1) Substrate-level phosphorylation occurs _____.1) _______
A)
during oxidative phosphorylation B) in glycolysis
C) in the
citric acid cycle D) in both glycolysis and the citric acid cycle
D
2) The molecule that functions as the reducing agent (electron donor) in a redox or oxidation-reduction reaction _____.
A) gains electrons and gains potential energy B) gains electrons and loses potential energy C) loses electrons and loses potential energy D) loses electrons and gains potential energy
C
3) Which of the listed statements describes the results of the following reaction? C6H12O6 + 6 O2 → 6 CO2 + 6 H2O + Energy
3) _______
A) CO2 is reduced and O2 is oxidized. B) O2 is reduced and CO2 is
oxidized.
C) C6H12O6 is oxidized and O2 is reduced. D) O2 is
oxidized and H2O is reduced.
C
4) High levels of citric acid inhibit the enzyme phosphofructokinase, a key enzyme in glycolysis. Citric acid binds to the enzyme at a different location from the active site. This is an example of _____. 4) _______
A) allosteric regulation
B) competitive inhibition
C)
the specificity of enzymes for their substrates
D) positive feedback regulation
A
5) Why is glycolysis considered to be one of the first metabolic
pathways to have evolved?
A) It is found in prokaryotic cells
but not in eukaryotic cells.
B) It does not involve organelles
or specialized structures, does not require oxygen, and is present in
most organisms. C) It produces much less ATP than does oxidative
phosphorylation.
D) It requires the presence of
membrane-enclosed cell organelles found only in eukaryotic cells.
B
6) Which of the following occurs in the cytosol of a eukaryotic cell? 6) _______
A) oxidation of pyruvate to acetyl CoA B) fermentation and
chemiosmosis
C) glycolysis and fermentation D) citric acid cycle
C
7) Which of the following normally occurs regardless of whether or not oxygen (O2) is present?
A) oxidative phosphorylation (chemiosmosis)
B) citric acid cycle
C) fermentation
D) glycolysis
D
8) In liver cells, the inner mitochondrial membranes are about five times the area of the outer mitochondrial membranes. What purpose must this serve? 8) _______
A) It increases the surface for oxidative phosphorylation.
B) It allows for an increased rate of glycolysis.
5) _______
C) It allows for an increased rate of the citric acid cycle.
D) It increases the surface for substrate-level phosphorylation.
A
9) The synthesis of ATP by oxidative phosphorylation, using the energy released by movement of protons across the membrane down their electrochemical gradient, is an example of _____. 9) _______
A) a reaction with a positive ΔG
B) allosteric
regulation
C) active transport
D) an endergonic reaction
coupled to an exergonic reaction
D
10) When hydrogen ions are pumped from the mitochondrial matrix across the inner membrane and into the intermembrane space, the result is the _____. 10) ______
A) lowering of pH in the mitochondrial matrix B) reduction of NAD+
C) creation of a proton-motive force D) formation of ATP
C
11) Energy released by the electron transport chain is used to pump H+ into which location in eukaryotic cells? 11) ______
A) mitochondrial inner membrane B) mitochondrial intermembrane space C) mitochondrial outer membrane D) mitochondrial matrix
B
12) The primary role of oxygen in cellular respiration is to _____. 12) ______
A) combine with carbon, forming CO2
B) yield energy in the form of ATP as it is passed down the respiratory chain
C) combine with lactate, forming pyruvate
D) act as an
acceptor for electrons and hydrogen, forming water
D
13) During aerobic respiration, which of the following directly donates electrons to the electron transport chain at the lowest energy level? 13) ______
A) ATP B) FADH2 C) ADP +P i D) NADH
B
14) During aerobic respiration, electrons travel downhill in which sequence?
A) glucose → pyruvate → ATP → oxygen
B) glucose → ATP →
electron transport chain → NADH
C) glucose → NADH → electron
transport chain → oxygen
D) food → glycolysis → citric acid cycle → NADH → ATP
C
15) The electron transport chain _____. 15) ______
A) takes place in the cytoplasm of prokaryotic cells
B) is a series of redox reactions
C) is a series of
substitution reactions
D) is driven by ATP consumption
B
16) Which one of the following is formed by the removal of a carbon (as CO2) from a molecule of pyruvate? 16) ______
A) acetyl CoA B) citrate
C) oxaloacetate D) glyceraldehyde 3-phosphate
A
In the presence of oxygen, the three-carbon compound pyruvate can be catabolized in the citric acid cycle. First, however, the pyruvate (1) loses a carbon, which is given off as a molecule of CO2, (2) is oxidized to form a two-carbon compound called acetate, and (3) is bonded to coenzyme A.
17) The three listed steps result in the formation of _____. 17) ______
A) acetyl CoA, NAD+, ATP, and CO2
B) acetyl CoA, FADH2, and CO2
C) acetyl CoA, NADH, and CO2
D) acetyl CoA, O2, and ATP
C
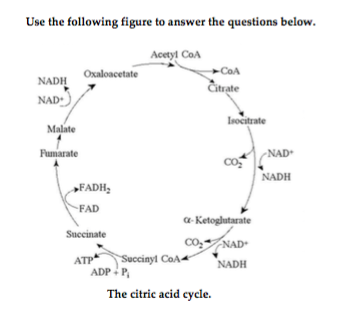
18) For each mole of glucose (C6H12O6) oxidized by cellular respiration, how many moles of CO2 are released in the citric acid cycle (see the accompanying figure)? 18) ______
A) 2 B) 4 C) 32 D) 6
B
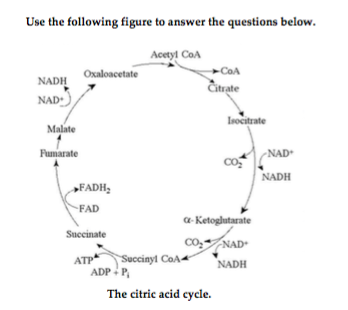
19) Which electron carrier(s) function in the citric acid cycle? 19) ______
A) ADP and ATP
B) NADH and FADH2
C) NAD+ only
D) the electron transport chain
B
20) Which kind of metabolic poison would most directly interfere with glycolysis?
A) an agent that reacts with NADH and oxidizes it to NAD+
B) an agent that reacts with oxygen and depletes its concentration in the cell
C) an agent that closely mimics the structure of glucose but is not metabolized
D) an agent that binds to pyruvate and inactivates it
C
21) Starting with one molecule of glucose, the energy-containing products of glycolysis are _____.21) ______
A) 2 NADH, 2 pyruvate, and 2 ATP
B) 2 NAD+, 2 pyruvate, and 2 ATP
C) 2 FADH2, 2 pyruvate, and 4 ATP
D) 6 CO2, 2 pyruvate, and 2 ATP
A
22) The free energy for the oxidation of glucose to CO2 and water is -686 kcal/mol and the free energy for the reduction of NAD+ to NADH is +53 kcal/mol. Why are only two molecules of NADH formed during glycolysis when it appears that as many as a dozen could be formed? 22) ______
A) Most of the free energy available from the oxidation of glucose
is used in the production of ATP in glycolysis.
B) Most of the
free energy available from the oxidation of glucose remains in
pyruvate, one of the products of glycolysis
. C) There is no CO2 or water produced as products of
glycolysis.
D) Glycolysis is a very inefficient reaction, with
much of the energy of glucose released as heat.
B
23) The oxygen consumed during cellular respiration is involved directly in which process or event?
A) accepting electrons at the end of the electron transport
chain
B) the citric acid cycle
C) the oxidation of
pyruvate to acetyl CoA
D) glycolysis
A
24) Plants photosynthesize _____.
A) only in the light but respire in light and dark
B) and
respire only in the light
C) only in the dark but respire only
in the light
D) only in the light but respire only in the dark
A
25) Early investigators thought the oxygen produced by photosynthetic plants came from carbon dioxide. In fact, it comes from _____.
A) water
C) glucose
B) electrons from NADPH
D) air
A
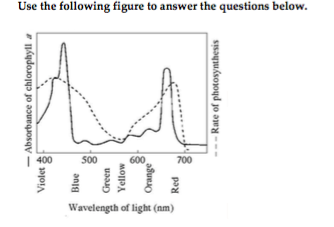
26) The figure shows the absorption spectrum for chlorophyll a and the action spectrum for photosynthesis. Why are they different? 26) ______
A) Oxygen given off during photosynthesis interferes with the
absorption of light.
B) Green and yellow wavelengths inhibit the
absorption of red and blue wavelengths.
C) Aerobic bacteria take
up oxygen, which changes the measurement of the rate of photosynthesis.
D) Other pigments absorb light in addition to chlorophyll a.
D
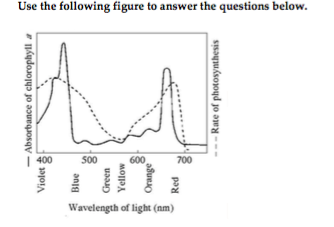
27) What wavelength of light in the figure is most effective in driving photosynthesis? 27) ______
A) 730 mm B) 575 mm C) 420 mm D) 625 mm
C
Theodor W. Engelmann illuminated a filament of algae with light that passed through a prism, thus exposing different segments of algae to different wavelengths of light. He added aerobic bacteria and then noted in which areas the bacteria congregated. He noted that the largest groups were found in the areas illuminated by the red and blue light.
28) What did Engelmann conclude about the congregation of bacteria in the red and blue areas? 28) ______
A) Bacteria are attracted to red and blue light and thus these
wavelengths are more reactive than other wavelengths. B) Bacteria
congregated in these areas because these areas had the most oxygen
being released.
C) Bacteria congregated in these areas due to an
increase in the temperature of the red and blue light.
D)
Bacteria congregated in these areas due to an increase in the
temperature caused by an increase in photosynthesis.
B
29) An outcome of Engelmann's experiment was to help determine the relationship between _____.
A) wavelengths of light and the amount of heat released
B) the
concentration of carbon dioxide and the rate of photosynthesis
C) wavelengths of light and the rate of photosynthesis
D) wavelengths of light and the rate of aerobic respiration
C
A spaceship is designed to support animal life for a multiyear voyage to the outer planets of the solar system. Plants will be grown to provide oxygen and to recycle carbon dioxide. Since the spaceship will be too far from the sun for photosynthesis, an artificial light source will be needed.
30) What wavelengths of light should be used to maximize plant growth with a minimum of energy expenditure? 30) ______
A) full-spectrum white light B) green light
C) UV light D) a
mixture of blue and red light
D
31) Why are there several structurally different pigments in the reaction centers of photosystems? 31) ______
A) Excited electrons must pass through several pigments before they
can be transferred to electron acceptors of the electron transport
chain.
B) They enable the reaction center to excite electrons to
a higher energy level.
C) This arrangement enables the plant to
absorb light energy of a variety of wavelengths.
D) They enable
the plant to absorb more photons from light energy, all of which are
at the same wavelength.
C
32) In autumn, the leaves of deciduous trees change colors. This is because chlorophyll is degraded and _____. 32) ______
A) carotenoids and other pigments are still present in the leaves
B) sugars are sent to most of the cells of the leaves
C) the
degraded chlorophyll changes into many other colors
D) water
supply to the leaves has been reduced
A
33) As electrons are passed through the system of electron carriers associated with photosystem II, they lose energy. What happens to this energy? 33) ______
A) It excites electrons of the reaction center of photosystem
I.
B) It is used to phosphorylate NAD+ to NADPH, the molecule
that accepts electrons from photosystem I.
C) It is used to establish and maintain a proton gradient.
D) It is lost as heat.
C
34) The final electron acceptor associated with photosystem I is _____.
A) NADP B) oxygen C) water D) NADPH
A
35) The electrons of photosystem II are excited and transferred to electron carriers. From which molecule or structure do the photosystem II replacement electrons come? 35) ______
A) water B) oxygen
C) the electron carrier, plastocyanin D)
photosystem I
A
36) Some photosynthetic organisms contain chloroplasts that lack photosystem II, yet are able to survive. The best way to detect the lack of photosystem II in these organisms would be to _____.
36) ______
A) test for liberation of O2 in the light B) test for CO2 fixation in the dark
C) determine if they have thylakoids in the chloroplasts D) do experiments to generate an action spectrum
A
37) What are the products of linear electron flow?
A) ADP and NADP+ B) ATP and NADPH
C) heat and fluorescence D)
ATP and P700
B
38) Assume a thylakoid is somehow punctured so that the interior of the thylakoid is no longer separated from the stroma. This damage will most directly affect the _____.
A) reduction of NADP+
B) synthesis of ATP
C) splitting
of water
D) flow of electrons from photosystem II to photosystem I
B
39) In a plant cell, where are the ATP synthase complexes located?
A) inner mitochondrial membrane only
B) thylakoid membrane and
inner mitochondrial membrane
C) thylakoid membrane only
D) thylakoid membrane and plasma membrane
B
40) In mitochondria, chemiosmosis moves protons from the matrix into the intermembrane space, whereas in chloroplasts, chemiosmosis moves protons from the _____.
A) intermembrane space to the matrix B) matrix to the stroma C) thylakoid space to the stromaD) stroma to the thylakoid space
D
41) Which of the following statements best describes the relationship between photosynthesis and respiration? 41) ______
A) Photosynthesis stores energy in complex organic molecules;
respiration releases energy from complex organic molecules
B)
Photosynthesis occurs only in plants; respiration occurs only in
animals.
C) Respiration runs the biochemical pathways of
photosynthesis in reverse.
D) Photosynthesis is catabolic; respiration is anabolic.
A
42) What is the relationship between the wavelength of light and the quantity of energy per photon?
A) They are logarithmically related. B) They are separate
phenomena.
C) They are inversely related. D) They have a direct,
linear relationship.
C
43) In a plant, the reactions that produce molecular oxygen (O2) take place in _____.
A) the light reactions and the Calvin cycle
B) neither the
light reactions nor the Calvin cycle
C) the light reactions alone
D) the Calvin cycle alone
C
44) A flask containing photosynthetic green algae and a control flask containing water with no algae are both placed under a bank of lights, which are set to cycle between 12 hours of light and 12 hours of dark. The dissolved oxygen concentrations in both flasks are monitored. Predict what the relative dissolved oxygen concentrations will be in the flask with algae compared to the control flask. The dissolved oxygen in the flask with algae will _____. 44) ______
A) always be lower
B) be higher in the light, but the same in
the dark
C) be higher in the light, but lower in the dark
D) always be higher
C
45) Where does the Calvin cycle take place? 45) ______
A) stroma of the chloroplast B) outer membrane of the chloroplast C) interior of the thylakoid (thylakoid space) D) thylakoid membrane
A
46) What is the primary function of the Calvin cycle? 46) ______
A) split water and release oxygen
B) transport RuBP out of the
chloroplast
C) use NADPH to release carbon dioxide
D) synthesize simple sugars from carbon dioxide
D
47) Which of the following sequences correctly represents the flow of electrons during photosynthesis?
A) NADPH → chlorophyll → Calvin cycle
B) NADPH → electron transport chain → O2
C) H2O → NADPH →
Calvin cycle
D) NADPH → O2 → C O2
C
48) Which of the following does NOT occur during the Calvin cycle? 48) ______
A) release of oxygen
B) regeneration of the CO2 acceptor
C) consumption of ATP
D) oxidation of NADPH
A
49) When electrons flow along the electron transport chains of mitochondria, which of the following changes occurs? 49) ______
A) The pH of the matrix increases.
B) ATP synthase pumps
protons by active transport.
C) NAD+ is oxidized.
D) The electrons gain free energy.
E) The cytochromes phosphorylate ADP to form ATP.
A
Theodor W. Engelmann illuminated a filament of algae with light that passed through a prism, thus exposing different segments of algae to different wavelengths of light. He added aerobic bacteria and then noted in which areas the bacteria congregated. He noted that the largest groups were found in the areas illuminated by the red and blue light.
50) In Engelmann experiment, if you ran the same experiment without passing light through a prism, what would you predict?50) ______
A) The bacteria would be relatively evenly distributed along the
algal filaments.
B) The number of bacteria present would
increase due to an increase in the carbon dioxide concentration. C)
There would be no difference in results.
D) The number of
bacteria would decrease due to a decrease in the temperature of the
water.
E) The number of bacteria present would decrease due to
an increase in the carbon dioxide concentration.
A