1) For a protein to be an integral membrane protein, it would have to be _____.
A) hydrophilic
B) hydrophobic
C) amphipathic, with at
least one hydrophobic region
D) exposed on only one surface of the membrane
Answer: C
2) You have a planar bilayer with equal amounts of saturated and unsaturated phospholipids. After testing the permeability of this membrane to glucose, you increase the proportion of unsaturated phospholipids in the bilayer. What will happen to the membrane's permeability to glucose?
A) Permeability to glucose will increase.
B) Permeability to
glucose will decrease.
C) Permeability to glucose will stay the
same.
D) You cannot predict the outcome. You simply have to make
the measurement.
Answer: A
3) According to the fluid mosaic model of cell membranes,
phospholipids _____.
A) can move laterally along the plane of
the membrane
B) frequently flip-flop from one side of the
membrane to the other
C) occur in an uninterrupted bilayer, with
membrane proteins restricted to the surface of the membrane
D) have hydrophilic tails in the interior of the membrane
Answer: A
4) The membranes of winter wheat are able to remain fluid when it is
extremely cold by _____. A) increasing the percentage of unsaturated
phospholipids in the membrane
B) increasing the percentage of
cholesterol molecules in the membrane
C) decreasing the number
of hydrophobic proteins in the membrane
D) cotransport of glucose and hydrogen
Answer: A
5) Some regions of the plasma membrane, called lipid rafts, have a
higher concentration of cholesterol molecules. At higher temperatures,
these regions _____.
A) are more fluid than the surrounding
membrane
B) are less fluid than the surrounding membrane
C) detach from the plasma membrane and clog arteries
D) have
higher rates of lateral diffusion of lipids and proteins into and out
of these regions
Answer: B
6) Singer and Nicolson's fluid mosaic model of the membrane proposed
that membranes_____. A) are a phospholipid bilayer between two layers
of hydrophilic proteins
B) are a single layer of phospholipids
and proteins
C) consist of protein molecules embedded in a fluid
bilayer of phospholipids
D) consist of a mosaic of polysaccharides and proteins
Answer: C
7) An animal cell lacking oligosaccharides on the external surface of its plasma membrane would likely be impaired in which function?
A) transporting ions against an electrochemical gradient
B)
cell-cell recognition
C) attaching the plasma membrane to the cytoskeleton
D) establishing a diffusion barrier to charged molecules
Answer: B
8) Which of these are NOT embedded in the hydrophobic portion of the
lipid bilayer at all? A) transmembrane proteins
B) integral
proteins
C) peripheral proteins
D) All of these are embedded in the hydrophobic portion of the lipid bilayer.
Answer: C
9) Why are lipids and proteins free to move laterally in
membranes?
A) The interior of the membrane is filled with liquid
water.
B) Lipids and proteins repulse each other in the
membrane.
C) Hydrophilic portions of the lipids are in the
interior of the membrane.
D) There are only weak hydrophobic
interactions in the interior of the membrane.
Answer: D
15) Cell membranes are asymmetrical. Which of the following
statements is the most likely explanation for the membrane's
asymmetrical nature?
A) Since the cell membrane forms a border
between one cell and another in tightly packed tissues such as
epithelium, the membrane must be asymmetrical
B) Since cell membranes communicate signals from one organism to
another, the cell membranes must be asymmetrical.
C) The two
sides of a cell membrane face different environments and carry out
different functions.
D) Proteins only function on the cytoplasmic side of the cell membrane, which results in the membrane's asymmetrical nature.
Answer: C
16) In what way do the membranes of a eukaryotic cell vary?
A)
Phospholipids are found only in certain membranes.
B) Certain
proteins are unique to each membrane.
C) Only certain membranes
of the cell are selectively permeable.
D) Some membranes have
hydrophobic surfaces exposed to the cytoplasm, while others have
hydrophilic surfaces facing the cytoplasm.
Answer: B
17) Which of the following is a reasonable explanation for why
unsaturated fatty acids help keep a membrane more fluid at lower
temperatures?
A) The double bonds form kinks in the fatty acid
tails, preventing adjacent lipids from packing tightly.
B) Unsaturated fatty acids have a higher cholesterol content and,
therefore, more cholesterol in membranes.
C) Unsaturated fatty
acids are more polar than saturated fatty acids.
D) The double
bonds block interaction among the hydrophilic head groups of the lipids
Answer: A
18) What kinds of molecules pass through a cell membrane most easily?
A) large and hydrophobic
B) small and hydrophobic
C)
large polar
D) ionic
Answer: B
19) Which of the following most accurately describes selective
permeability?
A) An input of energy is required for
transport.
B) Lipid-soluble molecules pass through a
membrane.
C) There must be a concentration gradient for
molecules to pass through a membrane.
D) Only certain molecules can cross a cell membrane.
Answer: D
20) Which of the following is a characteristic feature of a carrier protein in a plasma membrane?
A) It exhibits a specificity for a particular type of
molecule.
B) It requires the expenditure of cellular energy to
function.
C) It works against diffusion.
D) It has no hydrophobic regions.
Answer: A
21) Which of the following would likely move through the lipid
bilayer of a plasma membrane most rapidly?
A) CO2
B) an amino acid
C) glucose
D) K+
Answer: A
22) Which of the following allows water to move much faster across cell membranes?
A) the sodium-potassium pump
B) ATP
C) peripheral proteins
D) aquaporins
Answer: D
23) You are working on a team that is designing a new drug. For this
drug to work, it must enter the cytoplasm of specific target cells.
Which of the following would be a factor that determines whether the
molecule selectively enters the target cells?
A) hydrophobicity
of the drug molecule
B) lack of charge on the drug molecule
C) similarity of the
drug molecule to other molecules transported by the target cells
D) lipid composition of the target cells' plasma membrane
Answer: C
24) Diffusion _____.
A) is very rapid over long distances
B) requires an expenditure of energy by the cell
C) is a passive
process in which molecules move from a region of higher concentration
to a region of lower concentration
D) requires integral proteins
in the cell membrane
Answer: C
25) Which of the following processes includes all others?
A) osmosis
B) facilitated diffusion
C) passive transport
D) transport of an ion down its electrochemical gradient
Answer: C
26) When a cell is in equilibrium with its environment, which of the
following occurs for substances that can diffuse through the
cell?
A) There is random movement of substances into and out of
the cell.
B) There is directed movement of substances into and
out of the cell.
C) There is no movement of substances into and out of the cell.
D) All movement of molecules is directed by active transport.
Answer: A
27) Which of the following is true of osmosis?
A) Osmosis only
takes place in red blood cells.
B) Osmosis is an
energy-demanding or "active" process.
C) In osmosis,
water moves across a membrane from areas of lower solute concentration
to areas of higher solute concentration.
D) In osmosis, solutes
move across a membrane from areas of lower water concentration to
areas of higher water concentration.
Answer: C
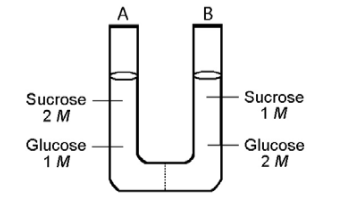
The solutions in the two arms of this U-tube are separated by a membrane that is permeable to water and glucose but not to sucrose. Side A is half-filled with a solution of 2 M sucrose and 1 M glucose. Side B is half-filled with 1 M sucrose and 2 M glucose. Initially, the liquid levels on both sides are equal.
28) Refer t o the figure. Initially, in terms of tonicity, the solution in side A with respect to the solution in side B is _____.
A) hypotonic
B) isotonic
C) saturated
D) hypertonic
Answer: B
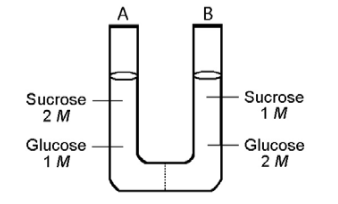
The solutions in the two arms of this U-tube are separated by a membrane that is permeable to water and glucose but not to sucrose. Side A is half-filled with a solution of 2 M sucrose and 1 M glucose. Side B is half-filled with 1 M sucrose and 2 M glucose. Initially, the liquid levels on both sides are equal.
29) Refer to the figure. After the system reaches equilibrium, what changes are observed?
A) The molarity of sucrose is higher than that of glucose on side
A.
B) The water level is higher in side A than in side B.
C) The water level is unchanged.
D) The water level is higher in side B than in side A.
Answer: B
30) A patient was involved a serious accident and lost a large
quantity of blood. In an attempt to replenish body fluids, distilled
water—equal to the volume of blood lost—is added to the blood directly
via one of his veins. What will be the most probable result of this
transfusion?
A) The patient's red blood cells will shrivel up
because the blood has become hypotonic compared to the cells.
B) The patient's red blood cells will swell and possibly burst
because the blood has become hypotonic compared to the cells.
C)
The patient's red blood cells will shrivel up because the blood has
become hypertonic compared to the cells.
D) The patient's red blood cells will burst because the blood has become hypertonic compared to the cells.
Answer: B
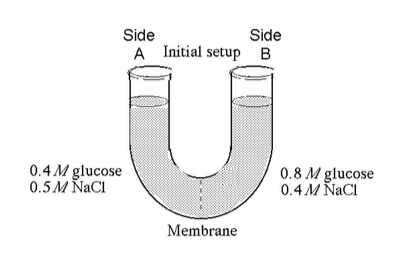
The solutions in the arms of a U-tube are separated at the bottom of the tube by a selectively permeable membrane. The membrane is permeable to sodium chloride but not to glucose. Side A is filled with a solution of 0.4 M glucose and 0.5 M sodium chloride (NaCl), and side B is filled with a solution containing 0.8 M glucose and 0.4 M sodium chloride. Initially, the volume in both arms is the same..
31) Refer to the figure. At the beginning of the experiment,
A) side A is hypertonic to side B.
B) side A is hypotonic to
side B.
C) side A is hypertonic to side B with respect to glucose.
D) side A is hypotonic to side B with respect to NaCl.
Answer: B
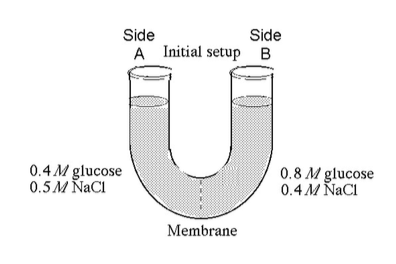
32) Refer to the figure. If you examine side A after three days, you
should find _____.
A) a decrease in the concentration of NaCl
and glucose and an increase in the water level
B) a decrease in the concentration of NaCl, an increase in water
level, and no change in the concentration of glucose
C) a
decrease in the concentration of NaCl and a decrease in the water
level
D) no change in the concentration of NaCl and glucose and
an increase in the water level
Answer: C
36) Celery stalks that are immersed in fresh water for several hours become stiff. Similar stalks left in a 0.15 M salt solution become limp. From this we can deduce that the fresh water_____.
A) and the salt solution are both hypertonic to the cells of the
celery stalks
B) is hypotonic and the salt solution is
hypertonic to the cells of the celery stalks
C) is hypertonic and the salt solution is hypotonic to the cells of the celery stalks
D) is isotonic and the salt solution is hypertonic to the cells of the celery stalks
Answer: B
37) What will happen to a red blood cell (RBC), which has an internal
ion concentration of about 0.9 percent, if it is placed into a beaker
of pure water?
A) The cell would shrink because the water in the
beaker is hypotonic relative to the cytoplasm of the RBC.
B) The cell would shrink because the water in the beaker is
hypertonic relative to the cytoplasm of the RBC.
C) The cell
would swell because the water in the beaker is hypotonic relative to
the cytoplasm of the RBC.
D) The cell will remain the same size because the solution outside the cell is isotonic.
Answer: C
38) Which of the following statements correctly describes the normal
tonicity conditions for typical plant and animal cells? The animal
cell is in _____.
A) a hypotonic solution, and the plant cell is
in an isotonic solution
B) an isotonic solution, and the plant
cell is in a hypertonic solution
C) a hypertonic solution, and the plant cell is in an isotonic solution
D) an isotonic solution, and the plant cell is in a hypotonic solution
Answer: D
39) In which of the following would there be the greatest need for osmoregulation?
A) an animal connective tissue cell bathed in isotonic body
fluid
B) a salmon moving from a river into an ocean
C) a
red blood cell surrounded by plasma
D) a plant being grown hydroponically in a watery mixture of designated nutrients
Answer: B
40) When a plant cell, such as one from a rose stem, is submerged in
a very hypotonic solution, what is likely to occur?
A) The cell
will burst.
B) Plasmolysis will shrink the interior.
C) The cell will become flaccid.
D) The cell will become turgid.
Answer: D
41) A sodium-potassium pump _____.
A) moves three potassium
ions out of a cell and two sodium ions into a cell while producing an
ATP for each cycle
B) move three sodium ions out of a cell and
two potassium ions into a cell while consuming an ATP for each
cycle
C) moves three potassium ions out of a cell and two sodium
ions into a cell while consuming 2 ATP in each cycle
D) move
three sodium ions out of a cell and two potassium ions into a cell and
generates an ATP in each cycle
Answer: B
42) The sodium-potassium pump is called an electrogenic pump because it _____.
A) pumps equal quantities of Na+ and K+ across the membrane
B)
contributes to the membrane potential
C) ionizes sodium and
potassium atoms
D) is used to drive the transport of other
molecules against a concentration gradient
Answer: B
43) Which of the following membrane activities requires energy from
ATP?
A) facilitated diffusion of chloride ions across the
membrane through a chloride channel
B) movement of Na+ ions from a lower concentration in a mammalian
cell to a higher concentration in the extracellular fluid
C)
movement of glucose molecules into a bacterial cell from a medium
containing a higher concentration of glucose than inside the
cell
D) movement of carbon dioxide out of a paramecium
Answer: B
44) The voltage across a membrane is called the _____.
A) chemical gradient
B) membrane potential
C) osmotic potential
D) electrochemical gradient
Answer: B
45) Ions diffuse across membranes through specific ion channels down _____.
A) their chemical gradients
B) their concentration
gradients
C) the electrical gradients
D) their electrochemical gradients
Answer: D
46) Which of the following would increase the electrochemical
gradient across a membrane? A) a sucrose-proton cotransporter
B)
a proton pump
C) a potassium channel
D) both a proton pump and a potassium channel
Answer: B
47) The phosphate transport system in bacteria imports phosphate into the cell even when the concentration of phosphate outside the cell is much lower than the cytoplasmic phosphate concentration. Phosphate import depends on a pH gradient across the membrane—more acidic outside the cell than inside the cell. Phosphate transport is an example of _____.
A) passive diffusion
B) facilitated diffusion
C) active
transport
D) cotransport
Answer: D
48) In some cells, there are many ion electrochemical gradients
across the plasma membrane even though there are usually only one or
two proton pumps present in the membrane. The gradients of the other
ions are most likely accounted for by _____.
A) cotransport proteins
B) ion channels
C) pores in the plasma membrane
D)
passive diffusion across the plasma membrane
Answer: A
49) Which of the following is most likely true of a protein that
cotransports glucose and sodium ions into the intestinal cells of an
animal?
A) Sodium and glucose compete for the same binding site
in the cotransporter.
B) Glucose entering the cell down its
concentration gradient provides energy for uptake of sodium ions
against the electrochemical gradient.
C) Sodium ions can move down their electrochemical gradient through
the cotransporter whether or not glucose is present outside the
cell.
D) A substance that blocks sodium ions from binding to the
cotransport protein will also block the transport of glucose.
Answer: D
50) Proton pumps are used in various ways by members of every domain
of organisms: Bacteria, Archaea, and Eukarya. What does this most
probably mean?
A) Proton gradients across a membrane were used
by cells that were the common ancestor of all three domains of life.
B) The high concentration of protons in the ancient atmosphere must
have necessitated a pump mechanism.
C) Cells of each domain
evolved proton pumps independently when oceans became more acidic.
D) Proton pumps are necessary to all cell membranes.
Answer: A
51) Several epidemic microbial diseases of earlier centuries incurred
high death rates because they resulted in severe dehydration due to
vomiting and diarrhea. Today they are usually not fatal because we
have developed which of the following?
A) antiviral medications
that are efficient and work well with most viruses
B)
intravenous feeding techniques
C) medications to slow blood
loss
D) hydrating drinks with high concentrations of salts and glucose
Answer: D
52) The force driving simple diffusion is _____, while the energy
source for active transport is _____.
A) the concentration
gradient; ADP
B) the concentration gradient; ATP
C) transmembrane pumps; electron transport
D) phosphorylated
protein carriers; ATP
Answer: B
53) An organism with a cell wall would most likely be unable to take in materials through _____.
A) osmosis
B) active transport
C) phagocytosis
D) facilitated diffusion
Answer: C
54) White blood cells engulf bacteria using _____.
A) phagocytosis
B) pinocytosis
C) osmosis
D) receptor-mediated exocytosis
Answer: A
55) Familial hypercholesterolemia is characterized by _____.
A)
defective LDL receptors on the cell membranes
B) poor attachment
of the cholesterol to the extracellular matrix of cells
C) a
poorly formed lipid bilayer that cannot incorporate cholesterol into
cell membranes D) inhibition of the cholesterol active transport
system in red blood cells
Answer: A
56) The difference between pinocytosis and receptor-mediated
endocytosis is that _____.
A) pinocytosis brings only water
molecules into the cell, but receptor-mediated endocytosis brings in
other molecules as well.
B) pinocytosis increases the surface
area of the plasma membrane, whereas receptor-mediated endocytosis
decreases the plasma membrane surface area.
C) pinocytosis is
nonselective in the molecules it brings into the cell, whereas
receptor-mediated endocytosis offers more selectivity.
D)
pinocytosis can concentrate substances from the extracellular fluid,
but receptor-mediated endocytosis cannot.
Answer: C
57) In receptor-mediated endocytosis, receptor molecules initially
project to the outside of the cell. Where do they end up after
endocytosis?
A) on the outside of vesicles
B) on the
inside surface of the cell membrane
C) on the inside surface of the vesicle
D) on the outer
surface of the nucleus
Answer: C
58) A bacterium engulfed by a white blood cell through phagocytosis
will be digested by enzymes contained in _____.
A)
lysosomes
B) Golgi vesicles
C) vacuoles
D) secretory vesicles
Answer: A
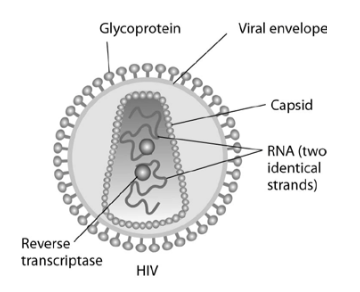
Human immunodeficiency virus (HIV) infects cells that have both CD4 and CCR5 cell surface molecules. The viral nucleic acid molecules are enclosed in a protein capsid, and the protein capsid is itself contained inside an envelope consisting of a lipid bilayer membrane and viral glycoproteins. One hypothesis for viral entry into cells is that binding of HIV membrane glycoproteins to CD4 and CCR5 initiates fusion of the HIV membrane with the plasma membrane, releasing the viral capsid into the cytoplasm. An alternative hypothesis is that HIV gains entry into the cell via receptor-mediated endocytosis, and membrane fusion occurs in the endocytotic vesicle. To test these alternative hypotheses for HIV entry, researchers labeled the lipids on the HIV membrane with a red fluorescent dye.
59) In an HIV-infected cell producing HIV virus particles, the viral
glycoprotein is expressed on the plasma membrane. How do the viral
glycoproteins get to the plasma membrane? They are synthesized
_____.
A) on ribosomes on the plasma membrane
B) by ribosomes in the rough ER and arrive at the plasma membrane in
the membrane of secretory vesicles
C) on free cytoplasmic
ribosomes and then inserted into the plasma membrane
D) by
ribosomes in the rough ER, secreted from the cell, and inserted into
the plasma membrane from the outside
Answer: B
Human immunodeficiency virus (HIV) infects cells that have both CD4 and CCR5 cell surface molecules. The viral nucleic acid molecules are enclosed in a protein capsid, and the protein capsid is itself contained inside an envelope consisting of a lipid bilayer membrane and viral glycoproteins. One hypothesis for viral entry into cells is that binding of HIV membrane glycoproteins to CD4 and CCR5 initiates fusion of the HIV membrane with the plasma membrane, releasing the viral capsid into the cytoplasm. An alternative hypothesis is that HIV gains entry into the cell via receptor-mediated endocytosis, and membrane fusion occurs in the endocytotic vesicle. To test these alternative hypotheses for HIV entry, researchers labeled the lipids on the HIV membrane with a red fluorescent dye.
60) What would be observed by live-cell fluorescence microscopy
immediately after HIV entry if HIV is endocytosed first, and then
later fuses with the endocytotic vesicle membrane?
A) A spot of
red fluorescence will be visible on the infected cell's plasma
membrane, marking the site of membrane fusion and HIV entry.
B) The red fluorescent dye-labeled lipids will appear in the
infected cell's interior.
C) A spot of red fluorescence will
diffuse in the infected cell's cytoplasm.
D) A spot of red
fluorescence will remain outside the cell after delivering the viral capsid.
Answer: B