The first law of thermodynamics can be given as ________.
A) ΔE
= q + w
B) ΔH°rxn = ΣnΔH°f(products) -
ΣmΔH°f(reactants)
C) for any spontaneous process, the
entropy of the universe increases
D) the entropy of a pure
crystalline substance at absolute zero is zero
E) ΔS =
qrev/T at constant temperature
A
A reaction that is spontaneous as written ________.
A) is very
rapid
B) will proceed without outside intervention
C) is
also spontaneous in the reverse direction
D) has an equilibrium
position that lies far to the left
E) is very slow
B
Of the following, only ________ is not a state
function.
A) S
B) H
C) q
D) E
E) T
C
When a system is at equilibrium, ________.
A) the reverse
process is spontaneous but the forward process is not
B) the
forward and the reverse processes are both spontaneous
C) the
forward process is spontaneous but the reverse process is not
D)
the process is not spontaneous in either direction
E) both
forward and reverse processes have stopped
D
A reversible process is one that ________.
A) can be reversed
with no net change in either system or surroundings
B) happens
spontaneously
C) is spontaneous in both directions
D) must
be carried out at low temperature
E) must be carried out at high temperature
A
Which of the following statements is true?
A) Processes that are
spontaneous in one direction are spontaneous in the opposite
direction.
B) Processes are spontaneous because they occur at an
observable rate.
C) Spontaneity can depend on the
temperature.
D) All of the statements are true.
C
The thermodynamic quantity that expresses the extent of randomness in
a system is ________.
A) enthalpy
B) internal energy
C)
bond energy
D) entropy
E) heat flow
D
For an isothermal process, ΔS = ________.
A) q
B)
qrev/T
C) qrev
D) Tqrev
E) q + w
B
Which one of the following is always positive when a spontaneous
process occurs?
A) ΔSsystem
B) ΔSsurroundings
C)
ΔSuniverse
D) ΔHuniverse
E) ΔHsurroundings
C
The entropy of the universe is ________.
A) constant
B)
continually decreasing
C) continually increasing
D)
zero
E) the same as the energy, E
C
The second law of thermodynamics states that ________.
A) ΔE = q
+ w
B) ΔH°rxn = Σ nΔH°f (products) - Σ mΔH°f (reactants)
C)
for any spontaneous process, the entropy of the universe
increases
D) the entropy of a pure crystalline substance is zero
at absolute zero
E) ΔS = qrev/T at constant temperature
C
Which of the following statements is false?
A) The change in
entropy in a system depends on the initial and final states of the
system and the path taken from one state to the other.
B) Any
irreversible process results in an overall increase in
entropy.
C) The total entropy of the universe increases in any
spontaneous process.
D) Entropy increases with the number of
microstates of the system.
A
Which one of the following processes produces a decrease of the
entropy of the system?
A) dissolving sodium chloride in
water
B) sublimation of naphthalene
C) dissolving oxygen in
water
D) boiling of alcohol
E) explosion of nitroglycerine
C
Consider a pure crystalline solid that is heated from absolute zero
to a temperature above the boiling point of the liquid. Which of the
following processes produces the greatest increase in the entropy of
the substance?
A) melting the solid
B) heating the
liquid
C) heating the gas
D) heating the solid
E)
vaporizing the liquid
E
Which one of the following correctly indicates the relationship
between the entropy of a system and the number of different
arrangements, W, in the system?
A) S = kW
B) S = k/W
C)
S = W/k
D) S = k lnW
E) S = Wk
D
The entropy change accompanying any process is given by the
equation:
A) ΔS = k lnWfinal
B) ΔS = k Wfinal - k
Winitial
C) ΔS = k ln(Wfinal / Winitial)
D) ΔS = k final - k
initial
E) ΔS = Wfinal - Winitial
C
ΔS is positive for the reaction ________.
A) 2H2 (g) + O2 (g) →
2H2O (g)
B) 2NO2 (g) → N2O4 (g)
C) CO2 (g) → CO2 (s)
D)
BaF2 (s) → Ba2+ (aq) + 2F- (aq)
E) 2Hg (l) + O2 (g) → 2HgO (s)
D
ΔS is positive for the reaction ________.
A) 2NO (g) + O2 (g) →
2NO2 (g)
B) 2N2 (g) + 3H2 (g) → 2NH3 (g)
C) C3H8 (g) + 5 O2
(g) → 3CO2 (g) + 4 H2O (g)
D) Mg (s) + Cl2 (g) → MgCl2
(s)
E) C2H4 (g) + H2 (g) → C2H6 (g)
C
ΔS is positive for the reaction ________.
A) CaO (s) + CO2 (g) →
CaCO3 (s)
B) N2 (g) + 3H2 (g) → 2NH3 (g)
C) 2SO3 (g) → 2SO2
(g) + O2 (g)
D) Ag+ (aq) + Cl- (aq) → AgCl (s)
E) H2O (l) →
H2O (s)
C
ΔS is positive for the reaction ________.
A) 2 Ca (s) + O2 (g) →
2 CaO (s)
B) 2 KClO3 (s) → 2KCl (s) + 3 O2 (g)
C) HCl (g) +
NH3 (g) → NH4Cl (s)
D) Pb2+ (aq) + 2Cl- (aq) → PbCl2 (s)
E)
CO2 (g) → CO2 (s)
B
Which reaction produces a decrease in the entropy of the
system?
A) CaCO3 (s) → CaO (s) + CO2 (g)
B) 2C (s) + O2 (g)
→ 2CO (g)
C) CO2 (s) → CO2 (g)
D) 2H2 (g) + O2 (g) → 2H2O
(l)
E) H2O (l) → H2O (g)
D
A decrease in the entropy of the system is observed for the reaction
________.
A) 4 NH3 (g) + 5 O2 (g) → 4 NO (g) + 6 H2O (g)
B)
2 HgO (s) → 2 Hg (l) + O2 (g)
C) UF6 (s) → U (s) + 3F2 (g)
D) K (s) + 1/2 I2 (g) → KI (s)
E) H2O (s) → H2O (g)
D
Which reaction produces an increase in the entropy of the
system?
A) Ag+ (aq) + Cl- (aq) → AgCl (s)
B) CO2 (s) → CO2
(g)
C) H2 (g) + Cl2 (g) → 2 HCl (g)
D) N2 (g) + 3 H2 (g) → 2
NH3 (g)
E) H2O (l) → H2O (s)
B
Which of the following reactions would have a negative ΔS?
A)
NH4Cl (s) → NH3 (g) + HCl (g)
B) PbCl2 (s) → Pb2+ (aq) + 2Cl-
(aq)
C) 2C (s) + O2 (g) → 2CO2 (g)
D) 2SO2 (g) + O2 (g) →
2SO3 (g)
E) H2O (l) → H2O (g)
D
ΔS is positive for the reaction ________.
A) Pb(NO3)2 (aq) +
2KI(aq) → PbI2 (s) + 2KNO3 (aq)
B) 2H2O (g) → 2H2 (g) + O2
(g)
C) H2O (g) → H2O (s)
D) NO (g) + O2 (g) → NO2
(g)
E) Ag+ (aq) + Cl- (aq) → AgCl (s)
B
For an isothermal process, the entropy change of the surroundings is
given by the equation:
A) ΔS = qsys T
B) ΔS = -qsys
T
C) ΔS = q lnT
D) ΔS = -q lnT
E) ΔS = -qsys / T
E
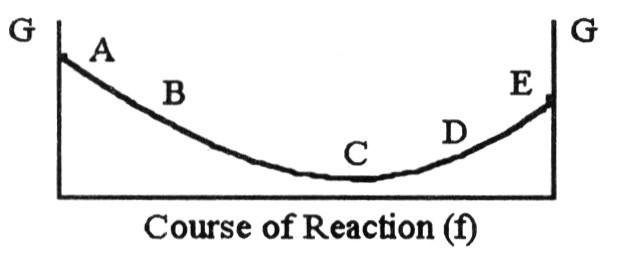
The equilibrium position corresponds to which letter on the graph of
G vs. f (course of reaction) below?
A) A
B) B
C)
C
D) D
E) E
C
For the reaction
C2H6 (g) → C2H4 (g) + H2 (g)
ΔH° is +137
kJ/mol and ΔS° is +120 J/K ∙ mol. This reaction is ________.
A)
spontaneous at all temperatures
B) spontaneous only at high
temperature
C) spontaneous only at low temperature
D)
nonspontaneous at all temperatures
B
For the reaction
2 C4H10 (g) + 13 O2 (g) → 8 CO2 (g) + 10 H2O
(g)
ΔH° is -125 kJ/mol and ΔS° is +253 J/K ∙ mol. This reaction
is ________.
A) spontaneous at all temperatures
B)
spontaneous only at high temperature
C) spontaneous only at low
temperature
D) nonspontaneous at all temperatures
E) unable
to determine without more information
A
A reaction that is not spontaneous at low temperature can become
spontaneous at high temperature if ΔH is ________ and ΔS is
________.
A) +, +
B) -, -
C) +, -
D) -, +
E)
+, 0
A
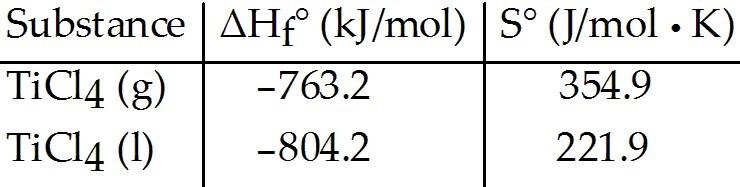
Given the following table of thermodynamic data, complete the
following sentence. The vaporization of TiCl4 is ________.
A)
spontaneous at all temperatures
B) spontaneous at low temperature
and nonspontaneous at high temperature
C) nonspontaneous at low
temperature and spontaneous at high temperature
D) nonspontaneous
at all temperatures
E) not enough information given to draw a conclusion
C

Consider the reaction:
Ag+ (aq) + Cl- (aq) → AgCl (s)
Given the following table of thermodynamic data, determine the
temperature (in °C) above which the reaction is nonspontaneous under
standard conditions.
A) 1230
B) 150
C) 432
D)
133
E) 1640
E

Consider the reaction:
NH3 (g) + HCl (g) → NH4Cl (s)
Given the following table of
thermodynamic data, determine the temperature (in °C) above which the
reaction is nonspontaneous.
A) This reaction is spontaneous at
all temperatures.
B) 618.1
C) 432.8
D) 345.0
E) 1235
D
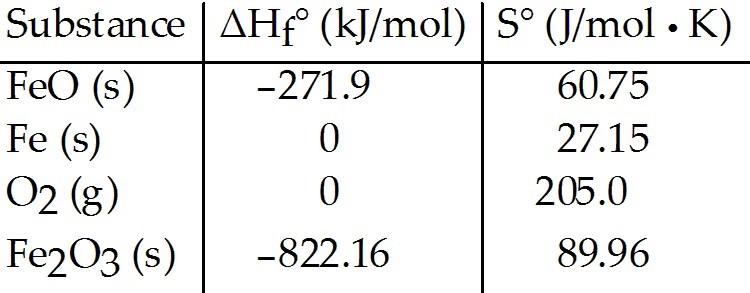
Consider the reaction:
FeO (s) + Fe (s) + O2 (g) → Fe2O3 (s)
Given the following
table of thermodynamic data, determine the temperature (in °C) at
which the reaction is nonspontaneous.
A) below 618.1
B)
above 2438
C) above 756.3
D) below 2438
E) This
reaction is spontaneous at all temperatures.
B
With thermodynamics, one cannot determine ________.
A) the speed
of a reaction
B) the direction of a spontaneous reaction
C)
the extent of a reaction
D) the value of the equilibrium
constant
E) the temperature at which a reaction will be spontaneous
A
Which one of the following statements is true about the equilibrium
constant for a reaction if ΔG° for the reaction is negative?
A) K
= 0
B) K = 1
C) K > 1
D) K < 1
C
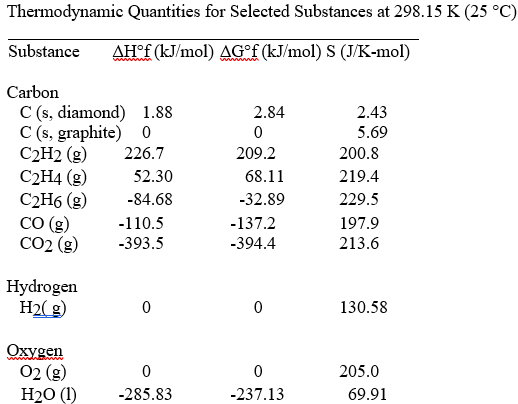
The value of ΔS° for the catalytic hydrogenation of acetylene to ethene,
C2H2 (g) + H2 (g) → C2H4 (g)
is ________ J/K∙ mol.
A) +18.6
B) +550.8
C)
+112.0
D) -112.0
E) -18.6
D

The combustion of acetylene in the presence of excess oxygen yields carbon dioxide and water:
2C2H2 (g) + 5O2 (g) → 4CO2 (g) + 2H2O (l)
The value of ΔS° for this reaction is ________ J/K ∙ mol.
A)
+689.3
B) +122.3
C) +432.4
D) -122.3
E) -432.4
E

The value of ΔS° for the reaction
2C (s, diamond) + O2 (g) → 2CO (g)
is ________ J/K ∙ mol.
A) -185.9
B) +185.9
C)
-9.5
D) +9.5
E) -195.7
B

The value of ΔS° for the catalytic hydrogenation of ethene to ethane,
C2H4 (g) + H2(g) → C2H6 (g)
is ________ J/K ∙ mol.
A) -101.9
B) -120.5
C)
-232.5
D) +112.0
E) +101.9
B

The value of ΔS° for the catalytic hydrogenation of acetylene to ethane,
C2H2 (g) + 2H2 (g) → C2H6 (g)
is ________ J/K ∙ mol.
A) -76.0
B) +440.9
C)
-232.5
D) +232.5
E) +28.7
C

The value of ΔS° for the oxidation of carbon to carbon monoxide,
2C (s, graphite) + O2 (g) → 2CO (g)
is ________ J/K ∙ mol. Carbon monoxide is produced in the
combustion of carbon with limited oxygen.
A) -12.8
B)
+408.6
C) -408.6
D) +179.4
E) +395.8
D

The value of ΔS° for the oxidation of carbon to carbon dioxide,
C (s, graphite) + O2 (g) → CO2(g)
is ________ J/K ∙ mol. The combustion of carbon, as in charcoal
briquettes, in the presence of abundant oxygen produces carbon
dioxide.
A) +424.3
B) +205.0
C) -205.0
D)
-2.9
E) +2.9
E

The combustion of ethene in the presence of excess oxygen yields carbon dioxide and water:
C2H4 (g) + 3O2 (g) → 2CO2 (g) + 2H2O (l)
The value of ΔS° for this reaction is ________ J/K ∙ mol.
A)
-267.4
B) -140.9
C) -347.6
D) +347.6
E) +140.9
A

The combustion of ethane in the presence of excess oxygen yields carbon dioxide and water:
2C2H6 (g) + 7O2 (g) → 4CO2 (g) + 6H2O (l)
The value of ΔS° for this reaction is ________ J/K ∙ mol.
A)
+718.0
B) -620.1
C) -718.0
D) -151.0
E) +151.0
B

The combustion of hydrogen in the presence of excess oxygen yields
liquid water: What is the value of ΔS° in J/K ∙ mol. for this
reaction?
A) -405.5
B) +405.5
C) -265.7
D)
-326.3
E) +265.7
D

The value of ΔS° for the oxidation of solid elemental sulfur to gaseous sulfur trioxide,
2S (s, rhombic) + 3O2(g) → 2SO3 (g)
is ________ J/K ∙ mol.
A) +19.3
B) -19.3
C)
+493.1
D) -166.4
E) -493.1
D

The value of ΔS° for the oxidation of solid elemental sulfur to gaseous sulfur dioxide,
S (s, rhombic) + O2(g) → SO2(g)
is ________ J/K ∙ mol.
A) +485.4
B) +248.5
C)
-11.6
D) -248.5
E) +11.6
E

The value of ΔS° for the decomposition of gaseous sulfur trioxide to solid elemental sulfur and gaseous oxygen,
2SO3 (g) → 2S (s, rhombic) + 3O2 (g)
is ________ J/K ∙ mol.
A) +19.3
B) -19.3
C)
+493.1
D) +166.4
E) -493.1
D

The value of ΔS° for the decomposition of gaseous sulfur dioxide to solid elemental sulfur and gaseous oxygen,
SO2 (g) → S (s, rhombic) + O2 (g)
is ________ J/K ∙ mol.
A) +485.4
B) +248.5
C)
-11.6
D) -248.5
E) +11.6
C

The value of ΔS° for the formation of POCl3 from its constituent elements,
P2 (g) + O2 (g) + 3Cl2 (g) → 2POCl3 (g)
is ________ J/K ∙ mol.
A) -442.0
B) +771.0
C)
-321.0
D) -771.0
E) +321.0
A

The value of ΔS° for the decomposition of POCl3 into its constituent elements,
2POCl3 (g) → P2 (g) + O2 (g) + 3Cl2 (g)
is ________ J/K ∙ mol.
A) +771.0
B) +442.0
C)
-321.0
D) -771.0
E) +321.0
B

The value of ΔS° for the formation of phosphorous trichloride from its constituent elements,
P2 (g) + 3Cl2 (g) → 2PCl3 (g)
is ________ J/K ∙ mol.
A) -311.7
B) +311.7
C)
-263.6
D) +129.4
E) -129.4
C

The value of ΔS° for the decomposition of phosphorous trichloride into its constituent elements,
2PCl3 (g) → P2 (g) + 3Cl2( g)
is ________ J/K ∙ mol.
A) -311.7
B) +311.7
C)
+263.6
D) +129.4
E) -129.4
C

The value of ΔS° for the formation of calcium chloride from its constituent elements,
Ca (s) + Cl2 (g) → CaCl2 (s)
is ________ J/K ∙ mol.
A) -104.6
B) +104.6
C)
+369.0
D) -159.8
E) +159.8
D

The value of ΔS° for the decomposition of calcium chloride into its constituent elements,
CaCl2 (s) → Ca (s) + Cl2 (g)
is ________ J/K ∙ mol.
A) -104.6
B) +104.6
C)
+369.0
D) -159.8
E) +159.8
E

The value of ΔH° for the oxidation of solid elemental sulfur to gaseous sulfur trioxide,
2S (s, rhombic) + 3O2( g) → 2SO3 (g)
is ________ kJ/mol.
A) +790.4
B) -790.4
C)
+395.2
D) -395.2
E) +105.1
B

The value of ΔH° for the decomposition of gaseous sulfur trioxide to its component elements,
2SO3 (g) → 2S (s, rhombic) + 3O2 (g)
is ________ kJ/mol.
A) +790.4
B) -790.4
C)
+395.2
D) -395.2
E) +105.1
A

The value of ΔH° for the oxidation of solid elemental sulfur to
gaseous sulfur dioxide,
S (s, rhombic) + O2 (g) → SO2 (g)
is ________ kJ/mol.
A) +269.9
B) -269.9
C)
+0.00
D) -11.6
E) +11.6
B

The value of ΔH° for the decomposition of gaseous sulfur dioxide to solid elemental sulfur and gaseous oxygen,
SO2 (g) → S (s,rhombic) + O2 (g)
is ________ kJ/mol.
A) 0.0
B) +135.0
C)
-135.90
D) -269.9
E) +269.9
E

The value of ΔH° for the formation of POCl3 from its constituent elements,
P2 (g) + O2 (g) + 3Cl2 (g) → 2POCl3 (g)
is ________ kJ/mol.
A) -1228.7
B) -397.7
C)
-686.5
D) +1228.7
E) +686.5
A

The value of ΔH° for the decomposition of POCl3 into its constituent elements,
2POCl3 (g) → P2 (g) + O2 (g) + 3Cl2 (g)
is ________ kJ/mol.
A) -1228.7
B) +1228.7
C)
-940.1
D) +940.1
E) 0.00
B

The value of ΔH° for the formation of phosphorous trichloride from its constituent elements,
P2 (g) + 3Cl2 (g) → 2PCl3 (g)
is ________ kJ/mol
A) -288.1
B) +432.4
C)
-720.5
D) +720.5
E) -432.4
C

The value of ΔH° for the decomposition of phosphorous trichloride into its constituent elements,
2PCl3 (g) → P2 (g) + 3Cl2 (g)
is ________ kJ/mol.
A) +576.2
B) -288.1
C)
+720.5
D) +288.1
E) -720.5
C

The value of ΔH° for the formation of calcium chloride from its constituent elements,
Ca (s) + Cl2 (g) → CaCl2 (s)
is ________ kJ/mol.
A) 0.00
B) -397.9
C)
+397.9
D) -795.8
E) +795.8
D

The value of ΔH° for the decomposition of calcium chloride into its constituent elements,
CaCl2 (s) → Ca (s) + Cl2 (g)
is ________ kJ/mol.
A) 0.00
B) -397.9
C)
+397.9
D) -795.8
E) +795.8
E

The value of ΔG° at 25 °C for the oxidation of solid elemental sulfur to gaseous sulfur trioxide,
2S (s, rhombic) + 3O2 (g) → 2SO3 (g)
is ________ kJ/mol.
A) +740.8
B) -370.4
C)
+370.4
D) -740.8
E) +185.2
D

The value of ΔG° at 25 °C for the oxidation of solid elemental sulfur to gaseous sulfur dioxide,
S (s, rhombic) + O2(g) → SO2 (g)
is ________ kJ/mol.
A) +395.2
B) +269.9
C)
-269.9
D) +300.4
E) -300.4
E

The value of ΔG° at 25 °C for the decomposition of gaseous sulfur trioxide to solid elemental sulfur and gaseous oxygen,
2SO3 (g) → 2S (s, rhombic) + 3O2 (g)
is ________ kJ/mol.
A) +740.8
B) -370.4
C)
+370.4
D) -740.8
E) +185.2
A

The value of ΔG° at 25 °C for the decomposition of gaseous sulfur
dioxide to solid elemental sulfur and gaseous oxygen,
SO2
(g) → S (s, rhombic) + O2 (g)
is ________ kJ/mol.
A) +395.2
B) +269.9
C) -269.9
D) +300.4
E) -300.4
D

The value of ΔG° at 25 °C for the formation of POCl3 from its constituent elements,
P2 (g) + O2 (g) + 3Cl2 (g) → 2POCl3 (g)
is ________ kJ/mol.
A) -1108.7
B) +1108.7
C)
-606.2
D) +606.2
E) -1,005
A

The value of ΔG° at 25 °C for the decomposition of POCl3 into its constituent elements,
2POCl3 (g) → P2 (g) + O2 (g) + 3Cl2 (g)
is ________ kJ/mol.
A) -1108.7
B) +1108.7
C)
-606.2
D) +606.2
E) -1,005
B

The value of ΔG° at 25 °C for the formation of phosphorous trichloride from its constituent elements,
P2 (g) + 3Cl2 (g) → 2PCl3 (g)
is ________ kJ/mol.
A) -539.2
B) +539.2
C)
-642.9
D) +642.9
E) -373.3
C

The value of ΔG° at 25 °C for the decomposition of phosphorous trichloride into its constituent elements,
2PCl3 (g) → P2 (g) + 3Cl2 (g)
is ________ kJ/mol.
A) -539.2
B) +539.2
C)
-642.9
D) +642.9
E) -373.3
D

The value of ΔG° at 25 °C for the formation of calcium chloride from its constituent elements,
Ca (s) + Cl2 (g) → CaCl2 (s)
is ________ kJ/mol.
A) -795.8
B) +795.8
C)
+763.7
D) +748.1
E) -748.1
E

The value of ΔG° at 25 °C for the decomposition of calcium chloride into its constituent elements,
CaCl2 (s) → Ca (s) + Cl2 (g)
is ________ kJ/mol.
A) -795.8
B) +795.8
C)
+763.7
D) +748.1
E) -748.1
D

The value of ΔG° at 373 K for the oxidation of solid elemental sulfur
to gaseous sulfur dioxide is ________ kJ/mol. At 298 K, ΔH° for this
reaction is -269.9 kJ/mol, and ΔS° is +11.6 J/K.
A)
-300.4
B) -274.2
C) -4,597
D) +300.4
E) +4,597
B

The value of ΔG° at 25 °C for the following reaction:
C2H4 (g) + H2 (g) → C2H6 (g)
is ________ kJ/mol. At 298 K, ΔH° for this reaction is -137.5
kJ/mol, and ΔS° is +120.5 J/K.
A) -35800
B) -173.4
C)
35800
D) -101.7
E) -274.2
B

Given the thermodynamic data in the table below, calculate the
equilibrium constant (at 298 K) for the reaction:
2 SO2 (g) + O2
(g) ⇌ 2 SO3 (g)
A) 2.40 × 1024
B) 1.06
C) 1.95
D) 3.82
× 1023
E) More data are needed.
A
The value of ΔG° for a reaction conducted at 25 °C is 3.05 kJ/mol.
The equilibrium constant for a reaction is ________ at this
temperature.
A) 0.292
B) -4.20
C) 0.320
D)
-1.13
E) More information is needed.
A
What is the equilibrium constant for a reaction at 25 °C? ΔG° for the
reaction is 2.8 kJ/mol.
A) 1.13
B) 0.32
C) -4.2
D)
-1.13
E) More information is needed.
B
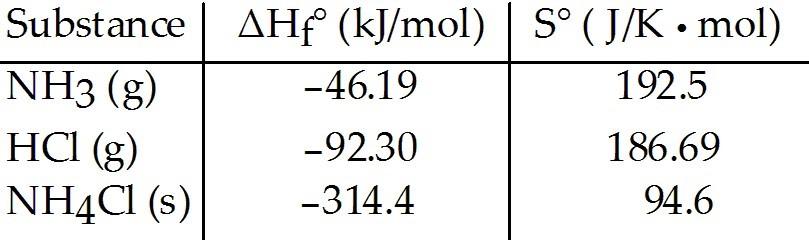
Consider the reaction between ammonia and hydrochloric acid to
produce ammonium chloride.
Given the following table of
thermodynamic data at 298 K:
The value of K for the reaction at 25 °C is ________.
A) 8.4 ×
104
B) 150
C) 1.1 × 10-16
D)
9.3 × 1015
E) 1.4 × 108
D
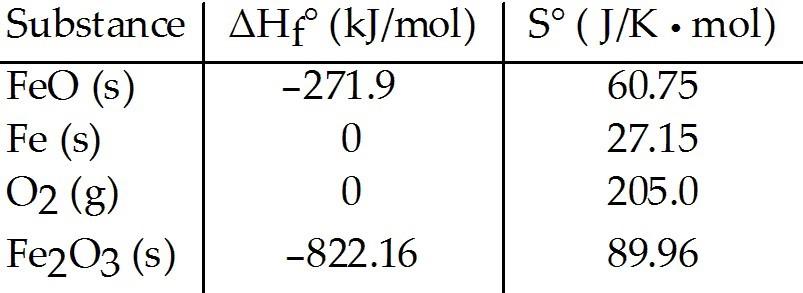
Consider the reaction:
FeO (s) + Fe (s) + O2(g) → Fe2O3 (s)
Given the following table
of thermodynamic data at 298 K:
The value K for the reaction at 25 °C is ________.
A)
370
B) 5.9 × 104
C) 3.8 × 10-14
D) 7.1 × 1085
E) 8.1 × 1019
D
Consider the formation of solid silver chloride from aqueous silver and chloride ions.
Given the following table of thermodynamic data at 298 K:
The value of K for the reaction at 25 °C is ________.
A)
1.9 × 10-10
B) 810
C) 1.8 × 104
D) 3.7 × 1010
E) 5.3 × 109
E
The normal boiling point of methanol is 64.7°C and the molar enthalpy
of vaporization if 71.8 kJ/mol. The value of ΔS when 1.75 mol of
CH3OH(I) vaporizes at 64.7 °C is ________ J/K
A) 0.372
B)
372
C) 1.94 × 103
D) 4.24 × 107
E) 1.94
B
The normal boiling point of water is 100.0 °C and its molar enthalpy
of vaporization is 40.67 kJ/mol. What is the change in entropy in the
system in J/K when 24.7 grams of steam at 1 atm condenses to a liquid
at the normal boiling point?
A) 88.8
B) -88.8
C)
-150
D) 373
E) -40.7
C
The normal boiling point of C2Cl3F3 is 47.6 °C and its molar enthalpy
of vaporization is 27.49 kJ/mol. What is the change in entropy in the
system in J/K when 28.6 grams of C2Cl3F3 vaporizes to a gas at the
normal boiling point?
A) -13.1
B) -4.19
C) 4.19
D)
13.1
E) 27.5
D
What is the change in entropy in the system in J/K when 112.2 grams
of ethanol at 1 atm condenses to a liquid at the normal boiling point?
The normal boiling point of ethanol (C2H5OH) is 78.3 °C and its molar
enthalpy of vaporization is 38.56 kJ/mol.
A) 382
B)
1199
C) -267
D) -1199
E) -382
C
Which one of the following processes produces a decrease in the
entropy of the system?
A) freezing of Fe(l) into Fe(s)
B)
evaporation of liquid ethanol into gaseous ethanol
C) dissolution
of LiOH(s) in water
D) melting ice to form water
E) mixing
of two gases into one container
A
ΔS is negative for the reaction ________.
A) Sr(NO3)2 (aq) +
2LiOH (aq) → Sr(OH)2 (s) + 2LiNO3 (aq)
B) 2H2O (g) → 2H2 (g) + O2
(g)
C) CH3OH (l) → CH3OH (g)
D) LiOH (aq) → Li+ (aq) + OH–
(aq)
E) C6H12O6 (s) → 6C (s) + 6H2 (g) + 3O2 (g)
A
Which of the following has the largest entropy?
A) HI
(g)
B) HBr (g)
C) HCl (g)
D) HCl (s)
E) HCl (l)
A
Which of the following has the largest entropy?
A) B2F4
(g)
B) BF3 (g)
C) B2H6 (g)
D) BCl3 (l)
E) B2O3 (s)
A
Which of the following has the largest entropy at 25 °C and
atm?
A) C3H4
B) C3H6
C) C3H8
D) H2
E) C2H6
C
Which of the following has the largest entropy at 25 °C and
atm?
A) Cl2
B) F2
C) I2
D) Br2
E) O3
C
The standard Gibbs free energy of formation of ________ is zero.
(a) H2O (l)
(b) O (g)
(c) CL2 (g)
A) (a) only
B) (b) only
C) (c) only
D) (b) and
(c)
E) (a), (b), and (c)
C
The standard Gibbs free energy of formation of ________ is zero.
(a)H2 O (l)
(b) Fe (s)
(c) I2 (s)
A) (a) only
B) (b) only
C) (c) only
D) (b) and
(c)
E) (a), (b), and (c)
D
The standard Gibbs free energy of formation of ________ is zero.
(a) Mn (s)
(b) I2 (s)
(c) Cu (s)
A) (a) only
B) (b) only
C) (c) only
D) (b) and
(c)
E) (a), (b), and (c)
E
The value of ΔG° at 261.0 °C for the formation of phosphorous trichloride from its constituent elements,
P2(g) +3CL2 (g) → 2PCL3(g)
is ________ kJ/mol. At 25.0 °C for this reaction, ΔH° is -720.5
kJ/mol, ΔG° is -643.9 kJ/mol, and ΔS° is -263.7 J/K
A)
-579.6
B) 6.81 × 104
C) 1.40 × 105
D) -651.7
E) -861.4
A
The value of ΔG° at 181.0 °C for the formation of calcium chloride
from calcium metal and chlorine gas is ________ kJ/mol. At 25.0 °C for
this reaction, ΔH° is -795.8 kJ/mol, ΔG° is -748.1 kJ/mol, and ΔS° is
-159.8 J/K
A) 7.18 × 104
B) -868.4
C)
-766.9
D) -723.2
E) 2.81 × 104
D
The signs of ΔH° and ΔS° must be ________ and ________, respectively,
for a reaction to be spontaneous at high temperatures but
nonspontaneous at low temperatures.
A) +, –
B) –, +
C)
–, –
D) +, +
E) +, 0
D
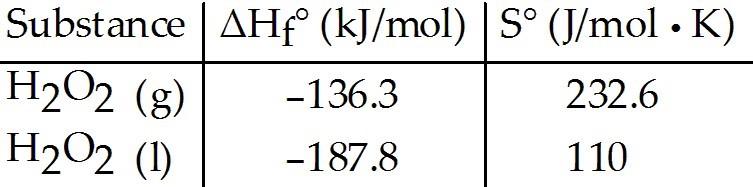
Given the following table of thermodynamic data, complete the
following sentence. The vaporization of H2O2 (l) is ________.
A)
nonspontaneous at low temperature and spontaneous at high
temperature
B) spontaneous at low temperature and nonspontaneous
at high temperature
C) spontaneous at all temperatures
D)
nonspontaneous at all temperatures
E) not enough information
given to draw a conclusion
A
For the reaction
C(s) + H2O(g) → CO(g) + H2(g)
ΔH° = 133.3 kJ/mol and ΔS° = 121.6 J/K ∙ mol at 298 K. At
temperatures greater than ________ °C this reaction is spontaneous
under standard conditions.
A) 273
B) 325
C) 552
D)
823
E) 1096
D
At what temperature in Kelvin will a reaction have ΔG = 0? ΔH = -24.2
kJ/mol and ΔS = -55.5 J/K-mol and assume both do not vary with
temperature.
A) 2.29
B) 2293
C) 298
D) 436
E) 0.436
D
At what temperature will a reaction be spontaneous? ΔH = +22.2 kJ/mol
and ΔS = +81.1 J/K-mol and assume both do not vary with
temperature.
A) at T > 298 K
B) at T < 274 K
C)
at T < 298 K
D) at T > 274 K
E) at all temperatures
D
For a given reaction, ΔH = +74.6 kJ/mol, and the reaction is
spontaneous at temperatures above the crossover temperature, 449 K.
The value of ΔS = __________ J/mol ∙ K, assuming that ΔH and ΔS do not
vary with temperature.
A) 166
B) 6020
C) -166
D)
-6020
E) 3.35 × 104
A
For a given reaction, ΔS = +69.0 J/mol∙K, and the reaction is
spontaneous at temperatures above the crossover temperature, 439 K.
The value of ΔH = __________ kJ/mol, assuming that ΔH and ΔS do not
vary with temperature.
A) 30.3
B) -30.3
C) 1.57 ×
10-4
D) -1.57 × 10-4
E) 6.36 × 10-3
A
For a given reaction with ΔH = -28.1 kJ/mol, the ΔG = 0 at 372 K. The
value of ΔS must be __________ J/K-mol, assuming that ΔH and ΔS do not
vary with temperature.
A) -75.5
B) 75.5
C) -7.55 ×
10-5
D) 7.55 × 10-5
E) -1.32 × 10-2
A
For a given reaction with ΔS = -50.8 J/K-mol, the ΔG = 0 at 395 K.
The value of ΔH must be __________ kJ/mol, assuming that ΔH and ΔS do
not vary with temperature.
A) -20.1
B) 20.1
C) -1.29 ×
10-4
D) 1.29 × 10-4
E) -7.78 × 10-3
A
What is the equilibrium constant for a reaction at 25 °C. The value
of ΔG° is -57.5 kJ/mol.
A) 8.4 × 10101
B) 10
C) 1.0
D) 1.2 × 1010
E) more information
is needed
D
If ΔG° for a reaction is less than zero, then ________.
A) K
> 1
B) K = 1
C) K < 1
D) K = 0
E) more
information is needed.
A
In the Haber process, ammonia gas is synthesized from nitrogen gas
and hydrogen gas. ΔG° at 298 K for this reaction is -33.3 kJ/mol. The
value of ΔG at 298 K for a reaction mixture that consists of 1.9 atm
nitrogen gas, 2.3 atm hydrogen gas, and 0.85 atm ammonia gas is
________.
A) -1.0
B) -4.09 × 103
C) -8.62
× 103
D) -118.0
E) -41.9
E
Phosphorous and chlorine gases combine to produce phosphorous
trichloride. ΔG° at 298 K for this reaction is -642.9 kJ/mol. The
value of ΔG at 298 K for a reaction mixture that consists of 1.9 atm
P2, 1.6 atm CL2, and PCL3 is ________.
A) -650.1
B)
-34.9
C) -714.1
D) -4.46 × 103
E) -7.86 × 103
A
The equilibrium constant for a reaction is 0.38 at 25 °C. What is the
value of ΔG° (kJ/mol) at this temperature?
A) 2.4
B)
-4.2
C) 200
D) 4.2
E) More information is needed.
A
The equilibrium constant for the following reaction is 3.0 ×
108 at 25 °C.
N2 (g) + 3H2 (g) ⇌ 2NH3 (g)
The value of ΔG° for this reaction is ________ kJ/mol.
A)
22
B) -4.1
C) 4.1
D) -48
E) -22
D
A reversible change produces the maximum amount of ________ that can be achieved by the system on the surroundings.
work
Calculate ΔG° (in kJ/mol) for the following reaction at 1 atm and 25 °C:
C2H6 (g) + O2 (g) → CO2 (g) + H2O (l) (unbalanced)
ΔGf° C2H6 (g) = -32.89 kJ/mol; ΔGf° CO2 (g) = -394.4 kJ/mol; ΔGf° H2O (l) = -237.13 kJ/mol
-2935.0
Calculate ΔG∘ (in kJ/mol) for the following reaction at 1 atm and 25 °C:
C2H6 (g) + O2 (g) → CO2 (g) + H2O (l) (unbalanced)
ΔHf∘ C2H6 (g) = -84.7 kJ/mol; S∘ C2H6 (g) = 229.5 J/K ∙ mol;
ΔHf∘ CO2 (g) = -393.5 kJ/mol; S∘ CO2 (g) = 213.6 J/K ∙ mol;
ΔHf∘ H2O (l) = -285.8 kJ/mol; S∘H2O (l) = 69.9 J/K ∙ mol;
S∘O2 (g) = 205.0 J/K ∙ mol
-2934.0
At what temperature (in K) will a reaction become spontaneous? ΔH is 115.0 kJ/mol and ΔS is 75.00 J/K ∙ mol.
1533
At what temperature (in K) will a reaction become spontaneous? ΔH is 65.0 kJ/mol and ΔS is 149.00 J/K ∙ mol.
436
What is the ΔG° (kJ/mol) for the formation of silver chloride at 25 °C? Ksp = 1.8 × 10-10
56
The melting of a substance at its melting point is an isothermal process.
true
The vaporization of a substance at its boiling point is an isothermal process.
true
The quantity of energy gained by a system equals the quantity of energy gained by its surroundings.
false
The entropy of a pure crystalline substance at 0 K is zero.
true
The more negative ΔG° is for a given reaction, the larger the value of the corresponding equilibrium constant, K.
true