment to occur?
A) The ATP must first have to attach to the tRNA.
B) The binding of the first two molecules must cause a 3-D change that opens another active site on the enzyme.
C) The ATP must be hydrolyzed to allow the amino acid to bind to the synthetase.
D) The tRNA molecule must have to alter its shape in order to be able to fit into the active site with the other two molecules.
E) The 3' end of the tRNA must have to be cleaved before it can have an attached amino acid.
Answer: B
Some of the drugs used to treat HIV patients are competitive inhibitors of the HIV reverse transcriptase enzyme. Unfortunately, the high mutation rate of HIV means that the virus rapidly acquires mutations with amino acid changes that make them resistant to these competitive inhibitors. Where in the reverse transcriptase enzyme would such amino acid changes most likely occur in drug-resistant viruses?
A) in or near the active site
B) at an allosteric site
C) at a cofactor binding site
D) in regions of the protein that determine packaging into the virus capsid
E) such mutations could occur anywhere with equal probability
Answer: A
Protein kinases are enzymes that transfer the terminal phosphate from ATP to an amino acid residue on the target protein. Many are located on the plasma membrane as integral membrane proteins or peripheral membrane proteins. What purpose may be served by their plasma membrane localization?
A) ATP is more abundant near the plasma membrane.
B) They can more readily encounter and phosphorylate other membrane proteins.
C) Membrane localization lowers the activation energy of the phosphorylation reaction.
D) They flip back and forth across the membrane to access target proteins on either side.
E) They require phospholipids as a cofactor.
Answer: B
How does a noncompetitive inhibitor decrease the rate of an enzyme reaction?
A) by binding at the active site of the enzyme
B) by changing the shape of the enzyme's active site
C) by changing the free energy change of the reaction
D) by acting as a coenzyme for the reaction
E) by decreasing the activation energy of the reaction
Answer: B
The mechanism in which the end product of a metabolic pathway inhibits an earlier step in the pathway is most precisely described as
A) metabolic inhibition.
B) feedback inhibition.
C) allosteric inhibition.
D) noncooperative inhibition.
E) reversible inhibition.
Answer: B
Which of the following statements describes enzyme cooperativity?
A) A multienzyme complex contains all the enzymes of a metabolic pathway.
B) A product of a pathway serves as a competitive inhibitor of an early enzyme in the pathway.
C) A substrate molecule bound to an active site of one subunit promotes substrate binding to the active site of other subunits.
D) Several substrate molecules can be catalyzed by the same enzyme.
E) A substrate binds to an active site and inhibits cooperation between enzymes in a pathway.
Answer: C
Allosteric enzyme regulation is usually associated with
A) lack of cooperativity.
B) feedback inhibition.
C) activating activity.
D) an enzyme with more than one subunit.
E) the need for cofactors.
Answer: D
Which of the following is an example of cooperativity?
A) the binding of an end product of a metabolic pathway to the first enzyme that acts in the pathway
B) one enzyme in a metabolic pathway passing its product to act as a substrate for the next enzyme in the pathway
C) a molecule binding at one unit of a tetramer, allowing faster binding at each of the other three
D) the effect of increasing temperature on the rate of an enzymatic reaction
E) binding of an ATP molecule along with one of the substrate molecules in an active site
Answer: C
Protein kinases are enzymes that catalyze phosphorylation of target proteins at specific sites, whereas protein phosphatases catalyze removal of phosphate(s) from phosphorylated proteins. Phosphorylation and dephosphorylation can function as an on-off switch for a protein's activity, most likely through
A) the change in a protein's charge leading to a conformational change.
B) the change in a protein's charge leading to cleavage.
C) a change in the optimal pH at which a reaction will occur.
D) a change in the optimal temperature at which a reaction will occur.
E) the excision of one or more peptides.
Answer: A
Besides turning enzymes on or off, what other means does a cell use to control enzymatic activity?
A) cessation of cellular protein synthesis
B) localization of enzymes into specific organelles or membranes
C) exporting enzymes out of the cell
D) connecting enzymes into large aggregates
E) hydrophobic interactions
Answer: B
In experimental tests of enzyme evolution, where a gene encoding an enzyme is subjected to multiple cycles of random mutagenesis and selection for altered substrate specificity, the resulting enzyme had multiple amino acid changes associated with altered substrate specificity. Where in the enzyme were these amino acid changes located?
A) only in the active site
B) only in the active site or near the active site
C) in or near the active site and at surface sites away from the active site
D) only at surface sites away from the active site
E) only in the hydrophobic interior of the folded protein
Answer: C
How might an amino acid change at a site distant from the active site of the enzyme alter the enzyme's substrate specificity?
A) by changing the enzyme's stability
B) by changing the enzyme's location in the cell
C) by changing the shape of the protein
D) by changing the enzyme's pH optimum
E) an amino acid change away from the active site cannot alter the enzyme's substrate specificity
Answer: C
Succinate dehydrogenase catalyzes the conversion of succinate to fumarate. The reaction is inhibited by malonic acid, which resembles succinate but cannot be acted upon by succinate dehydrogenase. Increasing the ratio of succinate to malonic acid reduces the inhibitory effect of malonic acid.
Based on this information, which of the following is correct?
A) Succinate dehydrogenase is the enzyme, and fumarate is the substrate.
B) Succinate dehydrogenase is the enzyme, and malonic acid is the substrate.
C) Succinate is the substrate, and fumarate is the product.
D) Fumarate is the product, and malonic acid is a noncompetitive inhibitor.
E) Malonic acid is the product, and fumarate is a competitive inhibitor.
Answer: C
Succinate dehydrogenase catalyzes the conversion of succinate to fumarate. The reaction is inhibited by malonic acid, which resembles succinate but cannot be acted upon by succinate dehydrogenase. Increasing the ratio of succinate to malonic acid reduces the inhibitory effect of malonic acid.
What is malonic acid's role with respect to succinate dehydrogenase?
A) It is a competitive inhibitor.
B) It blocks the binding of fumarate.
C) It is a noncompetitive inhibitor.
D) It is able to bind to succinate.
E) It is an allosteric regulator.
Answer: A
A series of enzymes catalyze the reaction X → Y → Z → A. Product A binds to the enzyme that converts X to Y at a position remote from its active site. This binding decreases the activity of the enzyme.
What is substance X?
A) a coenzyme
B) an allosteric inhibitor
C) a substrate
D) an intermediate
E) the product
Answer: C
A series of enzymes catalyze the reaction X → Y → Z → A. Product A binds to the enzyme that converts X to Y at a position remote from its active site. This binding decreases the activity of the enzyme.
With respect to the enzyme that converts X to Y, substance A functions as
A) a coenzyme.
B) an allosteric inhibitor.
C) the substrate.
D) an intermediate.
E) a competitive inhibitor.
Answer: B
Choose the pair of terms that correctly completes this sentence: Catabolism is to anabolism as ________ is to ________.
A) exergonic; spontaneous
B) exergonic; endergonic
C) free energy; entropy
D) work; energy
E) entropy; enthalpy
ANswer: B
What is the term for metabolic pathways that release stored energy by breaking down complex molecules?
A) anabolic pathways
B) catabolic pathways
C) fermentation pathways
D) thermodynamic pathways
E) bioenergetic pathways
Answer: B
Why does the oxidation of organic compounds by molecular oxygen to produce CO₂ and water release free energy?
A) The covalent bonds in organic molecules and molecular oxygen have more kinetic energy than the covalent bonds in water and carbon dioxide.
B) Electrons are being moved from atoms that have a lower affinity for electrons (such as C) to atoms with a higher affinity for electrons (such as O).
C) The oxidation of organic compounds can be used to make ATP.
D) The electrons have a higher potential energy when associated with water and CO₂ than they do in organic compounds.
E) The covalent bond in O₂ is unstable and easily broken by electrons from organic molecules.
Answer: B
Which of the following statements describes NAD⁺?
A) NAD⁺ is reduced to NADH during glycolysis, pyruvate oxidation, and the citric acid cycle.
B) NAD⁺ has more chemical energy than NADH.
C) NAD⁺ is oxidized by the action of hydrogenases.
D) NAD⁺ can donate electrons for use in oxidative phosphorylation.
E) In the absence of NAD⁺, glycolysis can still function.
Answer: A
The oxygen consumed during cellular respiration is involved directly in which process or event?
A) glycolysis
B) accepting electrons at the end of the electron transport chain
C) the citric acid cycle
D) the oxidation of pyruvate to acetyl CoA
E) the phosphorylation of ADP to form ATP
Answer: B
An electron loses potential energy when it
A) shifts to a less electronegative atom.
B) shifts to a more electronegative atom.
C) increases its kinetic energy.
D) increases its activity as an oxidizing agent.
E) moves further away from the nucleus of the atom.
Answer: B
The transport of pyruvate into mitochondria depends on the proton-motive force across the inner mitochondrial membrane. How does pyruvate enter the mitochondrion?
A) active transport
B) diffusion
C) facilitated diffusion
D) through a channel
E) through a pore
Answer: A
During cellular respiration, acetyl CoA accumulates in which location?
A) cytosol
B) mitochondrial outer membrane
C) mitochondrial inner membrane
D) mitochondrial intermembrane space
E) mitochondrial matrix
Answer: E
A young animal has never had much energy. He is brought to a veterinarian for help and is sent to the animal hospital for some tests. There they discover his mitochondria can use only fatty acids and amino acids for respiration, and his cells produce more lactate than normal. Of the following, which is the best explanation of his condition?
A) His mitochondria lack the transport protein that moves pyruvate across the outer mitochondrial membrane.
B) His cells cannot move NADH from glycolysis into the mitochondria.
C) His cells contain something that inhibits oxygen use in his mitochondria.
D) His cells lack the enzyme in glycolysis that forms pyruvate.
E) His cells have a defective electron transport chain, so glucose goes to lactate instead of to acetyl CoA.
Answer: A
During aerobic respiration, electrons travel downhill in which sequence?
A) food → citric acid cycle → ATP → NAD⁺
B) food → NADH → electron transport chain → oxygen
C) glucose → pyruvate → ATP → oxygen
D) glucose → ATP → electron transport chain → NADH
E) food → glycolysis → citric acid cycle → NADH → ATP
Answer: B
Where are the proteins of the electron transport chain located?
A) cytosol
B) mitochondrial outer membrane
C) mitochondrial inner membrane
D) mitochondrial intermembrane space
E) mitochondrial matrix
Answer: C
In cellular respiration, the energy for most ATP synthesis is supplied by
A) high energy phosphate bonds in organic molecules.
B) a proton gradient across a membrane.
C) converting oxygen to ATP.
D) transferring electrons from organic molecules to pyruvate.
E) generating carbon dioxide and oxygen in the electron transport chain.
Answer: B
During aerobic respiration, which of the following directly donates electrons to the electron transport chain at the lowest energy level?
A) NAD+
B) NADH
C) ATP
D) ADP + Pi
E) FADH2
Answer: E
Inside an active mitochondrion, most electrons follow which pathway?
A) glycolysis → NADH → oxidative phosphorylation → ATP → oxygen
B) citric acid cycle → FADH₂ → electron transport chain → ATP
C) electron transport chain → citric acid cycle → ATP → oxygen
D) pyruvate → citric acid cycle → ATP → NADH → oxygen
E) citric acid cycle → NADH → electron transport chain → oxygen
Answer: E
In chemiosmotic phosphorylation, what is the most direct source of energy that is used to convert ADP + Pi to ATP?
A) energy released as electrons flow through the electron transport system
B) energy released from substrate-level phosphorylation
C) energy released from movement of protons through ATP synthase, against the electrochemical gradient
D) energy released from movement of protons through ATP synthase, down the electrochemical gradient
E) No external source of energy is required because the reaction is exergonic.
Answer: D
Energy released by the electron transport chain is used to pump H⁺ into which location in eukaryotic cells?
A) cytosol
B) mitochondrial outer membrane
C) mitochondrial inner membrane
D) mitochondrial intermembrane space
E) mitochondrial matrix
Answer: D
The direct energy source that drives ATP synthesis during respiratory oxidative phosphorylation in eukaryotic cells is
A) oxidation of glucose to CO₂ and water.
B) the thermodynamically favorable flow of electrons from NADH to the mitochondrial electron transport carriers.
C) the final transfer of electrons to oxygen.
D) the proton-motive force across the inner mitochondrial membrane.
E) the thermodynamically favorable transfer of phosphate from glycolysis and the citric acid cycle intermediate molecules of ADP.
Answer: D
When hydrogen ions are pumped from the mitochondrial matrix across the inner membrane and into the intermembrane space, the result is the
A) formation of ATP.
B) reduction of NAD⁺.
C) restoration of the Na⁺/K⁺ balance across the membrane.
D) creation of a proton-motive force.
E) lowering of pH in the mitochondrial matrix.
Answer: D
Chemiosmotic ATP synthesis (oxidative phosphorylation) occurs in
A) all cells, but only in the presence of oxygen.
B) only eukaryotic cells, in the presence of oxygen.
C) only in mitochondria, using either oxygen or other electron acceptors.
D) all respiring cells, both prokaryotic and eukaryotic, using either oxygen or other electron acceptors.
E) all cells, in the absence of respiration.
Answer: D
What is proton-motive force?
A) the force required to remove an electron from hydrogen
B) the force exerted on a proton by a transmembrane proton concentration gradient
C) the force that moves hydrogen into the intermembrane space
D) the force that moves hydrogen into the mitochondrion
E) the force that moves hydrogen to NAD⁺
Answer: B
Phosphofructokinase is an important control enzyme in the regulation of cellular respiration. Which of the following statements correctly describes phosphofructokinase activity?
A) It is inhibited by AMP.
B) It is activated by ATP.
C) It is activated by citrate, an intermediate of the citric acid cycle.
D) It catalyzes the conversion of fructose 1,6-bisphosphate to fructose 6-phosphate, an early step of glycolysis.
E) It is an allosteric enzyme.
Answer: E
Phosphofructokinase is an allosteric enzyme that catalyzes the conversion of fructose 6-phosphate to fructose 1,6-bisphosphate, an early step of glycolysis. In the presence of oxygen, an increase in the amount of ATP in a cell would be expected to
A) inhibit the enzyme and thus slow the rates of glycolysis and the citric acid cycle.
B) activate the enzyme and thus slow the rates of glycolysis and the citric acid cycle.
C) inhibit the enzyme and thus increase the rates of glycolysis and the citric acid cycle.
D) activate the enzyme and increase the rates of glycolysis and the citric acid cycle.
E) inhibit the enzyme and thus increase the rate of glycolysis and the concentration of citrate.
Answer: A
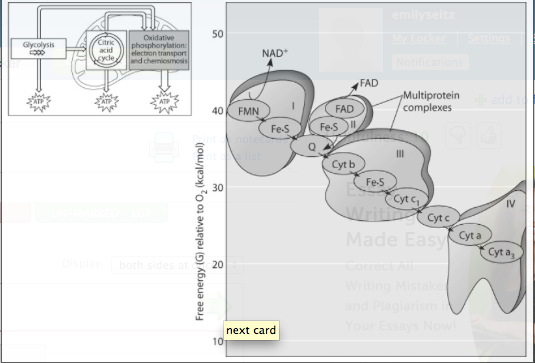
igure 9.3 shows the electron transport chain. Which of the following is the combination of substances that is initially added to the chain?
A) oxygen, carbon dioxide, and water
B) NAD⁺, FAD, and electrons
C) NADH, FADH₂, and protons
D) NADH, FADH₂, and O₂
E) oxygen and protons
Answer: D
***Which of the following most accurately describes what is happening along the electron transport chain in Figure 9.3?
A) Chemiosmosis is coupled with electron transfer.
B) Each electron carrier alternates between being reduced and being oxidized.
C) ATP is generated at each step.
D) Energy of the electrons increases at each step.
E) Molecules in the chain give up some of their potential energy.
Answer: B
***Which of the protein complexes labeled with Roman numerals in Figure 9.3 will transfer electrons to O₂?
A) complex I
B) complex II
C) complex III
D) complex IV
E) All of the complexes can transfer electrons to O₂.
Answer: D
What happens at the end of the chain in Figure 9.3?
A) 2 electrons combine with a proton and a molecule of NAD⁺.
B) 2 electrons combine with a molecule of oxygen and two hydrogen atoms.
C) 4 electrons combine with a molecule of oxygen and 4 protons.
D) 4 electrons combine with four hydrogen and two oxygen atoms.
E) 1 electron combines with a molecule of oxygen and a hydrogen atom.
Answer: C
Exposing inner mitochondrial membranes to ultrasonic vibrations will disrupt the membranes. However, the fragments will reseal "inside out." These little vesicles that result can still transfer electrons from NADH to oxygen and synthesize ATP. If the membranes are agitated further, however, the ability to synthesize ATP is lost.
After the first disruption, when electron transfer and ATP synthesis still occur, what must be present?
A) all of the electron transport proteins as well as ATP synthase
B) all of the electron transport system and the ability to add CoA to acetyl groups
C) the ATP synthase system
D) the electron transport system
E) plasma membranes like those bacteria use for respiration
Answer: A
The immediate energy source that drives ATP synthesis by ATP synthase during oxidative phosphorylation is the
A) oxidation of glucose and other organic compounds.
B) flow of electrons down the electron transport chain.
C) affinity of oxygen for electrons.
D) H⁺ concentration across the membrane holding ATP synthase.
E) transfer of phosphate to ADP.
Answer: D
The final electron acceptor of the electron transport chain that functions in aerobic oxidative phosphorylation is
A) oxygen.
B) water.
C) NAD⁺.
D) pyruvate.
E) ADP.
Answer: A
When electrons flow along the electron transport chains of mitochondria, which of the following changes occurs?
A) The pH of the matrix increases.
B) ATP synthase pumps protons by active transport.
C) The electrons gain free energy.
D) The cytochromes phosphorylate ADP to form ATP.
E) NAD⁺ is oxidized.
Answer: A
Which of the following are products of the light reactions of photosynthesis that are utilized in the Calvin cycle?
A) CO₂ and glucose
B) H₂O and O₂
C) ADP, Pi, and NADP⁺
D) electrons and H⁺
E) ATP and NADPH
Answer: E
Where does the Calvin cycle take place?
A) stroma of the chloroplast
B) thylakoid membrane
C) cytoplasm surrounding the chloroplast
D) interior of the thylakoid (thylakoid space)
E) outer membrane of the chloroplast
Answer: A
In autotrophic bacteria, where are the enzymes located that can carry on carbon fixation (reduction of carbon dioxide to carbohydrate)?
A) in chloroplast membranes
B) in chloroplast stroma
C) in the cytosol
D) in the nucleoid
E) in the infolded plasma membrane
Answer: C
When oxygen is released as a result of photosynthesis, it is a direct by-product of
A) reducing NADP⁺.
B) splitting water molecules.
C) chemiosmosis.
D) the electron transfer system of photosystem I.
E) the electron transfer system of photosystem II.
Answer: B
In the thylakoid membranes, what is the main role of the antenna pigment molecules?
A) split water and release oxygen to the reaction-center chlorophyll
B) harvest photons and transfer light energy to the reaction-center chlorophyll
C) synthesize ATP from ADP and Pi
D) transfer electrons to ferredoxin and then NADPH
E) concentrate photons within the stroma
Answer: B
Which of the events listed below occurs in the light reactions of photosynthesis?
A) NADP is produced.
B) NADPH is reduced to NADP⁺.
C) Carbon dioxide is incorporated into PGA.
D) ATP is phosphorylated to yield ADP.
E) Light is absorbed and funneled to reaction-center chlorophyll a.
Answer: E
Which statement describes the functioning of photosystem II?
A) Light energy excites electrons in the thylakoid membrane electron transport chain.
B) Photons are passed along to a reaction-center chlorophyll.
C) The P680 chlorophyll donates a pair of protons to NADP⁺, which is thus converted to NADPH.
D) The electron vacancies in P680⁺ are filled by electrons derived from water.
E) The splitting of water yields molecular carbon dioxide as a by-product.
Answer: D
Which of the following are directly associated with photosystem I?
A) harvesting of light energy by ATP
B) receiving electrons from the thylakoid membrane electron transport chain
C) generation of molecular oxygen
D) extraction of hydrogen electrons from the splitting of water
E) passing electrons to the thylakoid membrane electron transport chain
Answer: B
Some photosynthetic organisms contain chloroplasts that lack photosystem II, yet are able to survive. The best way to detect the lack of photosystem II in these organisms would be
A) to determine if they have thylakoids in the chloroplasts.
B) to test for liberation of O₂ in the light.
C) to test for CO₂ fixation in the dark.
D) to do experiments to generate an action spectrum.
E) to test for production of either sucrose or starch.
Answer: B
What are the products of linear photophosphorylation?
A) heat and fluorescence
B) ATP and P700
C) ATP and NADPH
D) ADP and NADP
E) P700 and P680
Answer: C
As a research scientist, you measure the amount of ATP and NADPH consumed by the Calvin cycle in 1 hour. You find 30,000 molecules of ATP consumed, but only 20,000 molecules of NADPH. Where did the extra ATP molecules come from?
A) photosystem II
B) photosystem I
C) cyclic electron flow
D) linear electron flow
E) chlorophyll
Answer: C
Assume a thylakoid is somehow punctured so that the interior of the thylakoid is no longer separated from the stroma. This damage will have the most direct effect on which of the following processes?
A) the splitting of water
B) the absorption of light energy by chlorophyll
C) the flow of electrons from photosystem II to photosystem I
D) the synthesis of ATP
E) the reduction of NADP⁺
Answer: D
What does the chemiosmotic process in chloroplasts involve?
A) establishment of a proton gradient across the thylakoid membrane
B) diffusion of electrons through the thylakoid membrane
C) reduction of water to produce ATP energy
D) movement of water by osmosis into the thylakoid space from the stroma
E) formation of glucose, using carbon dioxide, NADPH, and ATP
Answer: A
In mitochondria, chemiosmosis translocates protons from the matrix into the intermembrane space, whereas in chloroplasts, chemiosmosis translocates protons from
A) the stroma to the photosystem II.
B) the matrix to the stroma.
C) the stroma to the thylakoid space.
D) the intermembrane space to the matrix.
E) the thylakoid space to the stroma.
Answer: C
Where are the molecules of the electron transport chain found in plant cells?
A) thylakoid membranes of chloroplasts
B) stroma of chloroplasts
C) outer membrane of mitochondria
D) matrix of mitochondria
E) cytoplasm
Answer: A
Reduction of NADP⁺ occurs during
A) photosynthesis.
B) respiration.
C) both photosynthesis and respiration.
D) neither photosynthesis nor respiration.
E) photorespiration.
Answer:A
The splitting of carbon dioxide to form oxygen gas and carbon compounds occurs during
A) photosynthesis.
B) respiration.
C) both photosynthesis and respiration.
D) neither photosynthesis nor respiration.
E) photorespiration.
Answer: D
P680⁺ is said to be the strongest biological oxidizing agent. Why?
A) It is the receptor for the most excited electron in either photosystem.
B) It is the molecule that transfers electrons to plastoquinone (Pq) of the electron transfer system.
C) It transfers its electrons to reduce NADP⁺ to NADPH.
D) This molecule has a stronger attraction for electrons than oxygen, to obtain electrons from water.
E) It has a positive charge.
Answer: D
Some photosynthetic bacteria (e.g., purple sulfur bacteria) have only photosystem I, whereas others (e.g., cyanobacteria) have both photosystem I and photosystem II. Which of the following might this observation imply?
A) Photosystem II was selected against in some species.
B) Photosynthesis with only photosystem I is more ancestral.
C) Photosystem II may have evolved to be more photoprotective.
D) Linear electron flow is more primitive than cyclic flow of electrons.
E) Cyclic flow is more necessary than linear electron flow.
ANswer: B
electron flow may be photoprotective (protective to light-induced damage). Which of the following experiments could provide information on this phenomenon?
A) use mutated organisms that can grow but that cannot carry out cyclic flow of electrons and compare their abilities to photosynthesize in different light intensities against those of wild-type organisms
B) use plants that can carry out both linear and cyclic electron flow, or only one or another of these processes, and compare their light absorbance at different wavelengths and different light intensities
C) use bacteria that have only cyclic flow and look for their frequency of mutation damage at different light intensities
D) use bacteria with only cyclic flow and measure the number and types of photosynthetic pigments they have in their membranes
E) use plants with only photosystem I operative and measure how much damage occurs at different wavelengths
Answer: A
In metabolic processes of cell respiration and photosynthesis, prosthetic groups such as heme and iron-sulfur complexes are encountered in components of the electron transport chain. What do they do?
A) donate electrons
B) act as reducing agents
C) act as oxidizing agents
D) transport protons within the mitochondria and chloroplasts
E) both oxidize and reduce during electron transport
ANswer: E
In a cyanobacterium, the reactions that produce NADPH occur in
A) the light reactions alone.
B) the Calvin cycle alone.
C) both the light reactions and the Calvin cycle.
D) neither the light reactions nor the Calvin cycle.
E) the chloroplast, but is not part of photosynthesis.
Answer: A
The reactions that produce molecular oxygen (O₂) take place in
A) the light reactions alone.
B) the Calvin cycle alone.
C) both the light reactions and the Calvin cycle.
D) neither the light reactions nor the Calvin cycle.
E) the chloroplast, but are not part of photosynthesis.
Answer: A
The accumulation of free oxygen in Earth's atmosphere began
A) with the origin of life and respiratory metabolism.
B) with the origin of photosynthetic bacteria that had photosystem I.
C) with the origin of cyanobacteria that had both photosystem I and photosystem II.
D) with the origin of chloroplasts in photosynthetic eukaryotic algae.
E) with the origin of land plants.
Answer: C
A flask containing photosynthetic green algae and a control flask containing water with no algae are both placed under a bank of lights, which are set to cycle between 12 hours of light and 12 hours of dark. The dissolved oxygen concentrations in both flasks are monitored. Predict what the relative dissolved oxygen concentrations will be in the flask with algae compared to the control flask.
A) The dissolved oxygen in the flask with algae will always be higher.
B) The dissolved oxygen in the flask with algae will always be lower.
C) The dissolved oxygen in the flask with algae will be higher in the light, but the same in the dark.
D) The dissolved oxygen in the flask with algae will be higher in the light, but lower in the dark.
E) The dissolved oxygen in the flask with algae will not be different from the control flask at any time.
Answer: D
Where do the enzymatic reactions of the Calvin cycle take place?
A) stroma of the chloroplast
B) thylakoid membranes
C) matrix of the mitochondria
D) cytosol around the chloroplast
E) thylakoid space
Answer: A
What is the primary function of the Calvin cycle?
A) use ATP to release carbon dioxide
B) use NADPH to release carbon dioxide
C) split water and release oxygen
D) transport RuBP out of the chloroplast
E) synthesize simple sugars from carbon dioxide
Answer: E
The NADPH required for the Calvin cycle comes from
A) reactions initiated in photosystem I.
B) reactions initiated in photosystem II.
C) the citric acid cycle.
D) glycolysis.
E) oxidative phosphorylation.
Answer: A
Reactions that require CO₂ take place in
A) the light reactions alone.
B) the Calvin cycle alone.
C) both the light reactions and the Calvin cycle.
D) neither the light reactions nor the Calvin cycle.
E) the chloroplast, but is not part of photosynthesis.
Answer: B
Which of the following statements best represents the relationships between the light reactions and the Calvin cycle?
A) The light reactions provide ATP and NADPH to the Calvin cycle, and the cycle returns ADP, Pi, and NADP⁺ to the light reactions.
B) The light reactions provide ATP and NADPH to the carbon fixation step of the Calvin cycle, and the cycle provides water and electrons to the light reactions.
C) The light reactions supply the Calvin cycle with CO₂ to produce sugars, and the Calvin cycle supplies the light reactions with sugars to produce ATP.
D) The light reactions provide the Calvin cycle with oxygen for electron flow, and the Calvin cycle provides the light reactions with water to split.
E) There is no relationship between the light reactions and the Calvin cycle.
Answer: A
Three "turns" of the Calvin cycle generate a "surplus" molecule of glyceraldehyde 3-phosphate (G3P). Which of the following is a consequence of this?
A) Formation of a molecule of glucose would require nine "turns."
B) G3P more readily forms sucrose and other disaccharides than it does monosaccharides.
C) Some plants would not taste sweet to us.
D) The formation of sucrose and starch in plants involves assembling G3P molecules, with or without further rearrangements.
E) Plants accumulate and store G3P.
Answer: D
In the process of carbon fixation, RuBP attaches a CO₂ to produce a six-carbon molecule, which is then split to produce two molecules of 3-phosphoglycerate. After phosphorylation and reduction produces glyceraldehyde 3-phosphate (G3P), what more needs to happen to complete the Calvin cycle?
A) addition of a pair of electrons from NADPH
B) inactivation of RuBP carboxylase enzyme
C) regeneration of ATP from ADP
D) regeneration of RuBP
E) regeneration of NADP⁺
ANswer: D
The phylogenetic distribution of the enzyme rubisco is limited to
A) C₃ plants only.
B) C₃ and C₄ plants.
C) all photosynthetic eukaryotes.
D) all known photoautotrophs, both bacterial and eukaryotic.
E) all living cells.
Answer: D
Photorespiration occurs when rubisco reacts RuBP with
A) CO₂.
B) O₂.
C) glyceraldehyde 3-phosphate.
D) 3-phosphoglycerate.
E) NADPH.
ANswer: B
Plants photosynthesize only in the light. Plants respire
A) in the dark only.
B) in the light only.
C) both in light and dark.
D) never–they get their ATP from photophosphorylation.
E) only when excessive light energy induces photorespiration.
Answer: C
If the carbon atom of the incoming CO₂ molecule is labeled with a radioactive isotope of carbon, which organic molecules will be radioactively labeled after one cycle?
A) C only
B) B, C, D, and E
C) C, D, and E only
D) B and C only
E) B and D only
Answer: B
***If ATP used by this plant is labeled with radioactive phosphorus, which molecule or molecules of the Calvin cycle will be radioactively labeled first?
A) B only
B) B and C only
C) B, C, and D only
D) B and E only
E) B, C, D, and E
Answer: D
***Which molecule(s) of the Calvin cycle is (are) also found in glycolysis?
A) B, C, E, and 3-phosphoglycerate
B) B, C, and E only
C) 3-phosphoglycerate only
D) B, C, D, and 3-phosphoglycerate only
E) E only
Answer: D
To identify the molecule that accepts CO₂, Calvin and Benson manipulated the carbon-fixation cycle by either cutting off CO₂ or cutting off light from cultures of photosynthetic algae. They then measured the concentrations of various metabolites immediately following the manipulation. How would these experiments help identify the CO₂ acceptor? Study Figure 10.2 to help you in determining the correct answer.
A) The CO₂ acceptor concentration would decrease when either the CO₂ or light are cut off.
B) The CO₂ acceptor concentration would increase when either the CO₂ or light are cut off.
C) The CO₂ acceptor concentration would increase when the CO₂ is cut off, but decrease when the light is cut off.
D) The CO₂ acceptor concentration would decrease when the CO₂ is cut off, but increase when the light is cut off.
E) The CO₂ acceptor concentration would stay the same regardless of the CO₂ or light.
Answer: C
The light reactions of photosynthesis supply the Calvin cycle with
A) light energy.
B) CO₂ and ATP.
C) H₂O and NADPH.
D) ATP and NADPH.
E) sugar and O₂.
Answer: D
Which of the following sequences correctly represents the flow of electrons during photosynthesis?
A) NADPH → O₂ → CO₂
B) H₂O → NADPH → Calvin cycle
C) NADPH → chlorophyll → Calvin cycle
D) H₂O → photosystem I → photosystem II
E) NADPH → electron transport chain → O₂
Answer: B
Which of the following does not occur during the Calvin cycle?
A) carbon fixation
B) oxidation of NADPH
C) release of oxygen
D) regeneration of the CO₂ acceptor
E) consumption of ATP
Answer: C
Which process is most directly driven by light energy?
A) creation of a pH gradient by pumping protons across the thylakoid membrane
B) carbon fixation in the stroma
C) reduction of NADP⁺ molecules
D) removal of electrons from chlorophyll molecules
E) ATP synthesis
Answer:D