Name the enzyme classes


What is this reaction enzyme
oxioreductase
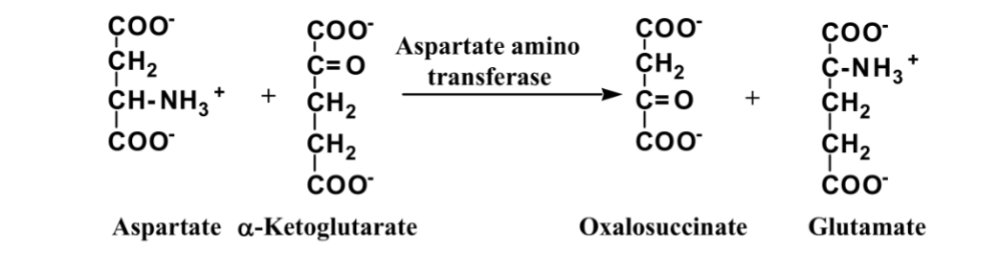
What is this reaction enzyme
transferase

What is this reaction enzyme
hydrolase
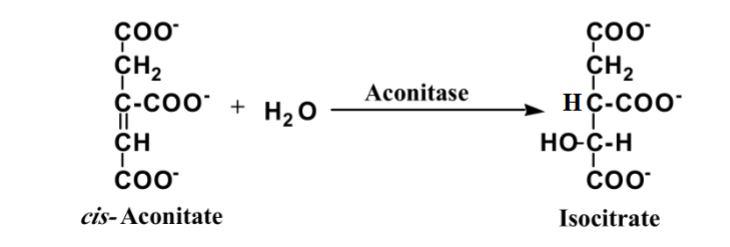
What is this reaction enzyme
Lyase
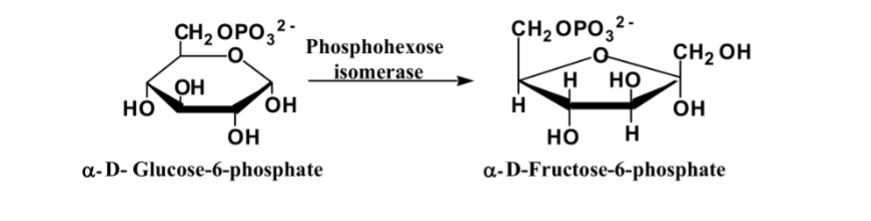
What is this reaction enzyme
Isomerase
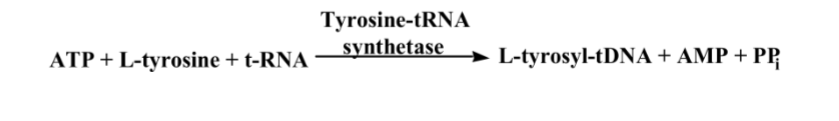
What is this reaction enzyme
Ligase
What is a reaction intermediate
transient chemical species formed and decayed in reactions
Define rate limiting step
step with highest activation energy in a reaction
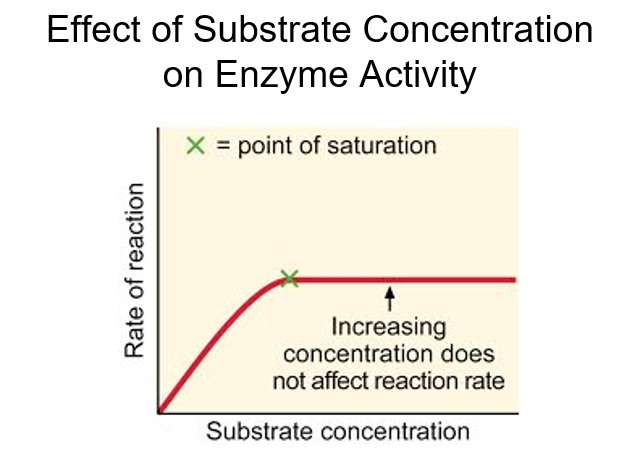
Explain effect of substrate concentration on enzyme activity
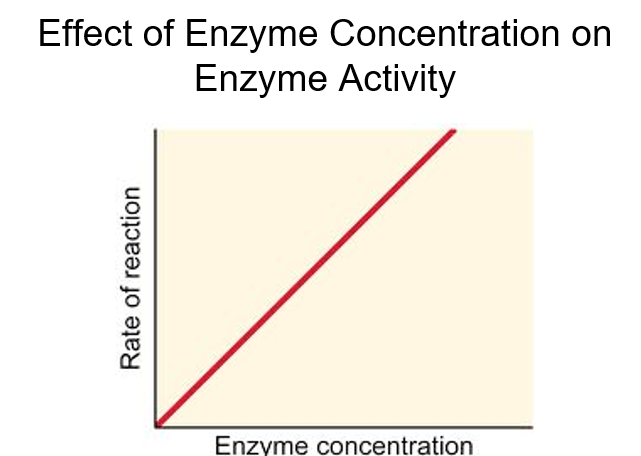
Explain effect of enzyme concentration on enzyme activity
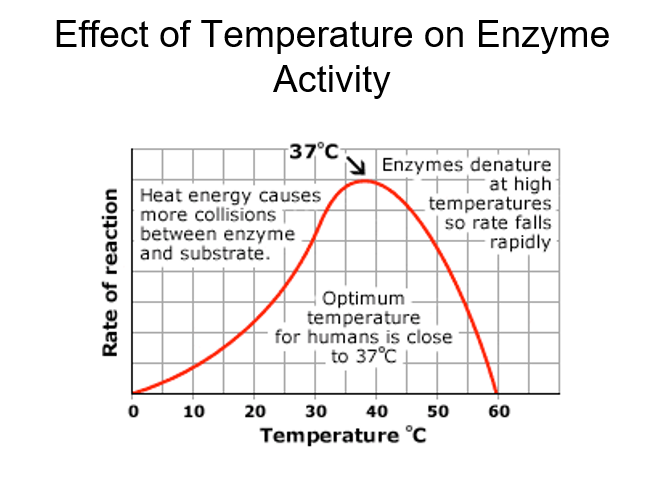
Explain effect of temperature on enzyme activity
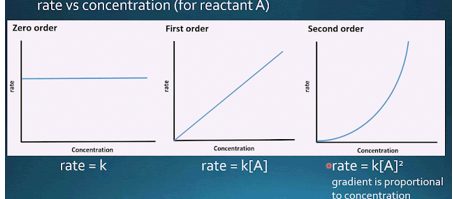
describe zero order kinetics
- rate is constant and does not depend on the amount of substance
- rate depends on presence of catalysts
- Enzyme-catalyzed reactions exhibit zero-order kinetics when the reactant concentrations are so high that the enzyme is completely saturated with reactant molecules
describe first order kinetics
- Rate is proportional with the amount of substrate present
- Description of a reaction whose rate depends on the first power of the concentration of a single reactant
Graphs for 0,1st, and 2nd order reactions
Michaelis Menton Equation explaination
- Has undergone modifications but is still the basic model for non-allosteric enzymes.
- KM - Inverse measure of the affinity of the enzyme for the substrate; Lower the KM, the higher the affinity
- Km=michaelis constant
- Enzymes can
only process so fast.
- At low substrate → rate increases almost linearly
- At high substrate → enzyme becomes saturated, rate levels off
- v: reaction rate
- [S]: substrate concentration
- Vmax: maximum rate (when enzyme is saturated)
- Km: Michaelis constant (substrate concentration at half Vmax)
what is lineweaver burk graph
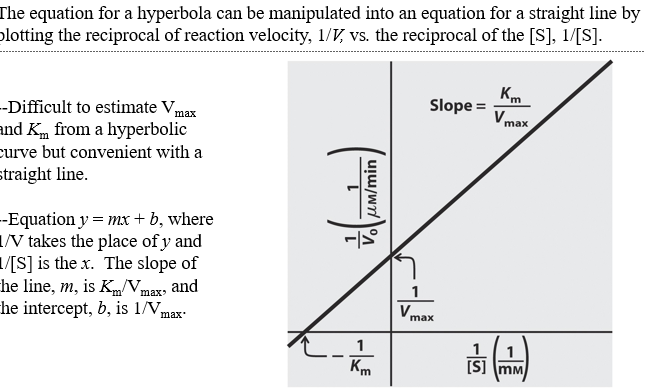
- double reciprocal plot of enzyme kinetics
What are the equations for x intercept, y intercept, and slope on lineweaver burk graph
x int= -1/Km
y int =1/Vmax
slope=Km/Vmax
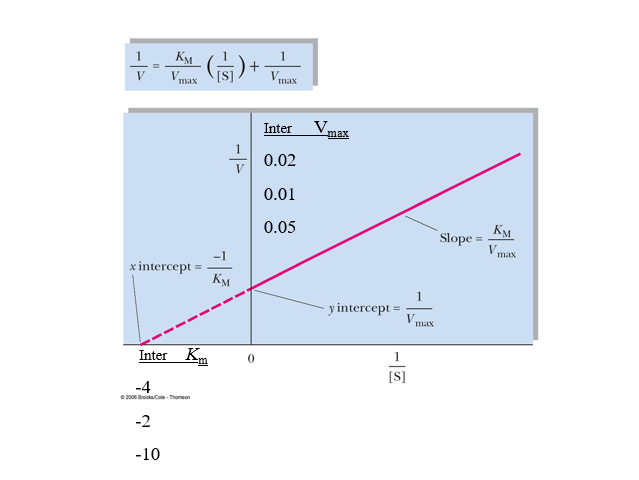
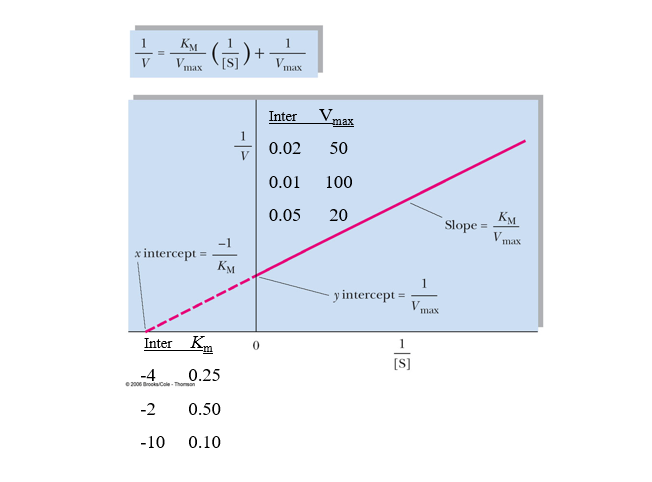
practice lineweaver burk calcs
reversible and irreversible inhibitors

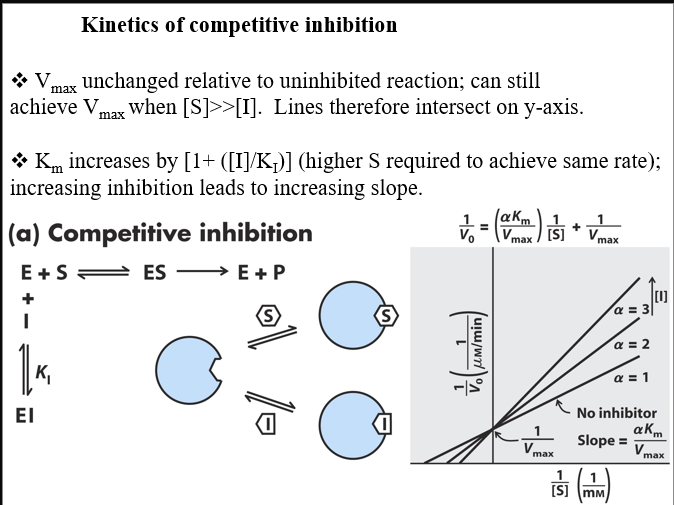
what competitive inhibitors do in terms of enzymatic output and graph information
what non-competitive inhibitors do in terms of enzymatic output and graph information
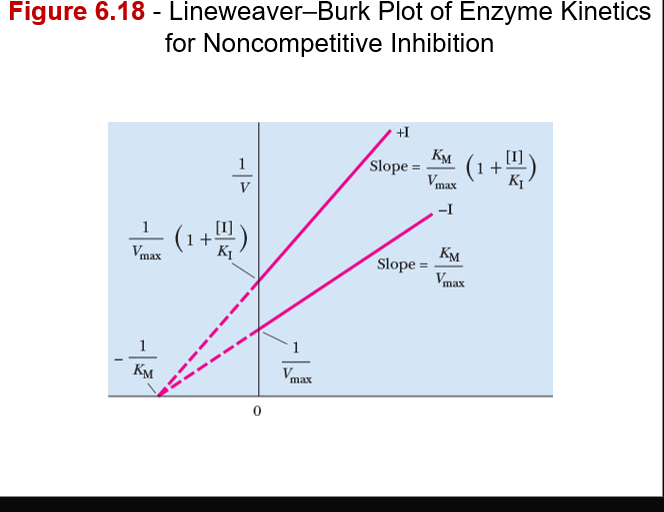
- Inhibitor does not interfere with substrate binding
–Value of Vmax decreases, and value of KM remains the same
- Increasing substrate concentration cannot overcome noncompetitive inhibition
–Inhibitor and substrate are not competing for the same site