1) In the fractionation of homogenized cells using centrifugation,
the primary factor that determines whether a specific cellular
component ends up in the supernatant or the pellet is the___.
a)
Percentage of carbohydrates in the component
b) Presence or
absence of lipids in the component
c) Relative solubility of the
component
d) size and weight of the component
Answer: D
2) The advantage of light microscopy over electron microscopy is
that___.
a) Light microscopy allows one to view dynamic processes
in living cells
b) light microscopy provides for higher
magnification than electron microscopy
c) light microscopy
provides higher contrast than electron microscopy
d) light
microscopy provides for higher resolving power than electron microscopy
Answer: A
3) Which of the following macro-molecules leaves the nucleus of a
eukaryotic cell through pores in the nuclear membrane?
a)
DNA
b) phospholipids
c) glycogen
d) mRNA
Answer: D
4) Large number of ribosomes are present in cells that specialize in
producing which of the following molecules?
a) Nucleic
acids
b) proteins
c) glycogen
d) lipids
Answer: B
5) The Golgi apparatus has a polarity, or sidedness to its structure
and function. Which of the following statements correctly describes
this polarity?
a) Lipids in the membrane of the Golgi may be
sorted and modified as they move from one side of the Golgi to the
other
b) Transport vesicles fuse with one side of the Golgi and
leave from the opposite side
c) proteins in the membrane of the
Golgi may be sorted and modified as they move from one side of the
Golgi to the other
d) All of the listed responses correctly
describe polarity characteristics of the Golgi function.
Answer: D
6) Cyanide binds with at least one molecule involved in producing
ATP. If a cell is exposed to cyanide, most of the cyanide will be
found within the___.
a) Mitochondria
b) lysosomes
c)
peroxisomes
d) endoplasmic reticulum
Answer: A
7) The evolution of eukaryotic cells most likely involved__.
a)
Anaerobic archaea taking up residence inside a large bacterial host
cell to escape toxic oxygen-the anaerobic bacterium evolved into
chloroplasts
b) acquisition of an endomembrane system and
subsequent evolution of mitochondria from a portion of the
Golgi
c) endosymbiosis of an aerobic bacterium in a larger host
cell – the endosymbiont evolved into mitochondria
d) an
endosymbiotic fungal cell evolving into the nucleus
Answer: C
8) Researches tried to explain how vesicular transport in cells by
attempting to assemble the transport components. They set up
microtubular tracks along which vesicles could be transported, and
they added vesicles and ATP (because they knew the transport process
requires energy). Yet, when they put everything together there was no
movement or transport of vesicles. What were they missing?
a)
Endoplasmatic reticulum
b) motor proteins
c) contractile
microfilaments
d) an axon
Answer: D
9) The cell walls of bacteria, fungi, and plant cells an the
extracellular matrix cells are all external to the plasma membrane.
Which of the following is a characteristic common to all of these
extracellular structures?
a) They must block water and small
molecules to regulate the exchange of matter and energy with their
environment
b) they are composed of a mixture of lipids and
nucleotides
c) they must provide a rigid structure that maintains
an appropriate ratio of cell surface area to volume
Answer: B
10) Suppose a young boy is always tired and fatigued, suffering from
a metabolic disease. Which of the following organelles is most likely
involved in this disease?
a) Golgi apparatus
b)
mitochondria
c) ribosomes
d) lysosomes
Answer: B
11) Spherocytosis is a human blood disorder associated with a
defective cytokeletal protein in the blood cell (RBCs). What do you
suspect is the consequence of such a defect?
a) Abnormally shaped
RBCs
b) adherence of RBCs to blood vessel walls, causing plaque
formation
c) an insufficient supply of oxygen-transportation
proteins in the RBCs
d) an insufficient supply of ATP in the RBCs
Answer: A
12) H.V. Wilson worked with sponges to gain some insight into exactly
what was responsible for holding adjacent cells together. He exposed
two species of differently pigmented sponges to a chemical that
disrupted the cell-cell interaction (cell junctions), and the cells of
the sponges dissociated. Wilson then mixed the cells of the two
species and removed the chemical that caused the cells to dissociate.
Wilson found that the sponges reassembled into two separate species.
The cells from one species did not interact or form associations with
the cells of the other species. How do you explain the results of
Wilson's experiments?
a) One cell functioned as the nucleus for
each organism, thereby attracting only cells of the same
pigment
b) The molecules responsible for cell-cell adhesion (cell
junctions) were irreversibly destroyed during the experiment.
c)
The molecule responsible for cell-cell adhesion (cell junctions)
differed between the two species of sponge
d) The two species of
sponge had different enzymes that functioned in the reassembly process.
Answer: C
13) For a protein to be an integral membrane protein, it would have
to be __.
a) exposed on only one surface of the membrane
b)
hydrophilic
c) amphipathic, with at least one hydrophobic
region
d) hydrophobic
Answer: C
14) The membranes of winter wheat are able to remain fluid when it is
extremely cold by___.
a) Cotransport of glucose and hydrogen
b) decreasing the number of hydrophobic proteins in the
membrane
c) increasing the percentage of cholesterol molecules in
the membrane
d) increasing the percentage of unsaturated
phospholipids in the membrane.
Answer: D
15) Singer and Nicolson's fluid mosaic model of the membrane proposed
that membranes____.
a) consist of protein molecules embedded in a
fluid bilayer of phospholipids.
b) consist of a mosaic of
polysaccharides and proteins.
c) are a phospholipid bilayer
between two layers of hydrophilic proteins.
d) are a single layer
of phospholipids and proteins.
Answer: A
16) An animal cell lacking oligosaccharides on the external surface
of its plasma membrane would likely be impaired in which
function?
a) attaching the plasma to the cytoskeleton
b)
cell-cell recognition
c) establishing the diffusion barrier to
charged molecules
d) transporting ions against an electrochemical gradient
Answer: B
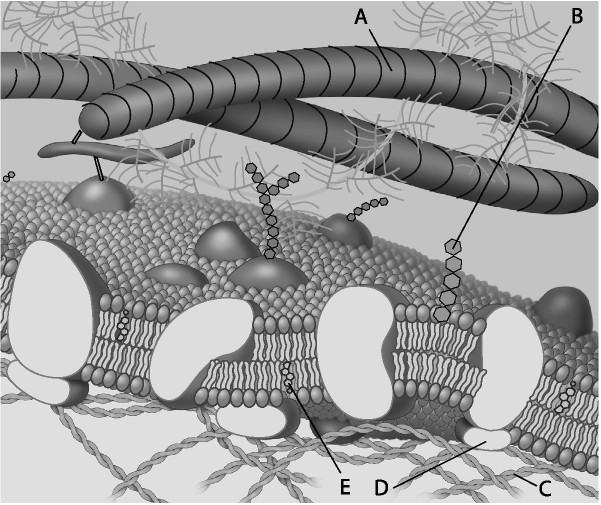
For the following question, match the labeled component of the cell
membrane in the figure with its description
17) Which
component is a peripheral protein?
a) A
b) B
c)
C
d) D
Answer: D
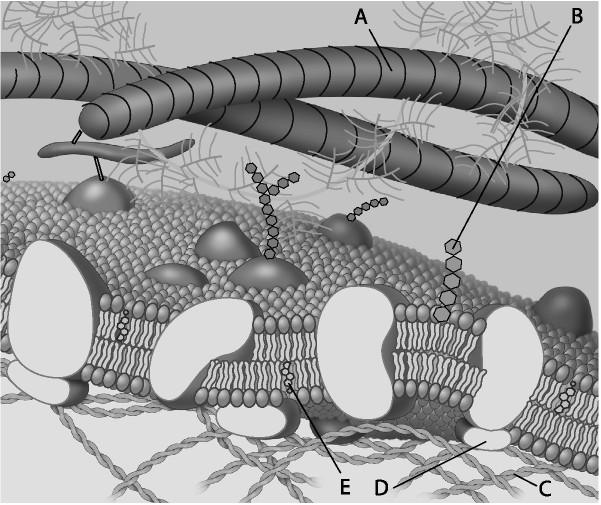
18) Which component is cholesterol?
a) A
b) B
c)
C
d) D
Answer: D
19)Which of the following allows water to move faster across cell
membranes?
a) Aquaporins
b) the sodium-potassium
pump
c) peripheral proteins
d) ATP
Answer: A
20)Which of the following would likely move through the lipid bilayer
of a plasma membrane most rapidly?
a) An amino acid
b)
glucose
c) K+
d) CO2
Answer: D
21) Which of the following processes includes all other?
a)
Passive transport
b) transport of an ion down its electrochemical
gradient
c) osmosis
d) facilitated diffusion
Answer: A
22) You are working on a team that is designating a new drug. For
this drug to work, it must enter the cytoplasm of specific target
cells. Which of the following would be a factor that determines
whether the molecule selectively enters the target cells?
a)
Lipid composition of the target cell's plasma membrane
b)
similarity of the drug molecule to other molecules transported by the
target cells
c) hydrophobicity of the drug molecule
d) lack
of charge on the drug molecule
Answer: B
23) Which of the following is true of osmosis?
a) Osmosis only
takes place in red blood cells
b) In osmosis, water moves across
a membrane from areas of lower solute concentration to areas of higher
solute concentration
c) In osmosis, solutes move across a
membrane from areas of lower water concentration to areas of higher
water concentration
d) Osmosis is an energy-demanding or “active” process
Answer: B
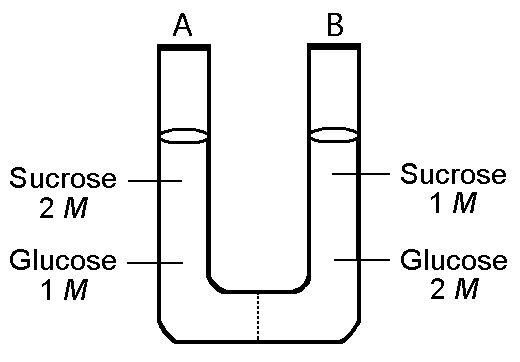
The solution in the two arms of this U-tube are separated by a membrane that is permeable to water and glucose but not to sucrose. Side A is half-filled with a solution of 2 M sucrose and 1 M glucose. Side B is half-filled with 1 M sucrose and 2 M glucose. Initially, the liquid levels on both sides are equal.
24) Refer to the figure. Initially, in terms of tonicity, the
solution in side A with respect to that in side B is
a)
saturated
b) hypertonic
c) hypotonic.
d) Isotonic
Answer: D
25) A patient was involved a serious accident and lost a large
quantity of blood. In an attempt to replenish body fluids, distilled
water - equal to the volume of blood lost – is added to the blood
directly via one of his veins. What will be the most probable result
of this transfusion?
a) The patient's red blood cells will burst
because the blood has become hypertonic compared to the cells.
b)
The patient's red blood cells will swell and possibly burst because
the blood has become hypotonic compared to the cells.
c) The
patient's red blood cells will shrivel up because the blood has become
hypotonic compared to the cells.
d) The patient's red blood cells
will shrivel up because the blood has become hypertonic compared to
the cells.
Answer: B
26) Most cells cannot harness heat to perform work because___
a)
heat is not a form of energy
b) heat can never be used to do
work
c) heat must remain constant during work
d) temperature
is usually uniform throughout a cell
Answer: D
27) Which of the following involves a decrease in entropy?
a)
Reactions that separate monomers
b) depolymerization
reactions
c) hydrolysis reactions
d) condensation reactions
Answer: C
28)Which of the following statements is a logical consequence of the
second law of thermodynamics?
a) Every chemical reaction must
increase the total entropy of the universe
b) energy can be
transferred or transformed, but it cannot be created o
destroyed
c) if the entropy of a system increases there must be a
corresponding decrease in the entropy of the universe
d) if there
is an increase in the energy of a system, there must be a
corresponding decrease in the energy of the rest of the universe
Answer: A
29) Which of the following types of reactions would decrease the
entropy within the cell?
a) Catabolic reactions
b)
digestion
c) anabolic reactions
d) hydrolysis
Answer: C
30) Biological evolution of life on Earth, from simple
prokaryote-like cells to large multicellular eukaryotic organisms
___.
a) has occurred in accordance with the laws of
thermodynamics.
b) has been made possible by expending Earth's
energy resources.
c) has occurred in accordance with the laws of
thermodynamics, by expending Earth's energy resources and causing an
increase in the entropy of the planet.
d) has caused an increase
in the entropy of the planet.
Answer: A
31) You have discovered an enzyme that can catalyze two different
chemical reactions. Which of the following is most likely to be
corrected?
a) Either the enzyme has two distinct active sites or
the reactants involved in the two reactions are very similar in size
and shape.
b) The enzyme is subject to competitive inhibition and
allosteric regulation.
c) The enzyme contains α-helices and
β-pleated sheets.
d) Two types of allosteric regulation occur:
The binding of one molecule activates the enzyme, while the binding of
a different molecule inhibits it.
Answer: A
32) A chemical reaction that has a positive G is best described
as____.
a) Spontaneous
b) exergonic
c)
endergonic
d) enthalpic
Answer: C
33) The active site of an enzyme is the region that____
a) is
inhibited by the presence of a co-enzyme or a co-factor
b) binds
noncompetitive inhibitors of the enzyme
c) is involved in the
catalytic reaction of the enzyme.
d) binds allosteric regulators
of the enzyme.
Answer: C
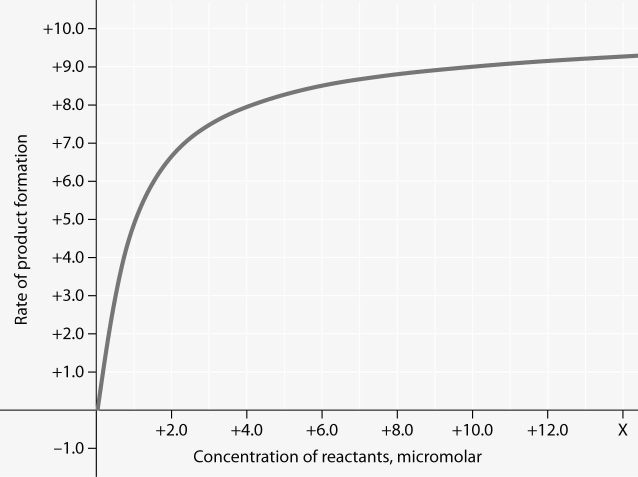
Rate of an enzyme-catalyzed reaction as a function of varying reactant concentration, with the concentration of enzyme constant
34) In the figure, why does the reaction rate plateau at higher
reactant concentrations?
a) The reaction nears equilibrium at
high reactant concentrations.
b) Feedback inhibition by product
occurs at high reactant concentrations.
c) Most enzyme molecules
are occupied by substrate at high reactant concentrations.
d)
The rate of the reverse reaction increases with reactant concentration.
Answer: C
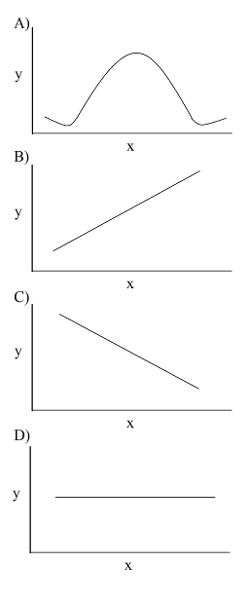
35) You collected data on the effect of pH on the function of the enzyme catalase in human cells. Which of the following graphs would you expect?
a) A
b) B
c) C
d) D
Answer: A
36) Substrate level phosphorylation occurs___
a) in
glycolysis
b) in the citric acid
c) during oxidative
phosphorylation
d) in both glycolysis and the citric acid
Answer: D
37) Which of the listed statements describes the results of the
following reaction?
C6H12O6 + 6 O2 → 6 CO2 + 6 H2O +
Energy
a) CO2 is reduced and O2 is oxidized.
b) C6H12O6 is
oxidized and O2 is reduced.
c) O2 is reduced and CO2 is
oxidized.
d) O2 is oxidized and H2O is reduced.
Answer: B
38) When a molecule of NAD+ (nicotinamide adenine dinucleotide) gains
a hydrogen atom (not a proton), the molecule becomes_____.
a)
Redoxed
b) dehydrogenated
c) oxidized
d) reduced
Answer: D
39) Which of the following statements about NAD+ is true?
a) Is
the absence of NAD+, glycolysis can still function
b) NAD+ has
more chemical energy than NADH
c) NAD+ is reduced to NADH during
glycolysis, pyruvate oxidation, and the citric acid cycle
d) NAD+
can donate electron for use in oxidative phosphorilation
Answer: C
40) Substrate-level phosphorylation accounts for approximately what
percentage of the ATP formed by the reactions of glycolysis?
a)
100%
b) 0%
c) 38%
d) 2%
Answer: A
41) In glycolysis, for each molecule of glucose oxidized to
pyruvate____
a) two molecules of ATP are used and six molecules
of ATP are produced.
b) two molecules of ATP are used and four
molecules of ATP are produced.
c) two molecules of ATP are used
and two molecules of ATP are produced.
d) four molecules of ATP
are used and two molecules of ATP are produced.
Answer: B
42) Carbon dioxide (CO2) is released during which of the following
stages of cellular respiration?
a) glycolysis and the oxidation
of pyruvate to acetyl CoA
b) oxidative phosphorylation and
fermentation
c) oxidation of pyruvate to acetyl CoA and the
citric acid cycle
d) fermentation and glycolysis
Answer: C
43) During aerobic respiration H2O is formed. Where does the oxygen
atom for the formation of the water come from?
a) pyruvate
(C₃H₃O₃-)
b) glucose (C₆H₁₂O₆)
c) molecular oxygen
(O₂)
d) carbon dioxide (CO₂)
Answer: C
44) The primary role of oxygen in cellular respiration is to
____
a) combine with lactate forming pyruvate
b) combine
with carbon, forming CO2
c) yield energy in the form of ATP as it
is passed down the respiratory chain
d) act as an acceptor for
electron and hydrogen, forming water
Answer: D
45) Where are the proteins of the electron transport chain
located?
a) mitochondrial inner membrane
b) mitochondrial
matrix
c) mitochondrial outer membrane
d) mitochondrial
intermembrane space
Answer: A
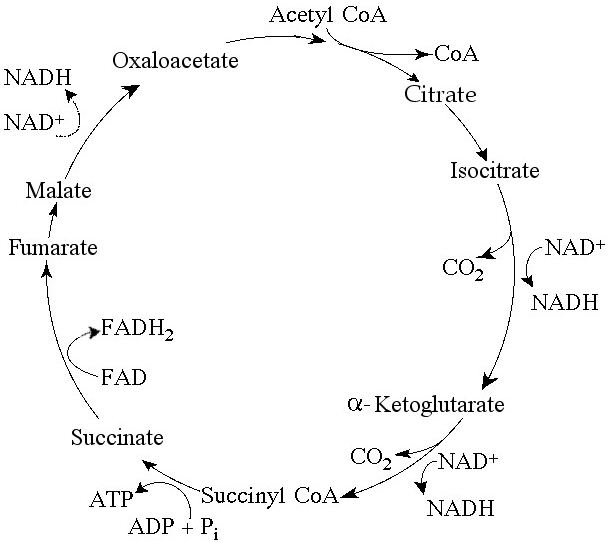
Use the following figure to answer the questions below.
46) Starting with citrate, which of the following combinations of
products would result from three acetyl CoA molecules entering the
citric acid cycle ( see the accompanying figure)?
a) 3 ATP, 6
CO₂, 9 NADH, and 3 FADH₂
b) 1 ATP, 2 CO₂, 3 NADH, and 1 FADH₂
c) 38 ATP, 6 CO₂, 3 NADH, and 12 FADH₂
d) 3 ATP, 3 CO₂, 3
NADH, and 3 FADH₂
Answer: A
47) Which of the following events tales place in the electron
transport chain?
a) substrate-level phosphorylation
b)
breakdown of glucose into two pyruvate molecules.
c) the
breakdown of an acetyl group to carbon dioxide
d) the extraction
of energy from high-energy electrons remaining from glycolysis and the
citric acid cycle
Answer: D
48) You have a friend who lot 7 kg (about 15 pounds) of fat on a
regimen of strict diet and exercise. How did the fat leave his
body?
a) It was converted to ATP, which weighs much less than
fat.
b) It was converted to urine and eliminated from the
body.
c) It was converted to heat and then released.
d) It
was released as CO₂ and H₂O.
Answer: D
49) Chemiosmotic ATP synthesis (oxidative phosphorylation) occurs in
____
a) only in mitochondria, using either oxygen or other
electron acceptors.
b) all cells, but only in the presence of
oxygen.
c) all respiring cells, both prokaryotic and eukaryotic,
using either oxygen or other electron acceptors.
d) only
eukaryotic cells, in the presence of oxygen.
Answer: C
50) Which of the following normally occurs regardless of whether or
not oxygen (O2) is present?
a) Glycolysis
b) oxidative
phosphorylation (chemiosmosis)
c) citric acid cycle
d) fermentation
Answer: A
51) What part of the cell is essential to aerobic
respiration?
a) Ribosomes
b) mitochondrion
c)
endoplasmatic reticulum
d) nucleus
Answer: B