Instructions for Side by Side Printing
- Print the notecards
- Fold each page in half along the solid vertical line
- Cut out the notecards by cutting along each horizontal dotted line
- Optional: Glue, tape or staple the ends of each notecard together
Biochem test 2
front 1 Name the enzyme classes | back 1  |
front 2  What is this reaction enzyme | back 2 oxioreductase |
front 3 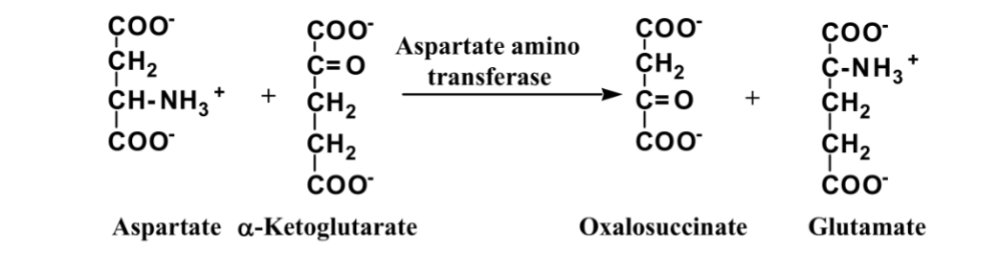 What is this reaction enzyme | back 3 transferase |
front 4  What is this reaction enzyme | back 4 hydrolase |
front 5 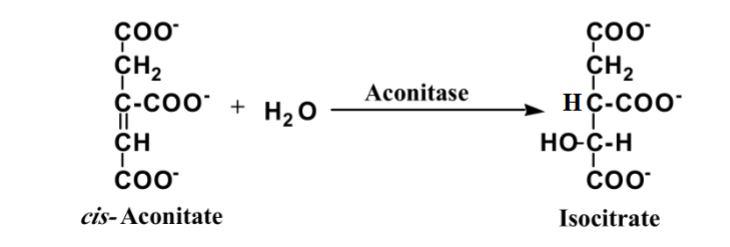 What is this reaction enzyme | back 5 Lyase |
front 6 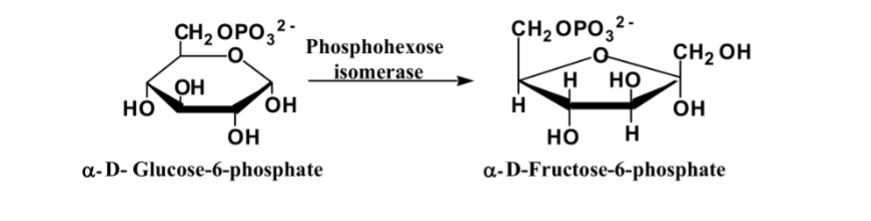 What is this reaction enzyme | back 6 Isomerase |
front 7 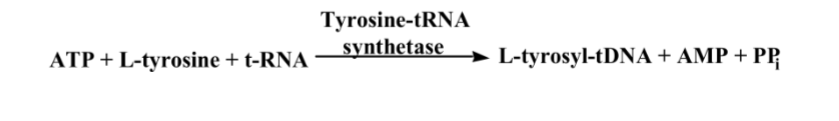 What is this reaction enzyme | back 7 Ligase |
front 8 What is a reaction intermediate | back 8 transient chemical species formed and decayed in reactions |
front 9 Define rate limiting step | back 9 step with highest activation energy in a reaction |
front 10 Explain effect of substrate concentration on enzyme activity | back 10 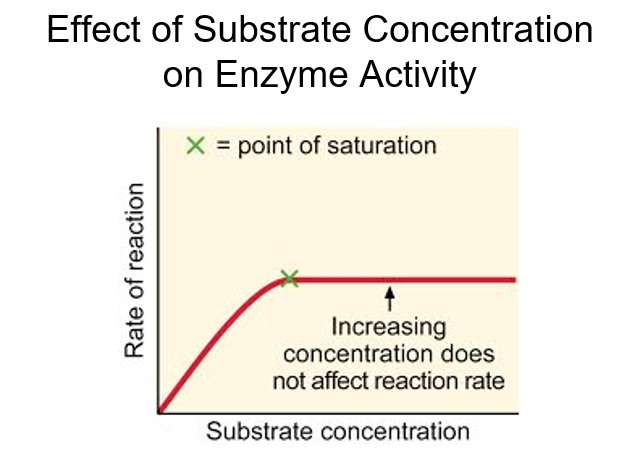 |
front 11 Explain effect of enzyme concentration on enzyme activity | back 11 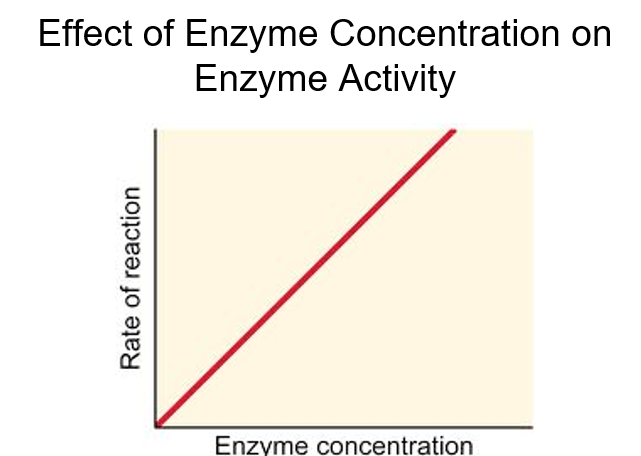 |
front 12 Explain effect of temperature on enzyme activity | back 12 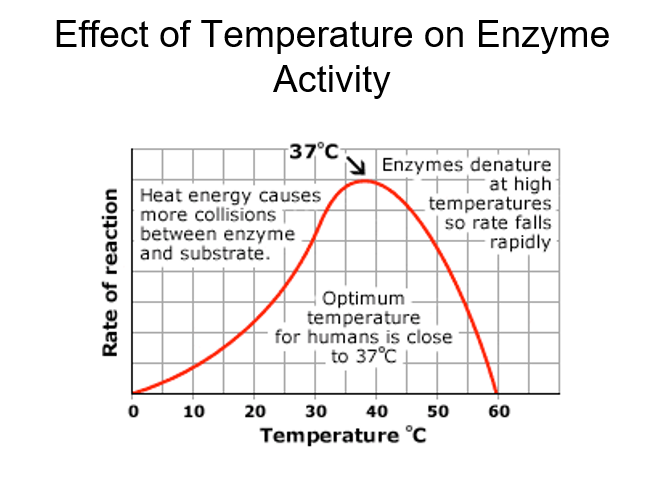 |
front 13 describe zero order kinetics | back 13
|
front 14 describe first order kinetics | back 14
|
front 15 Graphs for 0,1st, and 2nd order reactions | back 15 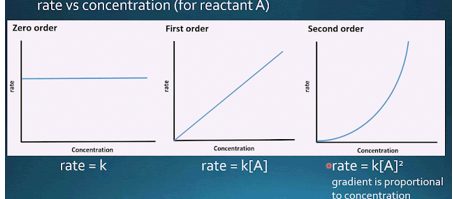 |
front 16 Michaelis Menton Equation explaination | back 16
|
front 17 what is lineweaver burk graph | back 17 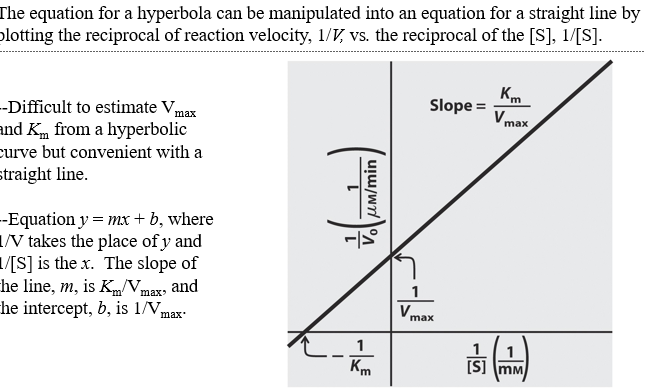
|
front 18 What are the equations for x intercept, y intercept, and slope on lineweaver burk graph | back 18 x int= -1/Km y int =1/Vmax slope=Km/Vmax |
front 19 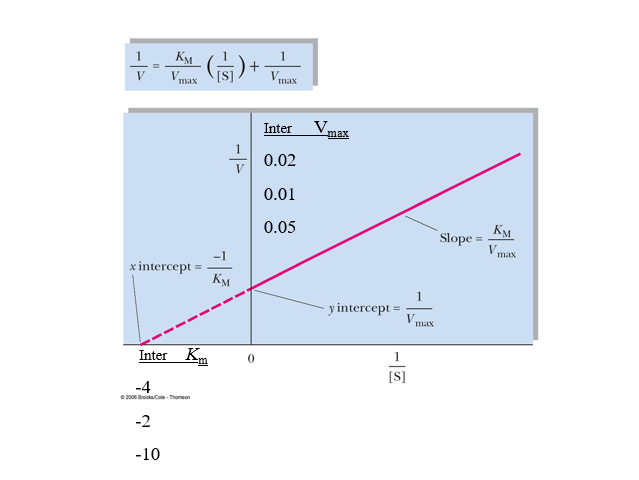 practice lineweaver burk calcs | back 19 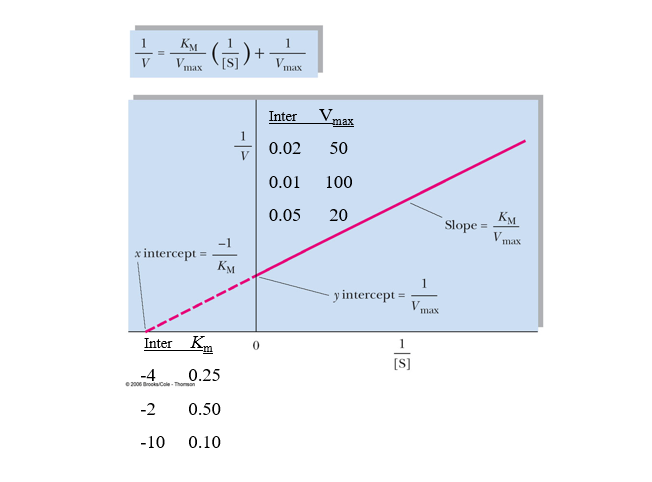 |
front 20 reversible and irreversible inhibitors | back 20  |
front 21 what competitive inhibitors do in terms of enzymatic output and graph information | back 21 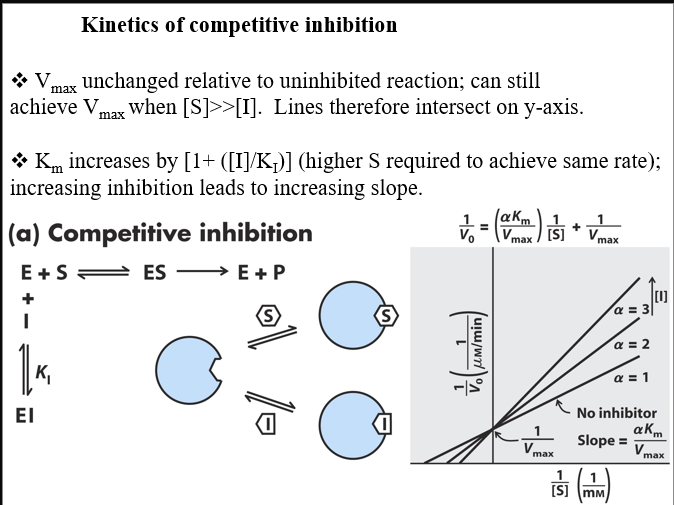 |
front 22 what non-competitive inhibitors do in terms of enzymatic output and graph information | back 22 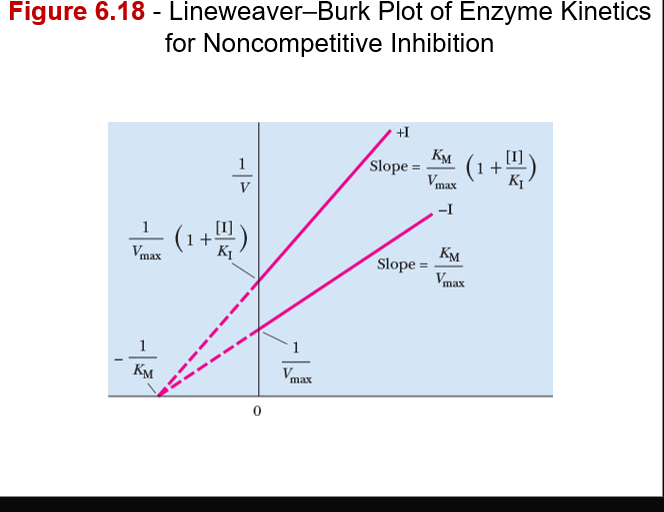
–Value of Vmax decreases, and value of KM remains the same
–Inhibitor and substrate are not competing for the same site |