1. Which of the following is not a theme that unifies
biology?
A) interaction with the environment
B) emergent
properties
C) evolution
D) reductionism
E) structure
and function
D
2. Which of these is a correct representation of the hierarchy of
biological organization from least to most complex?
A) organelle
of a stomach cell, digestive system, large intestine, small intestine,
intestinal tissue, organism
B) organelle of an intestinal cell,
digestive system, small intestine, large intestine, intestinal tissue,
organism
C) molecule, intestinal cell organelle, intestinal cell,
intestinal tissue, digestive system, organism
D) molecule, small
intestine, large intestine, intestinal tissue, digestive system,
organism
E) molecule, digestive system, digestive cell organelle,
small intestine, large intestine, intestinal cell, organism
C
3. The dynamics of any ecosystem include the following major
processes:
A) the flow of energy from sunlight to
producers
B) the flow of energy from sunlight to producers and
then to consumers
C) the recycling of chemical nutrients
D)
the flow of energy to producers and the recycling of nutrients
E)
the flow of energy from sunlight to producers and then to consumers,
and the recycling of chemical nutrients.
E
4. Once labor begins in childbirth, contractions increase in
intensity and frequency until delivery. The increasing labor
contractions of childbirth are an example of
A) a bioinformatic
system.
B) positive feedback.
C) negative feedback.
D)
feedback inhibition.
E) enzymatic catalysis.
B
5. Two species that belong to the same genus must also belong to the
same
A) kingdom.
B) phylum.
C) class.
D)
order.
E) all of the above
E
6. A controlled experiment is one in which
A) the experiment is
repeated many times to ensure that the results are accurate.
B)
the experiment proceeds at a slow pace to guarantee that the scientist
can carefully observe all reactions and process all experimental
data.
C) there are at least two groups, one of which does not
receive the experimental treatment.
D) there are at least two
groups, one differing from the other by two or more variables.
E)
there is one group for which the scientist controls all variables.
C
7. Which of the following statements is false?
A) Atoms of the
various elements differ in their number of subatomic
particles.
B) All atoms of a particular element have the same
number of protons in their nuclei.
C) The neutrons and protons
present in the nucleus of an atom are almost identical in mass; each
has a mass of about 1 dalton.
D) An atom is the smallest unit of
an element that still retains the properties of the element.
E)
Protons and electrons are electrically charged particles. Protons have
one unit of negative charge, and electrons have one unit of positive charge.
E
8. Calcium has an atomic number of 20 and an atomic mass of 40.
Therefore, a calcium atom must have
A) 20 protons.
B) 40
electrons.
C) 40 neutrons.
D) A and B only
E) A, B, and C
A
9. Which of the following best describes the relationship between the atoms described below?
Atom 1 Atom 2
31/15 P 32/15 P
A) They contain 31 and 32
electrons, respectively.
B) They are both phosphorus
cations.
C) They are both phosphorus anions.
D) They are
both isotopes of phosphorus.
E) They contain 31 and 32 protons, respectively.
D
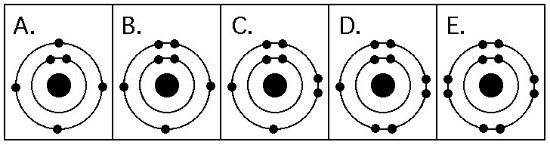
10. Which drawing depicts the electron configuration of oxygen (16/8 O )?
A) 1
B) 2
C) 3
D) 4
E) 5
C
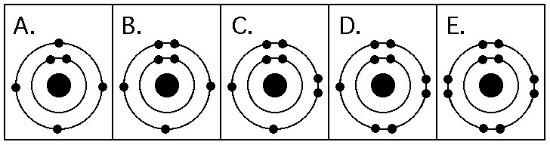
11. Which drawing depicts an atom that is inert or chemically unreactive?
A) 1
B) 2
C) 3
D) 4
E) 5
E
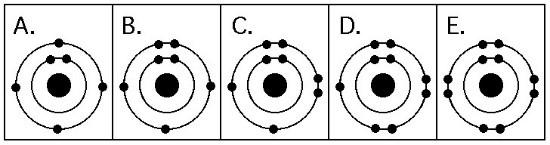
12. A molecule of carbon dioxide (CO2) is formed when one atom of
carbon (atomic number 6) is covalently bonded with two atoms of oxygen
(atomic number 8). What is the total number of electrons that must be
shared between the carbon atom and the oxygen atoms in order to
complete the outer electron shell of all three atoms?
A)
1
B) 2
C) 3
D) 4
E) 5
D
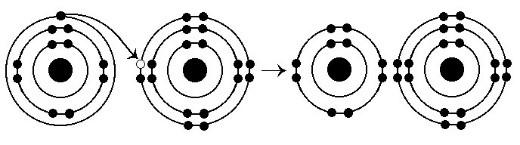
13. What results from the chemical reaction illustrated in Figure
2.3?
A) a cation with a net charge of +1
B) a cation with a
net charge of -1
C) an anion with a net charge of +1
D) an
anion with a net charge of -1
E) A and D
E
14. A group of molecular biologists is trying to synthesize a new
artificial compound to mimic the effects of a known hormone that
influences sexual behavior. They have turned to you for advice. Which
of the following compounds is most likely to mimic the effects of the
hormone?
A) a compound with the same number of carbon atoms as
the hormone
B) a compound with the same molecular mass (measured
in daltons) as the hormone
C) a compound with the same
three-dimensional shape as part of the hormone
D) a compound with
the same number of orbital electrons as the hormone
E) a compound
with the same number of hydrogen and nitrogen atoms as the hormone
C
15. The slight negative charge at one end of one water molecule is
attracted to the slight positive charge of another water molecule.
What is this attraction called?
A) a covalent bond
B) a
hydrogen bond
C) an ionic bond
D) a hydrophilic bond
E)
a hydrophobic bond
B
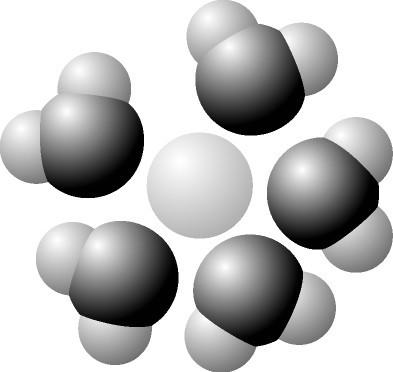
16. Based on your knowledge of the polarity of water molecules, the
solute molecule is most likely
A) positively charged.
B)
negatively charged.
C) without charge.
D)
hydrophobic.
E) nonpolar.
A
17. How many molecules of glycerol (C3H8O3) would be present in 1 L
of a 1 M glycerol solution?
A) 1
B) 14
C) 92
D) 1
× 10^7
E) 6.02 × 10^23
E
18. A given solution contains 0.0001(10^-4) moles of hydrogen ions
[H+] per liter. Which of the following best describes this
solution?
A) acidic: H+ acceptor
B) basic: H+
acceptor
C) acidic: H+ donor
D) basic: H+ donor
E) neutral
C
19. If the pH of a solution is increased from pH 5 to pH 7, it means
that the
A) concentration of H+ is twice (2X) what it was at pH
5.
B) concentration of H+ is half (1/2) what it was at pH
5.
C) concentration of OH- is 100 times greater than what it was
at pH 5.
D) concentration of OH- is one-hundredth (0.01X) what it
was at pH 5.
E) concentration of H+ is 100 times greater and the
concentration of OH- is one-hundredth what they were at pH 5.
C
20. One of the buffers that contribute to pH stability in human blood
is carbonic acid (H2CO3). Carbonic acid is a weak acid that
dissociates into a bicarbonate ion (HCO3-) and a hydrogen ion (H+).
Thus,
H2CO3 ↔ HCO3- + H+
If the pH of the blood drops, one
would expect:
A) a decrease in the concentration of H2CO3 and an
increase in the concentration of HCO3-.
B) the concentration of
hydroxide ion (OH-) to increase.
C) the concentration of
bicarbonate ion (HCO3-) to increase.
D) the HCO3- to act as a
base and remove excess H+ with the formation of H2CO3.
E) the
HCO3- to act as an acid and remove excess H+ with the formation of H2CO3.
D
21. Carbon dioxide (CO2) is readily soluble in water, according to
the equation CO2 + H2O → H2CO3. Carbonic acid (H2CO3) is a weak acid.
Respiring cells release CO2. What prediction can we make about the pH
of blood as that blood first comes in contact with respiring
cells?
A) Blood pH will decrease slightly.
B) Blood pH will
increase slightly.
C) Blood pH will remain unchanged.
D)
Blood pH will first increase, then decrease as CO2 combines with
hemoglobin.
E) Blood pH will first decrease, then increase
sharply as CO2 combines with hemoglobin.
A
22. How many electron pairs does carbon share in order to complete
its valence shell?
A) 1
B) 2
C) 3
D) 4
E) 8
D
23. Why are hydrocarbons insoluble in water?
A) The majority of
their bonds are polar covalent carbon-to-hydrogen linkages.
B)
The majority of their bonds are nonpolar covalent carbon-to-hydrogen
linkages.
C) They are hydrophilic.
D) They exhibit
considerable molecular complexity and diversity.
E) They are
lighter than water.
B
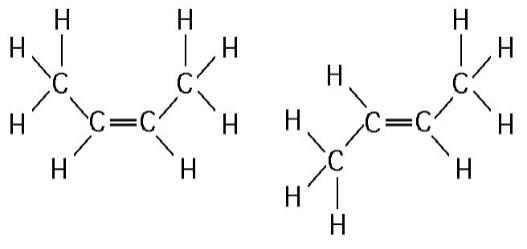
24. The two molecules shown in Figure 4.3 are best described
as
A) enantiomers.
B) radioactive isotopes.
C)
structural isomers.
D) nonisotopic isomers.
E) geometric isomers.
E
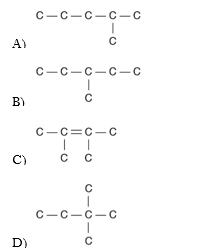
25. Three or four of the following illustrations depict different structural isomers of the organic compound with molecular formula C6H14. For clarity, only the carbon skeletons are shown; hydrogen atoms that would be attached to the carbons have been omitted. Which one, if any, is NOT a structural isomer of this compound?
A)
B)
C)
D)
E) Each of the illustrations in the other answer choices depicts a structural isomer of the compound with molecular formula C6H14.
C
26. A compound contains hydroxyl groups as its predominant functional
group. Which of the following statements is true concerning this
compound?
A) It lacks an asymmetric carbon, and it is probably a
fat or lipid.
B) It should dissolve in water.
C) It should
dissolve in a nonpolar solvent.
D) It won't form hydrogen bonds
with water.
E) It is hydrophobic.
B
27. A chemist wishes to make an organic molecule less acidic. Which
of the following functional groups should be added to the molecule in
order to do so?
A) carboxyl
B) sulfhydryl
C)
hydroxyl
D) amino
E) phosphate
D
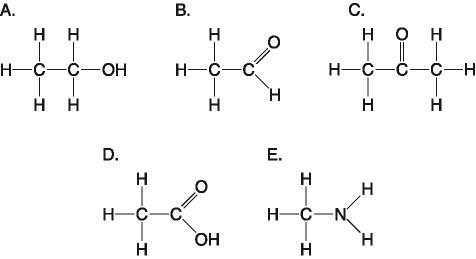
28. Which molecule is an alcohol?
A)
B)
C)
D)
E)
A
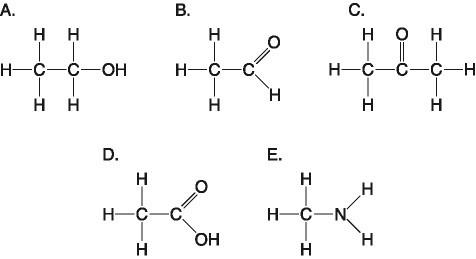
29. Which molecule has a carbonyl functional group in the form of an aldehyde?
A)
B)
C)
D)
E)
B
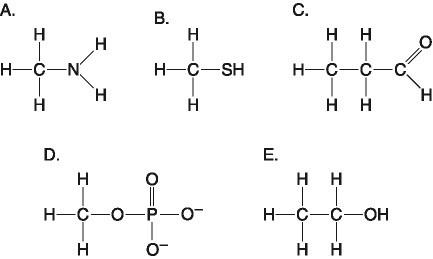
30. Which molecule functions to transfer energy between organic molecules?
A)
B)
C)
D)
E)
D
31. Which molecule is a thiol?
A)
B)
C)
D)
E)
B
32. How many molecules of water are needed to completely hydrolyze a
polymer that is 11 monomers long?
A) 12
B) 11
C)
10
D) 9
E) 8
C
33. The molecular formula for glucose is C6H12O6. What would be the
molecular formula for a molecule made by linking three glucose
molecules together by dehydration reactions?
A) C18H36O18
B)
C18H32O16
C) C6H10O5
D) C18H10O15
E) C3H6O3
B
34. All of the following statements concerning saturated fats are
true except
A) They are more common in animals than in
plants.
B) They have multiple double bonds in the carbon chains
of their fatty acids.
C) They generally solidify at room
temperature.
D) They contain more hydrogen than unsaturated fats
having the same number of carbon atoms.
E) They are one of
several factors that contribute to atherosclerosis.
B
35. All of the following contain amino acids except
A)
hemoglobin.
B) cholesterol.
C) antibodies.
D)
enzymes.
E) insulin.
B
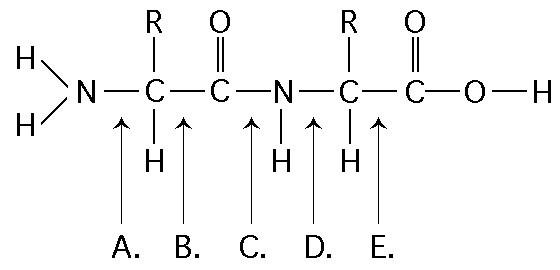
36. Which bond is a peptide bond?
A)
B)
C)
D)
E)
C
37. The tertiary structure of a protein is the
A) bonding
together of several polypeptide chains by weak bonds.
B) order in
which amino acids are joined in a polypeptide chain.
C) unique
three-dimensional shape of the fully folded polypeptide.
D)
organization of a polypeptide chain into an α helix or β pleated
sheet.
E) overall protein structure resulting from the
aggregation of two or more polypeptide subunits.
C
38. The R group or side chain of the amino acid serine is —CH2OH. The
R group or side chain of the amino acid alanine is —CH3. Where would
you expect to find these amino acids in a globular protein in aqueous
solution?
A) Serine would be in the interior, and alanine would
be on the exterior of the globular protein.
B) Alanine would be
in the interior, and serine would be on the exterior of the globular
protein.
C) Both serine and alanine would be in the interior of
the globular protein.
D) Both serine and alanine would be on the
exterior of the globular protein.
E) Both serine and alanine
would be in the interior and on the exterior of the globular protein.
B
39. If one strand of a DNA molecule has the sequence of bases
5'ATTGCA3', the other complementary strand would have the
sequence
A) 5'TAACGT3'.
B) 3'TAACGT5'.
C)
5'UAACGU3'.
D) 3'UAACGU5'.
E) 5'UGCAAU3'.
B
40. When biologists wish to study the internal ultrastructure of
cells, they most likely would use
A) a light microscope.
B)
a scanning electron microscope.
C) a transmission electronic
microscope.
D) A and B
E) B and C
C
41. Which of the following correctly lists the order in which
cellular components will be found in the pellet when homogenized cells
are treated with increasingly rapid spins in a centrifuge?
A)
ribosomes, nucleus, mitochondria
B) chloroplasts, ribosomes,
vacuoles
C) nucleus, ribosomes, chloroplasts
D) vacuoles,
ribosomes, nucleus
E) nucleus, mitochondria, ribosomes
E
42. Which of the following is a major cause of the size limits for
certain types of cells?
A) the evolution of larger cells after
the evolution of smaller cells
B) the difference in plasma
membranes between prokaryotes and eukaryotes
C) the evolution of
eukaryotes after the evolution of prokaryotes
D) the need for a
surface area of sufficient area to allow the cell's function
E)
the observation that longer cells usually have greater cell volume
D
43. Which structure is the site of the synthesis of proteins that may
be exported from the cell?
A) rough ER
B) lysosomes
C)
plasmodesmata
D) Golgi vesicles
E) tight junctions
A
44. The liver is involved in detoxification of many poisons and
drugs. Which of the following structures is primarily involved in this
process and therefore abundant in liver cells?
A) rough
ER
B) smooth ER
C) Golgi apparatus
D) Nuclear
envelope
E) Transport vesicles
B
45. Why isn't the mitochondrion classified as part of the
endomembrane system?
A) It only has two membrane layers.
B)
Its structure is not derived from the ER.
C) It has too many
vesicles.
D) It is not involved in protein synthesis.
E) It
is not attached to the outer nuclear envelope.
B
46. Centrioles, cilia, flagella, and basal bodies have remarkably
similar structural elements and arrangements. This leads us to which
of the following as a probable hypothesis?
A) Disruption of one
of these types of structure should necessarily disrupt each of the
others as well.
B) Loss of basal bodies should lead to loss of
all cilia, flagella, and centrioles.
C) Motor proteins such as
dynein must have evolved before any of these four kinds of
structure.
D) Evolution of motility, of cells or of parts of
cells, must have occurred only once.
E) Natural selection for
motility must select for microtubular arrays in circular patterns.
E
47. Which of the following makes it necessary for animal cells,
although they have no cell walls, to have intercellular
junctions?
A) Cell membranes do not distinguish the types of ions
and molecules passing through them.
B) Large molecules,
especially proteins, do not readily get through one, much less two
adjacent cell membranes.
C) Cell-to-cell communication requires
physical attachment of one cell to another.
D) Maintenance of
connective tissue shape requires cells to adhere to one
another.
E) The relative shapelessness of animal cells requires a
mechanism for keeping the cells aligned.
B
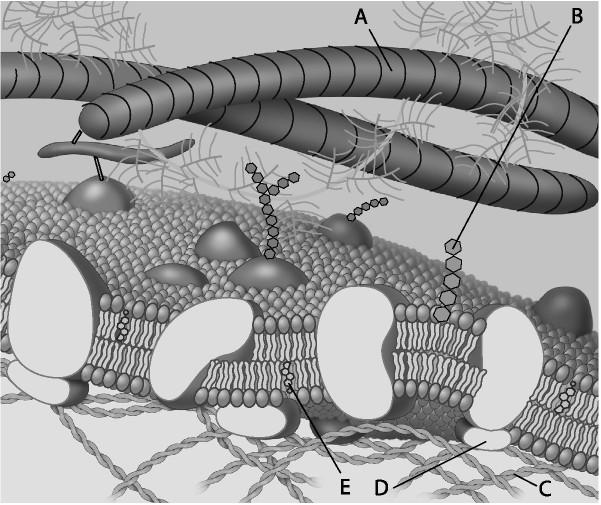
48. peripheral protein
A)
B)
C)
D)
E)
D
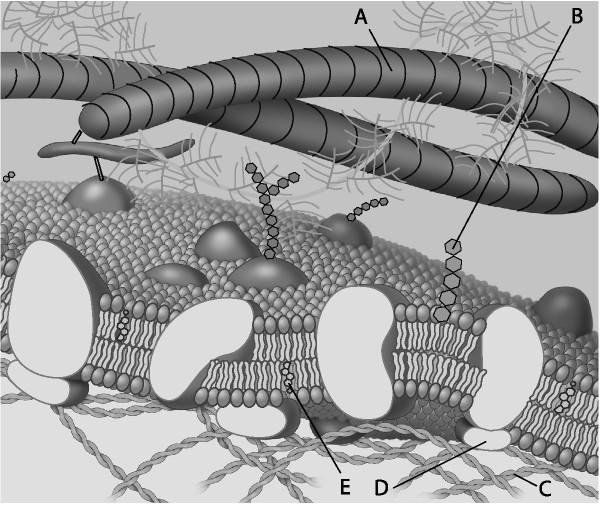
49. glycolipid
A)
B)
C)
D)
E)
B
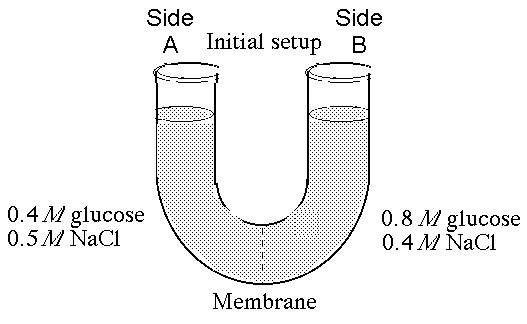
The solutions in the arms of a U-tube are separated at the bottom of the tube by a selectively permeable membrane. The membrane is permeable to sodium chloride but not to glucose. Side A is filled with a solution of 0.4 M glucose and 0.5 M sodium chloride (NaCl), and side B is filled with a solution containing 0.8 M glucose and 0.4 M sodium chloride. Initially, the volume in both arms is the same.
50. At the beginning of the experiment,
A) side A is hypertonic
to side B.
B) side A is hypotonic to side B.
C) side A is
isotonic to side B.
D) side A is hypertonic to side B with
respect to glucose.
E) side A is hypotonic to side B with respect
to sodium chloride.
B
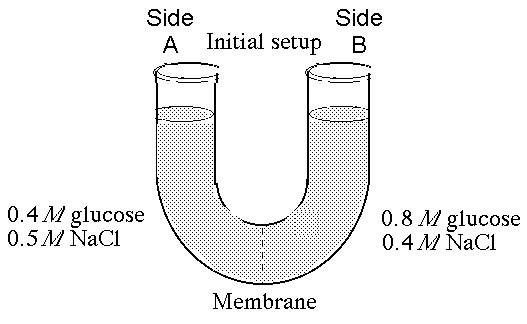
The solutions in the arms of a U-tube are separated at the bottom of the tube by a selectively permeable membrane. The membrane is permeable to sodium chloride but not to glucose. Side A is filled with a solution of 0.4 M glucose and 0.5 M sodium chloride (NaCl), and side B is filled with a solution containing 0.8 M glucose and 0.4 M sodium chloride. Initially, the volume in both arms is the same.
51. If you examine side A after 3 days, you should find
A) a
decrease in the concentration of NaCl and glucose and an increase in
the water level.
B) a decrease in the concentration of NaCl, an
increase in water level, and no change in the concentration of
glucose.
C) no net change in the system.
D) a decrease in
the concentration of NaCl and a decrease in the water level.
E)
no change in the concentration of NaCl and glucose and an increase in
the water level.
D
52. Which of the following membrane activities require energy from
ATP hydrolysis?
A) facilitated diffusion.
B) movement of
water into a cell
C) Na+ ions moving out of the cell
D)
movement of glucose molecules
E) movement of water into a paramecium
C
53. Which of the following is (are) true for anabolic pathways?
A) They do not depend on enzymes.
B) They are usually
highly spontaneous chemical reactions.
C) They consume energy to
build up polymers from monomers.
D) They release energy as they
degrade polymers to monomers.
C
54. Whenever energy is transformed, there is always an increase in
the
A) free energy of the system.
B) free energy of the
universe.
C) entropy of the system.
D) entropy of the
universe.
E) enthalpy of the universe.
D
55. Which of the following is an example of potential rather than
kinetic energy?
A) a boy mowing grass
B) water rushing over
Niagara Falls
C) a firefly using light flashes to attract a
mate
D) a food molecule made up of energy-rich
macromolecules
E) an insect foraging for food
D
56. Which of the following shows the correct changes in thermodynamic
properties for a chemical reaction in which amino acids are linked to
form a protein?
A) +△H, +△S, +△G
B) +△H, -△S, -△G
C)
+△H, -△S, +△G
D) -△H, -△S, +△G
E) -△H, +△S, +△G
C
57. What term is used to describe the transfer of free energy from
catabolic pathways to anabolic pathways?
A) feedback regulation
B) bioenergetics
C) energy coupling
D) entropy
E) cooperativity
C
58. When ATP releases some energy, it also releases inorganic
phosphate. What purpose does this serve (if any) in the cell?
A)
It is released as an excretory waste.
B) It can only be used to
regenerate more ATP.
C) It can be added to water and excreted as
a liquid.
D) It can be added to other molecules in order to
activate them.
E) It can enter the nucleus to affect gene expression.
D
59. A solution of starch at room temperature does not readily
decompose to form a solution of simple sugars because
A) the
starch solution has less free energy than the sugar solution.
B)
the hydrolysis of starch to sugar is endergonic.
C) the
activation energy barrier for this reaction cannot be surmounted.
D) starch cannot be hydrolyzed in the presence of so much
water.
E) starch hydrolysis is nonspontaneous.
C
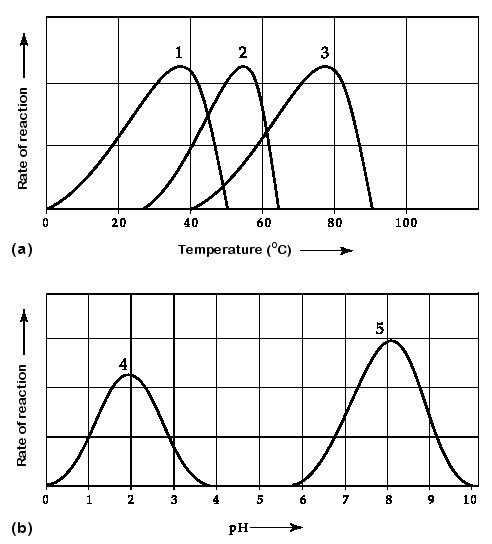
60. Which curve represents the behavior of an enzyme taken from a
bacterium that lives in hot springs at temperatures of 70°C or higher?
A) curve 1
B) curve 2
C) curve 3
D) curve 4
E) curve 5
C
61. When you have a severe fever, what may be a grave consequence if
this is not controlled?
A) destruction of your enzymes' primary
structure
B) removal of amine groups from your proteins
C)
change in the folding of enzymes
D) removal of the amino acids in
active sites
E) binding of enzymes to inappropriate substrates
C
62. An important group of peripheral membrane proteins are enzymes,
such as the phospholipases that attack the head groups of
phospholipids leading to the degradation of damaged membranes. What
properties must these enzymes exhibit?
A) resistance to
degradation
B) independence from cofactor interaction
C)
water solubility
D) lipid solubility
E) membrane spanning domains
C
63. When electrons move closer to a more electronegative atom, what
happens?
A) Energy is released.
B) Energy is
consumed.
C) The more electronegative atom is reduced.
D)
The more electronegative atom is oxidized.
E) A and C are correct.
E
64. Which of the following statements describes NAD+?
A) NAD+ is
reduced to NADH during both glycolysis and the citric acid
cycle.
B) NAD+ has more chemical energy than NADH.
C) NAD+
is reduced by the action of hydrogenases.
D) NAD+ can donate
electrons for use in oxidative phosphorylation.
E) In the absence
of NAD+, glycolysis can still function.
A
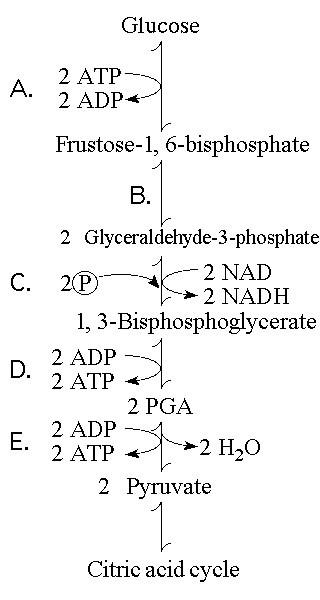
65. Which step shows a split of one molecule into two smaller molecules?
A)
B)
C)
D)
E)
B
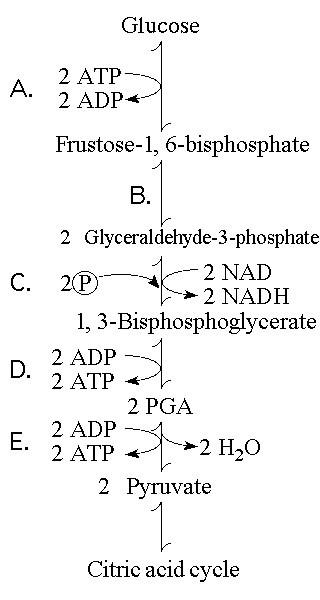
66. Which step involves an endergonic reaction?
A)
B)
C)
D)
E)
A
67. In glycolysis, for each molecule of glucose oxidized to
pyruvate
A) 2 molecules of ATP are used and 2 molecules of ATP
are produced.
B) 2 molecules of ATP are used and 4 molecules of
ATP are produced.
C) 4 molecules of ATP are used and 2 molecules
of ATP are produced.
D) 2 molecules of ATP are used and 6
molecules of ATP are produced.
E) 6 molecules of ATP are used and
6 molecules of ATP are produced.
B
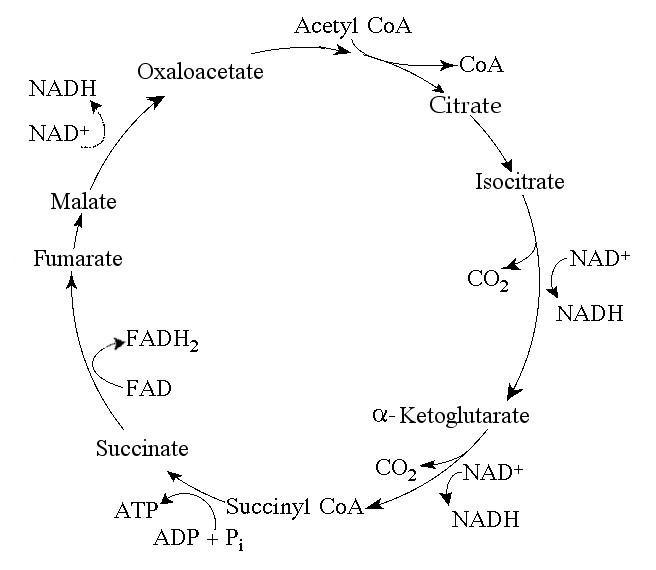
68. Starting with one molecule of isocitrate and ending with
fumarate, what is the maximum number of ATP molecules that could be
made through substrate-level phosphorylation?
A) 1
B)
2
C) 11
D) 12
E) 24
A
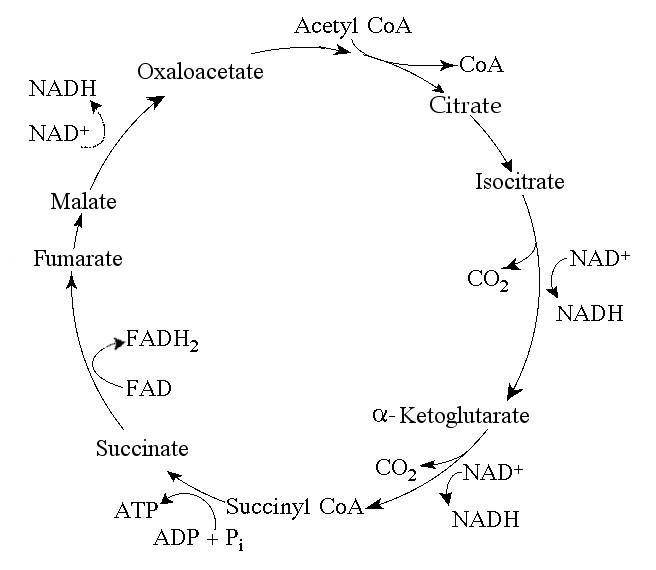
69. How many molecules of carbon dioxide (CO2) would be produced by
five turns of the citric acid cycle?
A) 2
B) 5
C)
10
D) 12
E) 60
C
70. During aerobic respiration, electrons travel downhill in which
sequence?
A) food → citric acid cycle → ATP → NAD+
B) food →
NADH → electron transport chain → oxygen
C) glucose → pyruvate →
ATP → oxygen
D) glucose → ATP → electron transport chain →
NADH
E) food → glycolysis → citric acid cycle → NADH → ATP
B
71. During aerobic respiration, which of the following directly
donates electrons to the electron transport chain at the lowest energy
level?
A) NAD+
B) NADH
C) ATP
D) ADP + Pi
E) FADH2
E
72. Which metabolic pathway is common to both cellular respiration
and fermentation?
A) the oxidation of pyruvate to acetyl
CoA
B) the citric acid cycle
C) oxidative
phosphorylation
D) glycolysis
E) chemiosmosis
D
73. Even though plants carry on photosynthesis, plant cells still use
their mitochondria for oxidation of pyruvate. When and where will this
occur?
A) in photosynthetic cells in the light, while
photosynthesis occurs concurrently
B) in non-photosynthesizing
cells only
C) in cells that are storing glucose only
D) in
photosynthesizing cells in dark periods and in other tissues all the
time
E) in photosynthesizing cells in the light and in other
tissues in the dark
D
74. Which of the following are products of the light reactions of
photosynthesis that are utilized in the Calvin cycle?
A) CO2 and
glucose
B) H2O and O2
C) ADP, Pi, and NADP+
D)
electrons and H+
E) ATP and NADPH
E
75. When oxygen is released as a result of photosynthesis, it is a
by-product of which of the following?
A) reducing NADP+
B)
splitting the water molecules
C) chemiosmosis
D) the
electron transfer system of photosystem I
E) the electron
transfer system of photosystem II
B
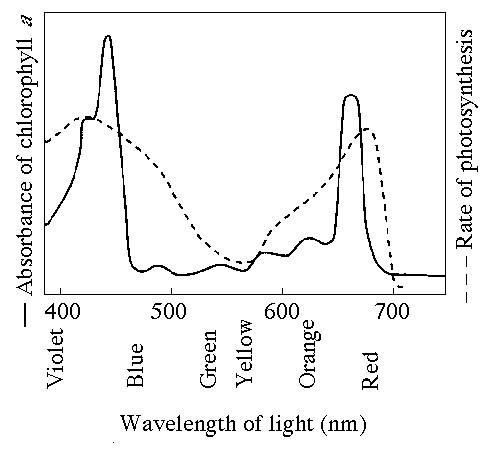
76. Figure 10.1 shows the absorption spectrum for chlorophyll a and
the action spectrum for photosynthesis. Why are they
different?
A) Green and yellow wavelengths inhibit the absorption
of red and blue wavelengths.
B) Bright sunlight destroys
photosynthetic pigments.
C) Oxygen given off during
photosynthesis interferes with the absorption of light.
D) Other
pigments absorb light in addition to chlorophyll a.
E) Aerobic
bacteria take up oxygen which changes the measurement of the rate of photosynthesis.
D
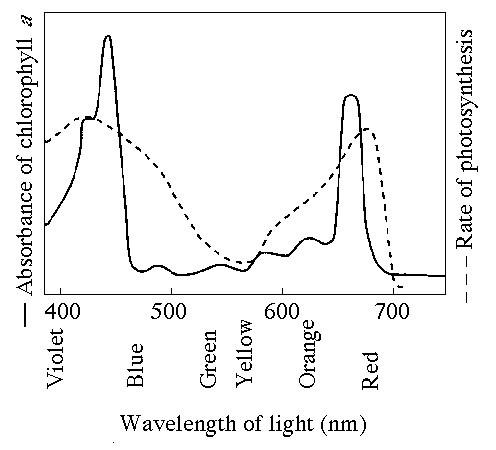
77. Compared with the lines for chlorophyll a in the figure, where
would you expect to find the lines to differ for chlorophyll
b?
A) The absorption spectrum line would be lowest for
chlorophyll b somewhat to the right of that for chlorophyll a
(500—600).
B) The rate of photosynthesis line for chlorophyll b
would be lowest from 600—700 nm.
C) The lines for the two types
of chlorophyll would be almost completely opposite.
D) The lines
for the two types of chlorophyll would be almost completely
identical.
E) The peaks of the line for absorbance of b would be
shifted to the left, and for rate of photosynthesis would be shifted
to the right.
A
78. What is the relationship between wavelength of light and the
quantity of energy per photon?
A) They have a direct, linear
relationship.
B) They are inversely related.
C) They are
logarithmically related.
D) They are separate phenomena.
E)
They are only related in certain parts of the spectrum.
B
79. The sugar that results from three "turns" of the Calvin
cycle is glyceraldehyde-3-phosphate (G3P). Which of the following is a
consequence of this?
A) Formation of a molecule of glucose would
require 9 "turns."
B) G3P more readily forms sucrose
and other disaccharides than it does monosaccharides.
C) Some
plants would not taste sweet to us.
D) The formation of starch in
plants involves assembling many G3P molecules, with or without further
rearrangements.
E) G3P is easier for a plant to store.
D
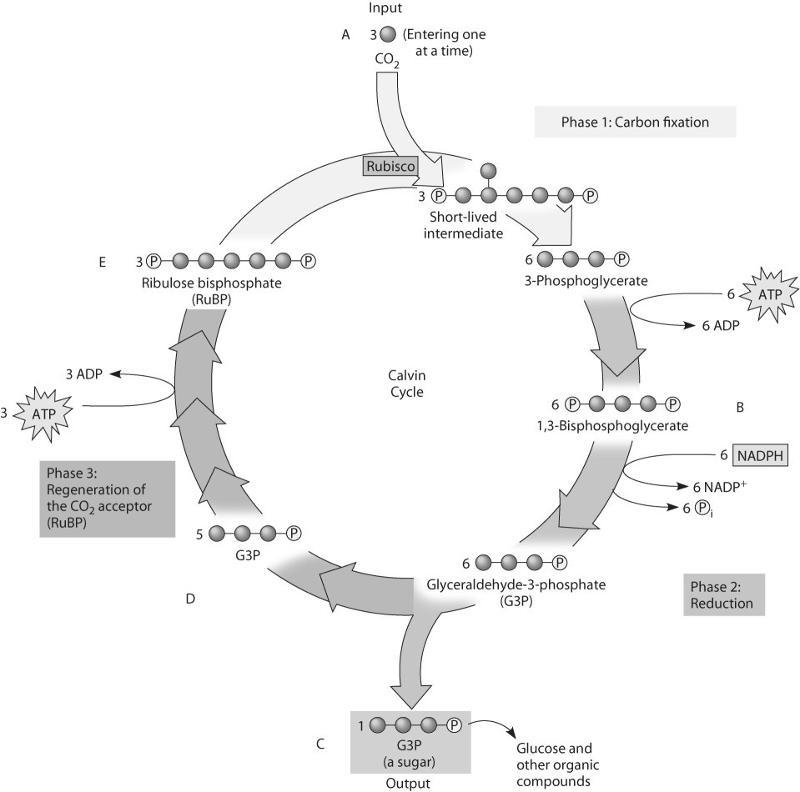
80. If ATP used by this plant is labeled with radioactive phosphorus,
in which molecules will the radioactivity be measurable after one
"turn" of the cycle?
A) in B only
B) in B and C
only
C) in B, C, and D only
D) in B and E only
E) in B,
C, D, and E
D
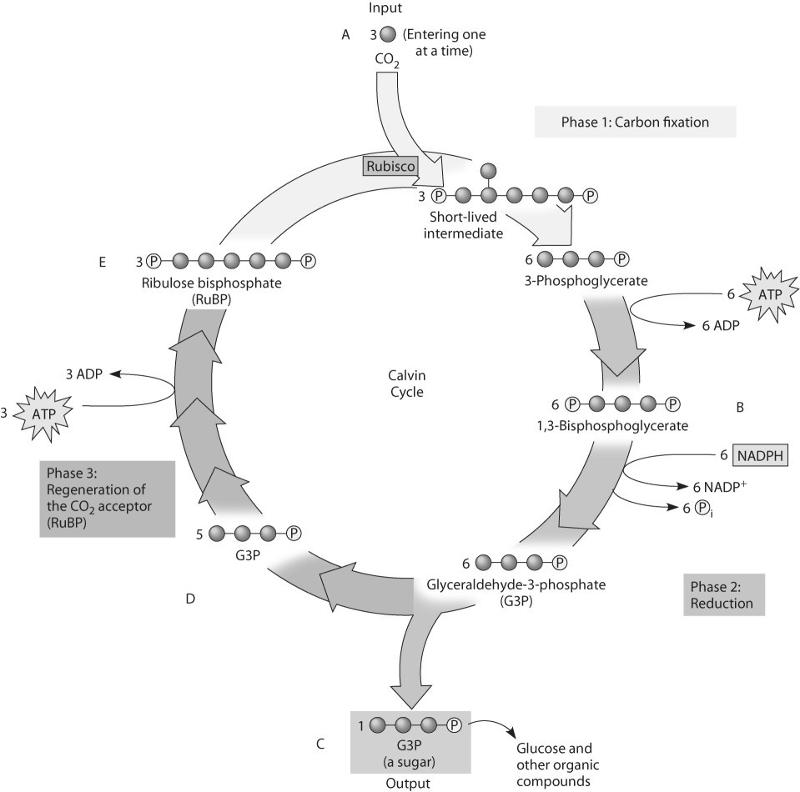
81. Which molecule(s) of the Calvin cycle is/are most like molecules
found in glycolysis?
A) A, B, C, and E
B) B, C, and
E
C) A only
D) C and D only
E) E only
D
82. In an experiment studying photosynthesis performed during the
day, you provide a plant with radioactive carbon (14C) dioxide as a
metabolic tracer. The 14C is incorporated first into oxaloacetate. The
plant is best characterized as a
A) C4 plant.
B) C3
plant.
C) CAM plant.
D) heterotroph.
E) chemoautotroph.
A
83. Synaptic signaling between adjacent neurons is like hormone
signaling in which of the following ways?
A) It sends its signal
molecules through the blood.
B) It sends its signal molecules
quite a distance.
C) It requires calcium ions.
D) It
requires binding of a signaling molecule to a receptor.
E) It
persists over a long period.
D
84. What would be true for the signaling system in an animal cell
that lacks the ability to produce GTP?
A) It would not be able to
activate and inactivate the G protein on the cytoplasmic side of the
plasma membrane.
B) It could activate only the epinephrine
system.
C) It would be able to carry out reception and
transduction, but would not be able to respond to a signal.
D)
Only A and C are true.
E) A, B, and C are true.
A
85. Which is true of transcription factors?
A) They regulate the
synthesis of DNA in response to a signal.
B) Some transcribe ATP
into cAMP.
C) They initiate the epinephrine response in animal
cells.
D) They control which genes are expressed.
E) They
are needed to regulate the synthesis of lipids in the cytoplasm.
D
86. The general name for an enzyme that transfers phosphate groups
from ATP to a protein is
A) phosphorylase.
B)
phosphatase.
C) protein kinase.
D) ATPase.
E) protease.
C
87. Which of the following is a correct association?
A) kinase
activity and the addition of a tyrosine
B) phosphodiesterase
activity and the removal of phosphate groups
C) GTPase activity
and hydrolysis of GTP to GDP
D) phosphorylase activity and the
catabolism of glucose
E) adenylyl cyclase activity and the
conversion of cAMP to AMP
C
88. At puberty, an adolescent female body changes in both structure
and function of several organ systems, primarily under the influence
of changing concentrations of estrogens and other steroid hormones.
How can one hormone, such as estrogen, mediate so many
effects?
A) Estrogen is produced in very large concentration and
therefore diffuses widely.
B) Estrogen has specific receptors
inside several cell types, but each cell responds in the same way to
its binding.
C) Estrogen is kept away from the surface of any
cells not able to bind it at the surface.
D) Estrogen binds to
specific receptors inside many kinds of cells, each of which have
different responses to its binding.
E) Estrogen has different
shaped receptors for each of several cell types.
D
89. If an adult person has a faulty version of the human-analog to
ced-4 of the nematode, which of the following might more likely
result?
A) neurodegeneration
B) activation of a
developmental pathway found in the worm but not in humans
C) a
form of cancer in which there is insufficient apoptosis
D)
webbing of fingers or toes
E) excess skin exfoliation
C
90. Starting with a fertilized egg (zygote), a series of five cell
divisions would produce an early embryo with how many cells?
A)
4
B) 8
C) 16
D) 32
E) 64
D
91. How do the daughter cells at the end of mitosis and cytokinesis
compare with their parent cell when it was in G1 of the cell
cycle?
A) The daughter cells have half the amount of cytoplasm
and half the amount of DNA.
B) The daughter cells have half the
number of chromosomes and half the amount of DNA.
C) The daughter
cells have the same number of chromosomes and half the amount of
DNA.
D) The daughter cells have the same number of chromosomes
and the same amount of DNA.
E) The daughter cells have the same
number of chromosomes and twice the amount of DNA.
D
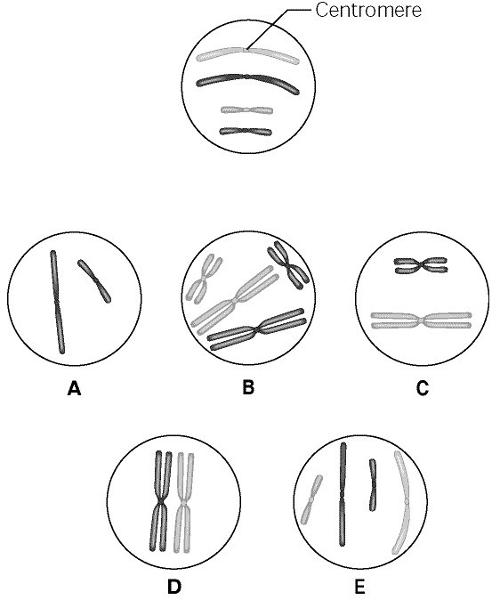
The lettered circle in Figure 12.1 shows a diploid nucleus with four chromosomes. There are two pairs of homologous chromosomes, one long and the other short. One haploid set is symbolized as black and the other haploid set is gray. The chromosomes in the unlettered circle have not yet replicated. Choose the correct chromosomal conditions for the following stages.
92. What is the first stage of mitosis, in which centrosomes are in
opposite poles of the cell?
A) telophase
B) anaphase
C)
prometaphase
D) metaphase
E) prophase
C
93. Which term describes centromeres uncoupling, sister chromatids
separating, and the two new chromosomes moving to opposite poles of
the cell?
A) telophase
B) anaphase
C)
prometaphase
D) metaphase
E) prophase
B
94. A cell containing 92 chromatids at metaphase of mitosis would, at
its completion, produce two nuclei each containing how many
chromosomes?
A) 12
B) 16
C) 23
D) 46
E) 92
D
95. If there are 20 centromeres in a cell at anaphase, how many
chromosomes are there in each daughter cell following
cytokinesis?
A) 10
B) 20
C) 30
D) 40
E) 80
A
96. A group of cells is assayed for DNA content immediately following
mitosis and is found to have an average of 8 picograms of DNA per
nucleus. Those cells would have __________ picograms at the end of the
S phase and __________ picograms at the end of G2.
A) 8;
8
B) 8; 16
C) 16; 8
D) 16; 16
E) 12; 16
D
97. Which of the following triggers the cell's passage past the G2
checkpoint into mitosis?
A) PDGF
B) MPF
C) protein
kinase
D) cyclin
E) Cdk
B
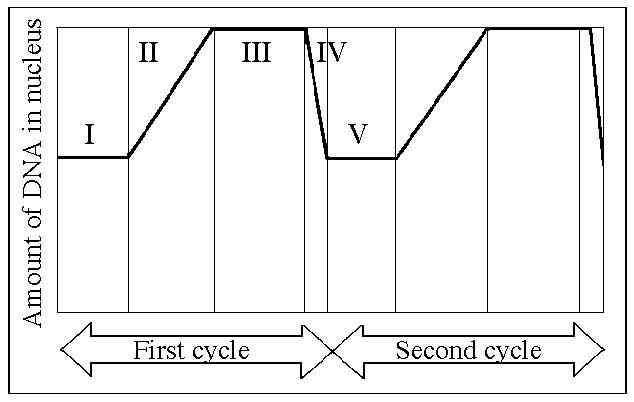
98. In the figure above, mitosis is represented by which
number?
A) I
B) II
C) III
D) IV
E) V
D
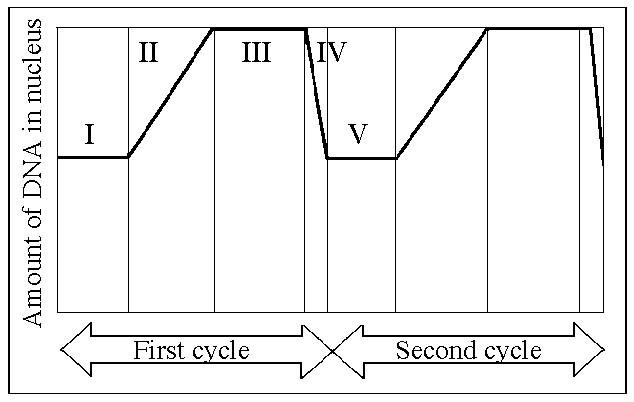
99. Which number represents DNA synthesis?
A) I
B)
II
C) III
D) IV
E) V
B
100. A particular cyclin called cyclin E forms a complex with a
cyclin-dependent kinase called Cdk 2. This complex is important for
the progression of the cell from G1 into the S phase of the cell
cycle. Which of the following statements is correct?
A) The
amount of cyclin E is greatest during the S phase.
B) The amount
of Cdk 2 is greater during G1 compared to the S phase.
C) The
amount of cyclin E is highest during G1.
D) The amount of Cdk 2
is greatest during G1.
E) The activity of the cyclin E/Cdk 2
complex is highest during G2.
C
101. Besides the ability of some cancer cells to overproliferate,
what else could logically result in a tumor?
A)
metastasis
B) changes in the order of cell cycle stages
C)
lack of appropriate cell death
D) inability to form
spindles
E) inability of chromosomes to meet at the metaphase plate
C
102. Cytosine makes up 38% of the nucleotides in a sample of DNA from
an organism. Approximately what percentage of the nucleotides in this
sample will be thymine?
A) 12
B) 24
C) 31
D)
38
E) It cannot be determined from the information provided.
A
103. In an analysis of the nucleotide composition of DNA, which of
the following will be found?
A) A = C
B) A = G and C =
T
C) A + C = G + T
D) G + C = T + A
C
104. What is meant by the description "antiparallel"
regarding the strands that make up DNA?
A) The twisting nature of
DNA creates nonparallel strands.
B) The 5' to 3' direction of one
strand runs counter to the 5' to 3' direction of the other
strand.
C) Base pairings create unequal spacing between the two
DNA strands.
D) One strand is positively charged and the other is
negatively charged.
E) One strand contains only purines and the
other contains only pyrimidines.
B
105. Eukaryotic telomeres replicate differently than the rest of the
chromosome. This is a consequence of which of the following?
A)
The evolution of telomerase enzyme
B) DNA polymerase that cannot
replicate the leading strand template to its 5' end
C) Gaps left
at the 5' end of the lagging strand because of the need for a 3' onto
which nucleotides can attach
D) Gaps left at the 3' end of the
lagging strand because of the need for a primer
E) The "no
ends" of a circular chromosome
C
106. At a specific area of a chromosome, the sequence of nucleotides
below is present where the chain opens to form a replication
fork:
3' C C T A G G C T G C A A T C C 5'
An RNA primer is
formed starting at the underlined T (T) of the template. Which of the
following represents the primer sequence?
A) 5' G C C T A G G
3'
B) 3' G C C T A G G 5'
C) 5' A C G T T A G G 3'
D)
5' A C G U U A G G 3'
E) 5' G C C U A G G 3'
D
107. What is the function of DNA polymerase III?
A) to unwind
the DNA helix during replication
B) to seal together the broken
ends of DNA strands
C) to add nucleotides to the end of a growing
DNA strand
D) to degrade damaged DNA molecules
E) to rejoin
the two DNA strands (one new and one old) after replication
C
108. Which of the following removes the RNA nucleotides from the
primer and adds equivalent DNA nucleotides to the 3' end of Okazaki
fragments?
A) helicase
B) DNA polymerase III
C)
ligase
D) DNA polymerase I
E) primase
D
109. A new DNA strand elongates only in the 5' to 3' direction
because
A) DNA polymerase begins adding nucleotides at the 5' end
of the template.
B) Okazaki fragments prevent elongation in the
3' to 5' direction.
C) the polarity of the DNA molecule prevents
addition of nucleotides at the 3' end.
D) replication must
progress toward the replication fork.
E) DNA polymerase can only
add nucleotides to the free 3' end.
E
110. When DNA is compacted by histones into 10 nm and 30 nm fibers,
the DNA is unable to interact with proteins required for gene
expression. Therefore, to allow for these proteins to act, the
chromatin must constantly alter its structure. Which processes
contribute to this dynamic activity?
A) DNA supercoiling at or
around H1
B) methylation and phosphorylation of histone
tails
C) hydrolysis of DNA molecules where they are wrapped
around the nucleosome core
D) accessibility of heterochromatin to
phosphorylating enzymes
E) nucleotide excision and reconstruction
B
111. If a cell were unable to produce histone proteins, which of the
following would be a likely effect?
A) There would be an increase
in the amount of "satellite" DNA produced during
centrifugation.
B) The cell's DNA couldn't be packed into its
nucleus.
C) Spindle fibers would not form during
prophase.
D) Amplification of other genes would compensate for
the lack of histones.
E) Pseudogenes would be transcribed to
compensate for the decreased protein in the cell.
B
112. Which of the following represents the order of increasingly
higher levels of organization of chromatin?
A) nucleosome, 30-nm
chromatin fiber, looped domain
B) looped domain, 30-nm chromatin
fiber, nucleosome
C) looped domain, nucleosome, 30-nm chromatin
fiber
D) nucleosome, looped domain, 30-nm chromatin fiber
E)
30-nm chromatin fiber, nucleosome, looped domain
A
113. The enzyme polynucleotide phosphorylase randomly assembles
nucleotides into a polynucleotide polymer. You add polynucleotide
phosphorylase to a solution of adenosine triphosphate and guanosine
triphosphate. How many artificial mRNA 3 nucleotide codons would be
possible?
A) 3
B) 4
C) 8
D) 16
E) 64
C
114. A particular triplet of bases in the template strand of DNA is
5' AGT 3'. The corresponding codon for the mRNA transcribed is
A)
3' UCA 5'.
B) 3' UGA 5'.
C) 5' TCA 3'.
D) 3'ACU
5'.
E) either UCA or TCA, depending on wobble in the first base.
A
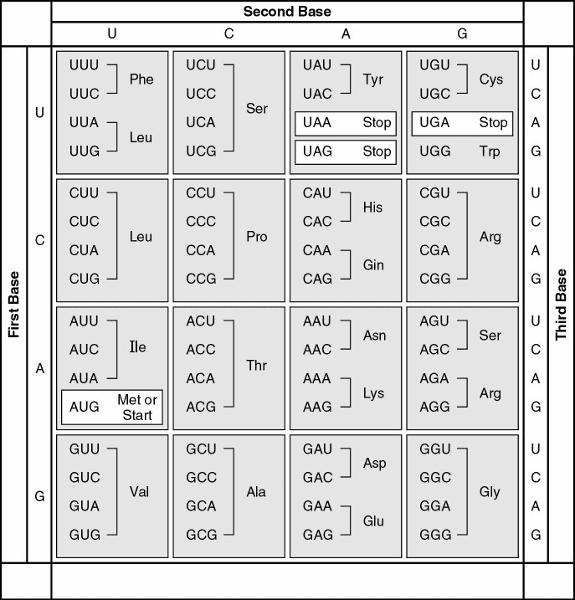
115. A possible sequence of nucleotides in the template strand of DNA
that would code for the polypeptide sequence phe-leu-ile-val would
be
A) 5' TTG-CTA-CAG-TAG 3'.
B) 3' AAC-GAC-GUC-AUA
5'.
C) 5' AUG-CTG-CAG-TAT 3'.
D) 3' AAA-AAT-ATA-ACA
5'.
E) 3' AAA-GAA-TAA-CAA 5'.
E
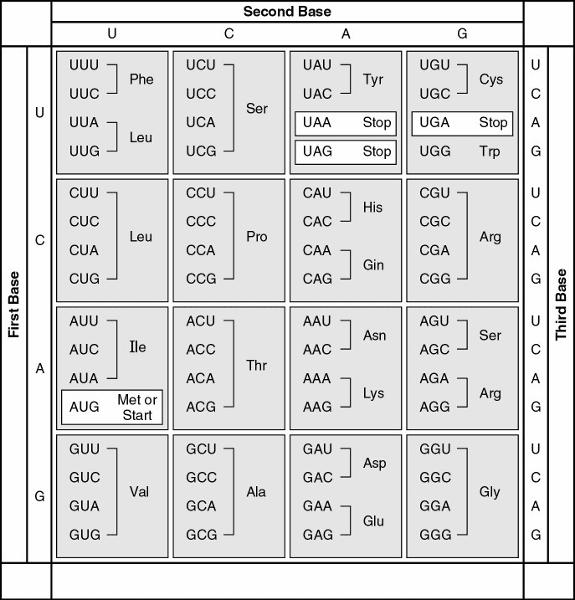
116. A peptide has the sequence NH2-phe-pro-lys-gly-phe-pro-COOH.
Which of the following sequences in the coding strand of the DNA could
code for this peptide?
A) 3' UUU-CCC-AAA-GGG-UUU-CCC
B) 3'
AUG-AAA-GGG-TTT-CCC-AAA-GGG
C) 5' TTT-CCC-AAA-GGG-TTT-CCC
D)
5' GGG-AAA-TTT-AAA-CCC-ACT-GGG
E) 5' ACT-TAC-CAT-AAA-CAT-TAC-UGA
C
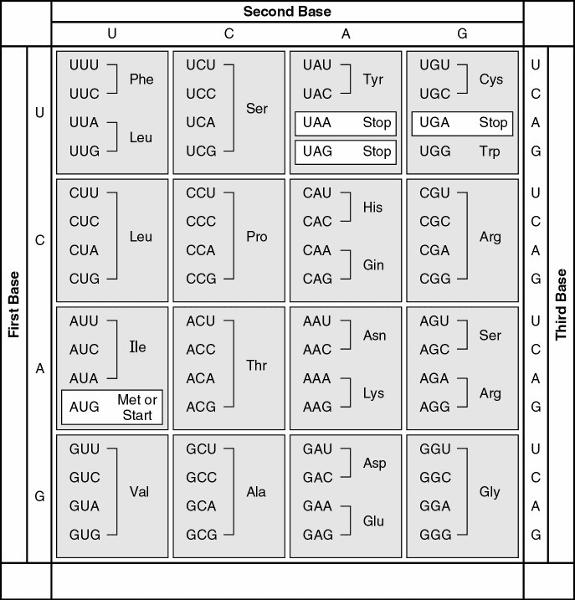
117. What is the sequence of a peptide based on the following mRNA
sequence?
5' . . . UUUUCUUAUUGUCUU 3'
A)
leu-cys-tyr-ser-phe
B) cyc-phe-tyr-cys-leu
C)
phe-leu-ile-met-val
D) leu-pro-asp-lys-gly
E) phe-ser-tyr-cys-leu
E
118. In which of the following actions does RNA polymerase differ
from DNA polymerase?
A) RNA polymerase uses RNA as a template,
and DNA polymerase uses a DNA template.
B) RNA polymerase binds
to single-stranded DNA, and DNA polymerase binds to double-stranded
DNA.
C) RNA polymerase is much more accurate than DNA
polymerase.
D) RNA polymerase can initiate RNA synthesis, but DNA
polymerase requires a primer to initiate DNA synthesis.
E) RNA
polymerase does not need to separate the two strands of DNA in order
to synthesize an RNA copy, whereas DNA polymerase must unwind the
double helix before it can replicate the DNA.
D
119. Which of the following help(s) to stabilize mRNA by inhibiting
its degradation?
A) TATA box
B) spliceosomes
C) 5' cap
and poly (A) tail
D) introns
E) RNA polymerase
C
120. A mutation in which of the following parts of a gene is likely
to be most damaging to a cell?
A) intron
B) exon
C) 5'
UTR
D) 3' UTR
E) All would be equally damaging.
B
121. Alternative RNA splicing
A) is a mechanism for increasing
the rate of transcription.
B) can allow the production of
proteins of different sizes from a single mRNA.
C) can allow the
production of similar proteins from different RNAs.
D) increases
the rate of transcription.
E) is due to the presence or absence
of particular snRNPs.
B
122. A particular triplet of bases in the coding sequence of DNA is
AAA. The anticodon on the tRNA that binds the mRNA codon is
A)
TTT.
B) UUA.
C) UUU.
D) AAA.
E) either UAA or TAA,
depending on first base wobble.
C
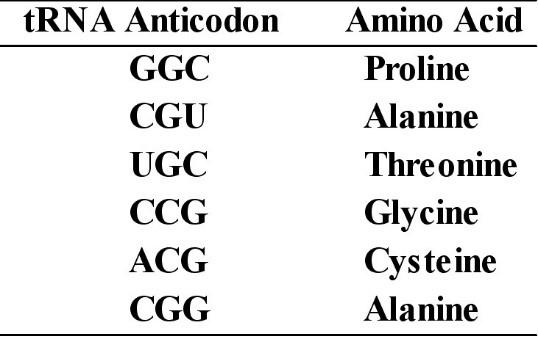
123. A part of an mRNA molecule with the following sequence is being read by a ribosome: 5' CCG-ACG 3' (mRNA). The following charged transfer RNA molecules (with their anticodons shown in the 3' to 5' direction) are available. Two of them can correctly match the mRNA so that a dipeptide can form.
The dipeptide that will form will be
A)
cysteine-alanine.
B) proline-threonine.
C)
glycine-cysteine.
D) alanine-alanine.
E) threonine-glycine.
B
124. As a ribosome translocates along an mRNA molecule by one codon,
which of the following occurs?
A) The tRNA that was in the A site
moves into the P site.
B) The tRNA that was in the P site moves
into the A site.
C) The tRNA that was in the A site moves to the
E site and is released.
D) The tRNA that was in the A site
departs from the ribosome via a tunnel.
E) The polypeptide enters
the E site.
A
125. Each of the following options is a modification of the sentence
THECATATETHERAT. Which of the following is analogous to a frameshift
mutation?
A) THERATATETHECAT
B) THETACATETHERAT
C)
THECATARETHERAT
D) THECATATTHERAT
E) CATATETHERAT
D
126. Which of the following DNA mutations is the most likely to be
damaging to the protein it specifies?
A) a base-pair
deletion
B) a codon substitution
C) a substitution in the
last base of a codon
D) a codon deletion
E) a point mutation
A
127. The role of a metabolite that controls a repressible operon is
to
A) bind to the promoter region and decrease the affinity of
RNA polymerase for the promoter.
B) bind to the operator region
and block the attachment of RNA polymerase to the promoter.
C)
increase the production of inactive repressor proteins.
D) bind
to the repressor protein and inactivate it.
E) bind to the
repressor protein and activate it.
E
128. When this is taken up by the cell, it binds to the repressor so
that the repressor no longer binds to the operator:
A)
operon
B) inducer
C) promoter
D) repressor
E) corepressor
B
129. Muscle cells and nerve cells in one species of animal owe their
differences in structure to
A) having different genes.
B)
having different chromosomes.
C) using different genetic
codes.
D) having different genes expressed.
E) having unique ribosomes.
D
130. Genomic imprinting, DNA methylation, and histone acetylation are
all examples of
A) genetic mutation.
B) chromosomal
rearrangements.
C) karyotypes.
D) epigenetic
phenomena.
E) translocation.
D
131. Eukaryotic cells can control gene expression by which of the
following mechanisms?
A) histone acetylation of
nucleosomes
B) DNA acetylation
C) RNA induced modification
of chromatin structure
D) repression of operons
E) induction
of operators in the promoter
A
132. A researcher found a method she could use to manipulate and
quantify phosphorylation and methylation in embryonic cells in
culture. In one set of experiments using this procedure in Drosophila,
she was readily successful in increasing phosphorylation of amino
acids adjacent to methylated amino acids in histone tails. Which of
the following results would she most likely see?
A) increased
chromatin condensation
B) decreased chromatin
concentration
C) abnormalities of mouse embryos
D) decreased
binding of transcription factors
E) inactivation of the selected genes
B
133. Which of the following describes the function of an enzyme known
as Dicer?
A) It degrades single-stranded DNA.
B) It degrades
single-stranded mRNA.
C) It degrades mRNA with no poly(A)
tail.
D) It trims small double-stranded RNAs into molecules that
can block translation.
E) It chops up single-stranded DNAs from
infecting viruses.
D
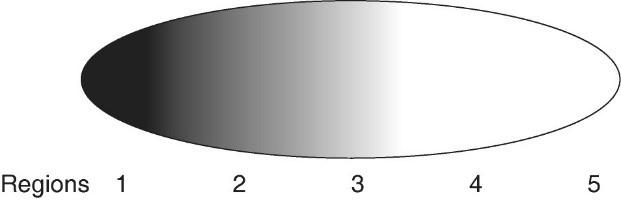
134. The researcher measures the concentration of the polypeptides from different regions in the early embryo and finds the following pattern (darker shading = greater concentration):
Which of the following would be his most logical assumption?
A)
The substance has moved quickly from region 5 to region 1.
B)
Some other material in the embryo is causing accumulation in region 1
due to differential binding.
C) The cytosol is in constant
movement, dispersing the polypeptide.
D) The substance is
produced in region 1 and diffuses toward region 5.
E) The
substance must have entered the embryo from the environment near
region 1.
D
135. In most cases, differentiation is controlled at which
level?
A) replication of the DNA
B) nucleosome
formation
C) transcription
D) translation
E)
post-translational activation of the proteins
C
136. Your brother has just purchased a new plastic model airplane. He
places all the parts on the table in approximately the positions in
which they will be located when the model is complete. His actions are
analogous to which process in development?
A)
morphogenesis
B) determination
C) induction
D)
differentiation
E) pattern formation
E
137. If a Drosophila female has a homozygous mutation for a maternal
effect gene,
A) she will not develop past the early embryonic
stage.
B) all of her offspring will show the mutant phenotype,
regardless of their genotype.
C) only her male offspring will
show the mutant phenotype.
D) her offspring will show the mutant
phenotype only if they are also homozygous for the mutation.
E)
only her female offspring will show the mutant phenotype.
B
138. Tumor suppressor genes
A) are frequently overexpressed in
cancerous cells.
B) are cancer-causing genes introduced into
cells by viruses.
C) can encode proteins that promote DNA repair
or cell-cell adhesion.
D) often encode proteins that stimulate
the cell cycle.
E) all of the above
C
139. The cancer-causing forms of the Ras protein are involved in
which of the following processes?
A) relaying a signal from a
growth factor receptor
B) DNA replication
C) DNA
repair
D) cell-cell adhesion
E) cell division
A
140. Viral genomes vary greatly in size and may include from four
genes to several hundred genes. Which of the following viral features
is most apt to correlate with the size of the genome?
A) size of
the viral capsomeres
B) RNA versus DNA genome
C) double
versus single strand genomes
D) size and shape of the
capsid
E) glycoproteins of the envelope
D
141. Most human-infecting viruses are maintained in the human
population only. However, a zoonosis is a disease that is transmitted
from other vertebrates to humans, at least sporadically, without
requiring viral mutation. Which of the following is the best example
of a zoonosis?
A) rabies
B) herpesvirus
C)
smallpox
D) HIV
E) hepatitis virus
A
142. In many ways, the regulation of the genes of a particular group
of viruses will be similar to the regulation of the host genes.
Therefore, which of the following would you expect of the genes of the
bacteriophage?
A) regulation via acetylation of histones
B)
positive control mechanisms rather than negative
C) control of
more than one gene in an operon
D) reliance on transcription
activators
E) utilization of eukaryotic polymerases
C
143. Why do RNA viruses appear to have higher rates of
mutation?
A) RNA nucleotides are more unstable than DNA
nucleotides.
B) Replication of their genomes does not involve the
proofreading steps of DNA replication.
C) RNA viruses replicate
faster.
D) RNA viruses can incorporate a variety of nonstandard
bases.
E) RNA viruses are more sensitive to mutagens.
B
144. Which of the following can be effective in preventing viral
infection in humans?
A) getting vaccinated
B) taking
nucleoside analogs that inhibit transcription
C) taking
antibiotics
D) applying antiseptics
E) taking vitamins
A
145. Antiviral drugs that have become useful are usually associated
with which of the following properties?
A) ability to remove all
viruses from the infected host
B) interference with the viral
reproduction
C) prevention of the host from becoming
infected
D) removal of viral proteins
E) removal of viral mRNAs
B