Antibodies
- globulin proteins
- term immunoglobulins(ig) used for antibodies
- made in response to an antigen, in which they can recognize and bind to
- antigen-binding sites at least two identical sites that binds to epitopes
- basis of humoral immune response
What are B cells?
- Type of lymphocyte that develops & matures in bone marrow.
- circulate in blood and lymph system
- activated when their receptors recognize and respond to antigens by making specific antibodies
- can become memory cells
How are B cells activated?What happens when they are activated?
- Process begins when B cells are exposed to free, or extracellular antigens
- activated when a B-cell's immnuoglobulins (located on its surface) binds to the epitope for which they become specific
- when activated B-cells under go proliferation
- Usually needs assistance from T Helper cells
Define: Major histocompatibility complex (MHC)
- A collection of genes that is made up of glycoproteins that are found on the plasma membrane on cells that help the immune system recognize self from nonself; preventing the immune system from making antibodies that would be harmful to the host.
MHC of class II
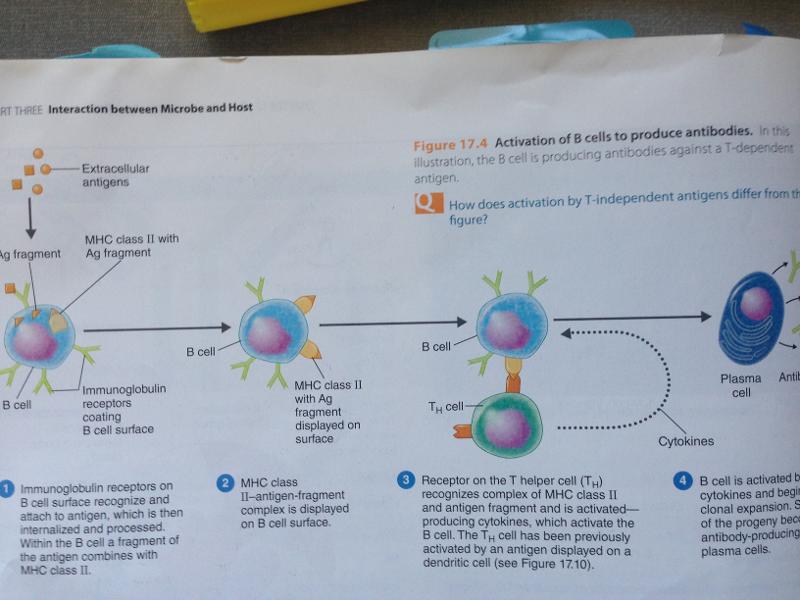
- only found on antigen-presenting cells (ie. macrophages, b-cells, dendrites)
- When an antigen activates a B-cells, fragments of the antigen and the MHC class to are presented on the surface of the cell for the receptors of the T helper cells to identify.
Define: plasma cell
- an activated B cell proliferates into a large clone of cells some of which differentiate in to antibody producing plasma cells
- a mature B lymphocyte that is specialized for antibody (immunoglobulin) production
Define: memory cell
- clones of activate B cells (that do not become plasma cells) and Helper T cells that exist as long-lived but non proliferating memory cells responsible for the more rapid secondary response when the antigen is encountered on a subsequent occasion.
- vaccinations are used to initiate the production of memory cells.
define clonal selection
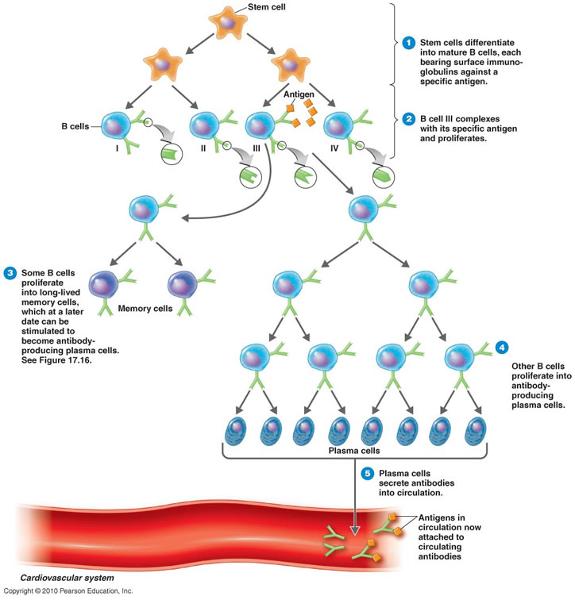
- the development of clones of B and T cells against a specific antigen
antigen-antibody binding
- binding of an antibody to an antigen protects the host by tagging foreign cells and molecules for destruction by phagocytes and complement.
- antibody binds to antigens epitope
agglutination
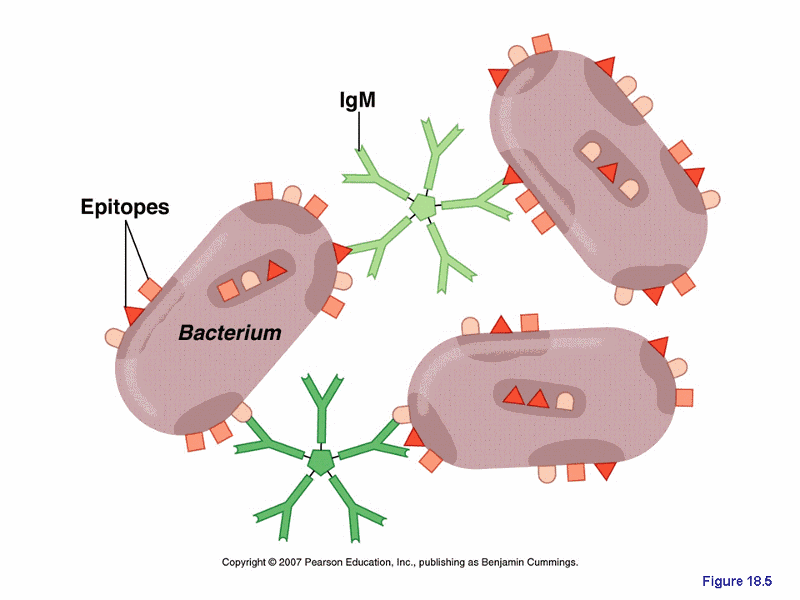
- one of the possible outcomes of antigen-antibody binding
- antibodies cause antigens to clump together
- the two antigen binding sites on an Ig combine with epitopes on two different foreign cells, making them clump with makes them easily ingested by phagocytes
- reduces the number of infection units to be dealt with
Opsonization
- one of the possible outcomes of antigen-antibody binding
- the antigen is coated with antibodies that enhance the ingestion and lysis by phagocytic cells
antibody-dependent cell-mediated cytotoxicity
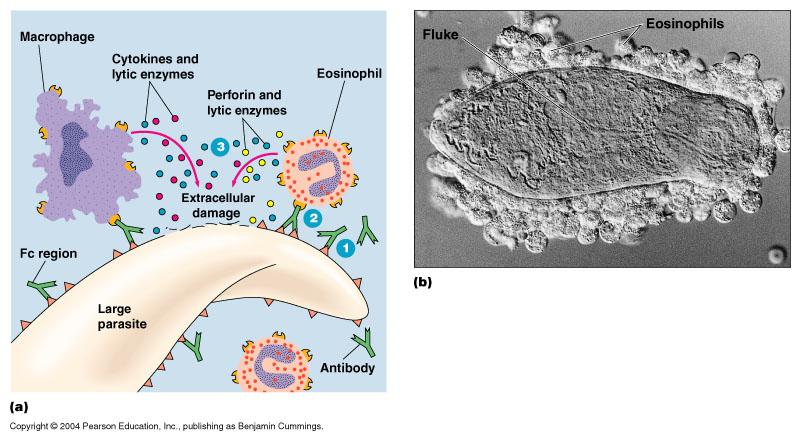
- one of the possible outcomes of antigen-antibody binding
- the pathogen is first coated with antibodies then a variety of cells of the immune system bind to the Fc regions of the on the antibodies & thus the pathogen, the attacking cells them secrete substances such as perforin and lytic enzymes that cause the pathogen to be lysed
neutralization
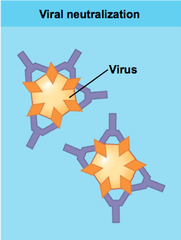
- possible out come of antigen-antibody binding
- IgG antibodies inactivate microbes by blocking their attachment to host cells; neutralize toxins in a similar way
activation of the complement system
- one of the possible outcomes of and antigen-antibody binding
- the attachment of microbes of an antibody-complement complex
- causes inflammation and cell lysis
- attracts phagocytes and other defensive immune system cells to the area
T cells
- lymphocytes develop from stem cells in bone marrow; reach maturity in the thymus where they go through a process called thymic selection in which the T cells that will not specifically recognize self-molecules of MHC are weeded out---> important in preventing the body from attacking its own tissues
- respond to the need to combat intracellular pathogens
- also the way in which the immune system recognizes cells that are nonself
- to recognize an antigen they require antigen fragments be presented by antigen presenting cells.
- do not have immunoglobulins on surface but rather TCRs ( T cell receptor)
T Helper Cells (CD4+ T Cells)
- TH1 cells activate cells involved with cellular immunity: macrophages, Tc cells, Natural killer cells
- TH2 cells are associated with allergic reactions and parasitic infections; stimulate eosinphils, IgM, & IgE
- Th17 cells activate innate immunity and responses to extracellular bacteria; situated in the skin and in the lining of the gastrointestinal tract
- T helper cells, or CD4+ T cells, are activated by MHC class II on APCs. After binding on an APC, CD4+ T cells secrete cytokines that activate other T cells and B cells
T regulatory cells (Treg)
- main function is to combat autoimmunity by suppressing T cells that might act on the body's self
T Cytotoxic Cells (CD8+ T cells)
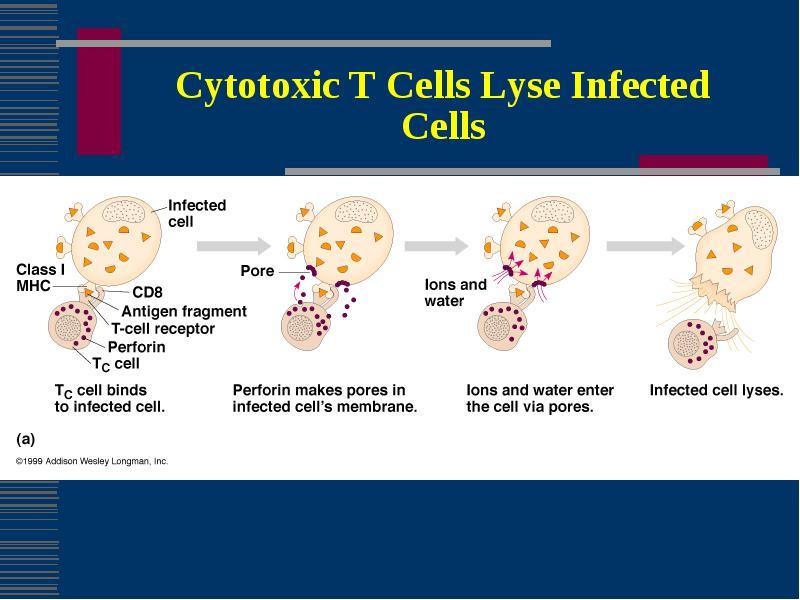
- T cytotoxic cells are activated by endogenous antigens and MHC class I on a target cells and are transformed into CTLs (Cytotoxic T Lymphocyte)
- CTLs attache to the target cells and releases perforin then granzymes resulting in Apoptosis (programmed cell death)
- Primarlily the target cells are self cells that have been altered by infection with a pathogen especially viruses
Define: Antigen
- a substance that causes the body to produce antibodies
- include toxins, bacteria, foreign blood cells, and the cells of transplanted organs
- either proteins or large polysaccharides (nucleic acids and lipids only pathogenic when combined with proteins and polysaccharides
monomer antibody structure
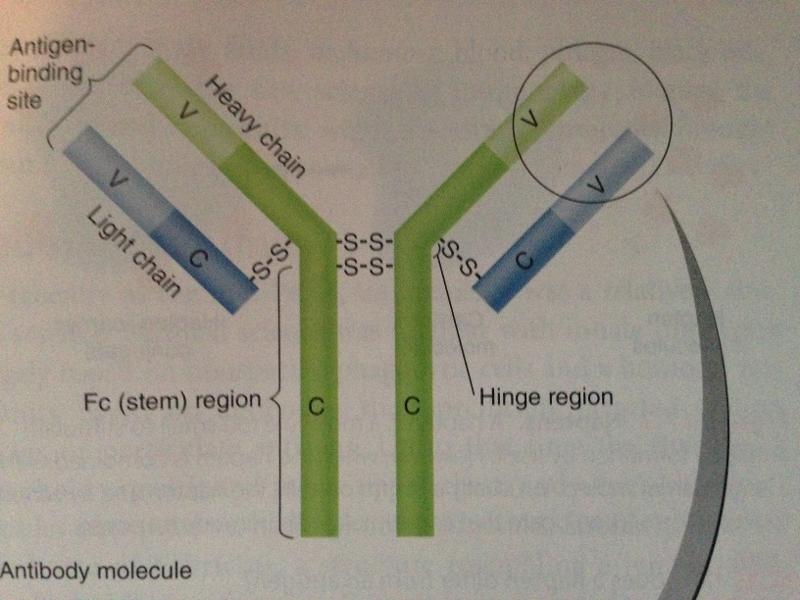
- two antigen-binding sites that form a Y's arms called variable (V) regions
- the stem of the antibody monomer is called the constant (C) regions; the same for a particular class of immunoglobulin; there are 5 major C class
- The stem of the of the "Y" shaped antibody is called the Fc region
- If left exposed after both antigen-binding sites attach to antigen, the Fc regions of adjacent adjacent antibodies can bind complement---> destruction of antigen; Fc region may also bind to a cell leaving antigen binding sites free to react with antigens
Define: epitope or antigenic determinant
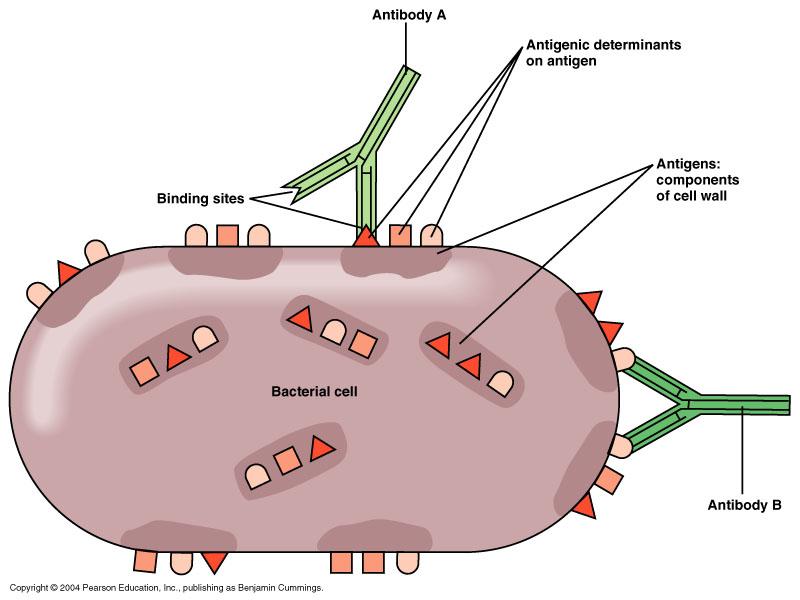
- specific regions on an antigen that antibodies recognize, respond and can bind to
- nature of this interaction depends on the size, shape, and chemical structures of the binding site on the antibody molecule
- antigens carries more than one epitope
IgG
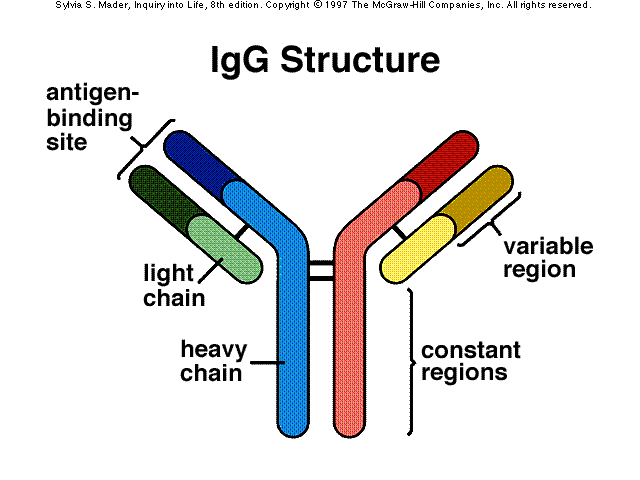
- structure: monomer
- 80% of all antibodies in serum
- in regions of inflammation; readily crossed blood vessels into tissue
- Location: blood, lymph and intestine
- Known functions: when bound to pathogens enhances phagocytosis, neutralizes toxins and viruses, trigger the complement system, protect against circulating bacteria and viruses
IgM
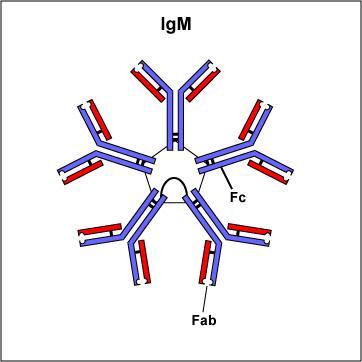
- stucture: pentamer (remember M for macro= their large size)
- up to 5-10% of antibodies
- located in blood, lymph, B cell surface(as monomer); large size prevents IgMs from moving about freely
- known functions: especially effective against micro organisms and agglutinating antigens; predominant antibody involved in the response to the ABO blood group antigens on the surface of RBCs; first antibodies produces in response to initial infection
- short lived (5days) which makes it useful in diagnosing if there are high concentrations of IgM against a pathogen, its likely that the disease observed is caused by that pathogen
IgA
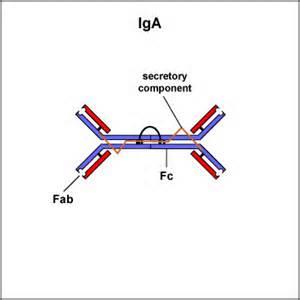
- structure: dimer( with secretory component)
- up to 10-15% in serum, but most common antibodies in mucous membranes and in body secretions with this is consideration, it is the most abundant Ig in the body
- locations: secretions(tears, saliva, mucus, intestine, milk) blood, lymph
- Functions: prevent attachment of microbial pathogens to mucosal surfaces
IgD
- structure monmer
- make up only about 0.2% of antibodies in serum
- location: B cell surface, blood, lymph
- Functions: not know; presence on B cell functions in initiation of immune response
IgE
- 0.002% of antibodies in serum
- location: bound to mast and basophil cells throughout body; blood
- Functions: aides in allergic reactions- when antigen like pollen touches the IgE antibodies that are attached to cell, that cell releases histamine; possibly lysis of parasitic worms
antigen-presenting Cells (APCs)
- include B cells, dendritic cells, and machrophages
- dendritic cells are the primary APCs
- activated macrophages are effective phagocytes and APCs
- APCs carry antigens to lymphoid tissues where T cells recognize the antigen are located
- generally present fragments of antigen to T cell
dendritic cells
- found in the skin, lymph nodes, spleen, various body tissues; are principle antigen presenting cells; engulf and breakdown microbes and display pieces like flags to T cells
review: cytokines
- chemical messengers produced by all cells of the immune system in response to a stimulus
- protein or glycoprotein
- acts only on a cell that it has a receptor for
review interferons
- family of cytokines
- protect cell from viral infection
review: tumor necrosis factor (TNF-alpha)
- important family of cytokine
- strong factor in inflammatory reactions of autoimmune diseases
Active vs passive immunity
- Immunity is acquired actively when a person is exposed to microorganisms or foreign substances and the immune system responds
- immunity is acquired passively when antibodies are transferred from one person to another
Naturally acquired active immunity
- person is exposed to antigens, becomes ill and then recovers
- antigens enter the body naturally; body induces antibodies and specialized lymphocytes
Naturally acquired passive immunity
- the natrual transfer of antibodies from mother to fetus via placenta to infant via the mother's milk
artificially acquired active immunity
- antigens are introduced in vaccines; body produces antibodies and specialized lymphocytes
artificially acquired passive immunity
- involves the injection of antibodies rather than antigens into the body. these antibodies comes from an animal or person who already is already immune to the disease
cell-mediated immunity
- an immune response that does not involve antibodies, but rather involves the activation of phagocytes, antigen-specific cytotoxic T-lymphocytes, and the release of various cytokines in response to an antigen
cytokine storm
- the harmful overproduction of cytokines due to cytokines stimulating cells to produce more cytokines
- can do significant damage to tissues
attenuated vaccines
- consist of live attenuated (weakened) microorganisms; attenuated virus vanccines generally provide life long immunity
- closely mimic an actual infection
inactivated vaccines
- consist of killed bacteria or viruses
- examples: rabies, influenza
subunit vaccines
- consist of antigenic fragments of a microorganism; this include recombinant vaccines and toxoids; typical require boosters
- ex: recombinant vaccine: hep B vaccine; toxoid: tetanus and diphtheria
Conjugated vaccines
- combine the desired antigen with a protein that boosts the immune response
- developed more recently to deal with the poor immune response of children to vaccines based on capsular polysaccharides
- ex: Haemophilus influenzae type b (Hib)
Nucleic Acid (DNA) Vaccines
- cause recipient by the injection of plasmids of naked DNA into muscle resulting in the production of the protein antigen encoded in the DNA
- protein antigents are carried to the red bone marrow and stimulate both humoral and cellular immunity
serology
- the science that studies serum and immune responses that are evident in serum
antiserum
- solution of antibodies used in the identification of medically important microorganisms; if an unknown bacterium is isolated from a patient it can be tested against known antisera and identified quickly
slide agglutination
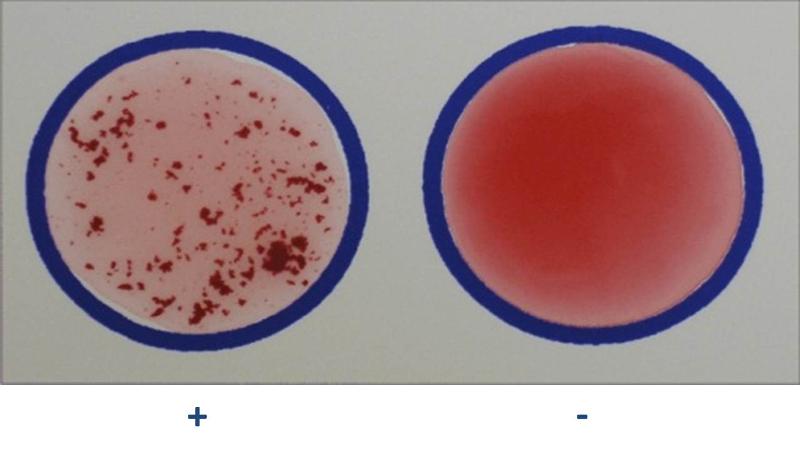
- procedure in which samples of an unknown bacterium are placed in a drop of saline on several slides; then a different known antiserum is added to each sample
- the bacteria agglutinate (clump) when mixed with antibodies produced in response to that species or strain of bacterium.
- positive test is indicated by the presence of agglutination
define: agglutination reactions
- the interaction of particulate antigens(cells that carry antigens) with antibodies leads to these reaction
direct agglutination tests
- detects antibodies against relatively large cellular antigens, such as those on RBCs, bacteria, and fungi
titer
- concentration of serum antibodies
- often done to detect immunity to a disease
indirect (passive) agglutination test:

- antibodies cause visible agglutination of soluble antigens affixed to latex sphere
- latex aggluination test (common)
Hemoagglutination
- when aggluntination reactions involve the clumping of RBCs; routinely done in blood typing
enzyme-linked immunosorbent assay (ELISA)
- most widely used group of test: enzyme immunoassay (EIA)
- antigen-antibody reactions are detected by enzyme activity; if the indicator enzyme is present in the test well, an antigen antibody reaction has occurred
direct ELISA
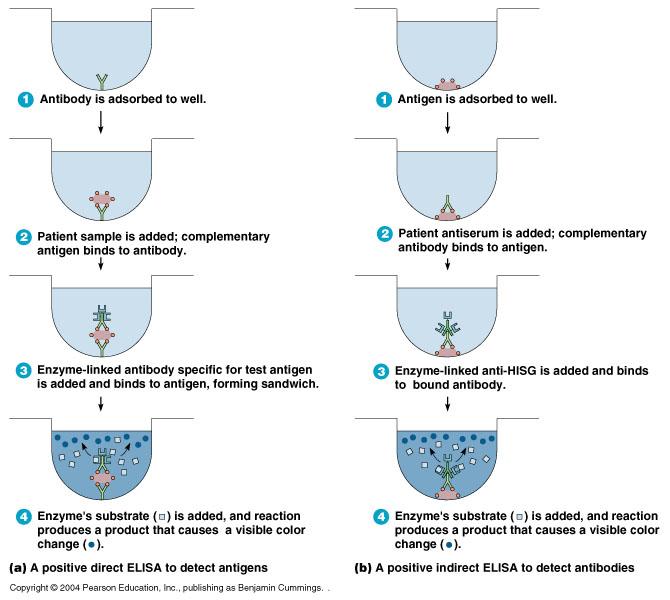
detects antigens
- antibody is absorbed to well
- patient sample is added; complementary antigen binds to antibody
- enzyme-linked antibody specific for test is added & bidned to antigen, forming a sandwich
- enzyme's substrate is added, and reaction produces a product that causes a visible color change
indirect ELISA
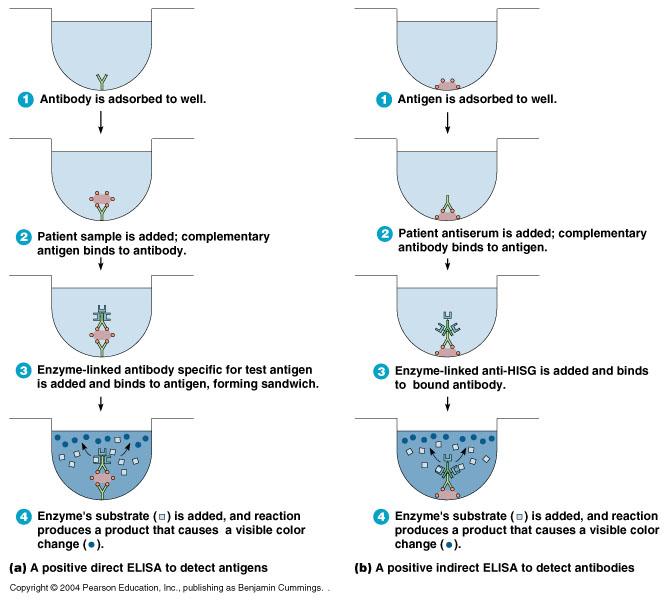
detects antibodies
- antigen is absorbed to well
- patient serum is added; complementary antibody binds to antigen
- Enzyme-linked anti-HISG (anti-human serum globulin) is added and binds to bound antibody
- Enzymes substrate is added, and reaction produces a product that causes a visible color change
fluorescent antibody (FA) techniques
- combines fluorescent dyes with antibodies to make them fluoresce when exposed to ultraviolet light
- can identify microorganisms in clinical specimens and detect the presence of specific antibody in serum
- procedures are quick, sensitive and very specific
direct FA tests
- usually used to identify a microorganism in a clinical specimen
- the specimen containing the antigen to be identified is fixed onto a slide; fluorescein-labeled antibodies are then added and slide is incubated; slide is washed to removed any antibody not bound to antigen and then examined under fluorescence microscope for yellow-green fluorescence; the residual antibody will be visible even if the antigen is submicroscopic size.
indirect FA tests
- used to detect the presence of a specific antibody in serum following exposure to microorganism
anti-human immune serum globulin (anti-HISG)
- an antibody that that reacts specifically with human antibodies
Western blotting (immunoblotting)
- can be used to identify a specific protein in a mixture
- serum antibodies separated by electrophoresis are identified with an enzyme-linked antibody
monoclonal antibodies
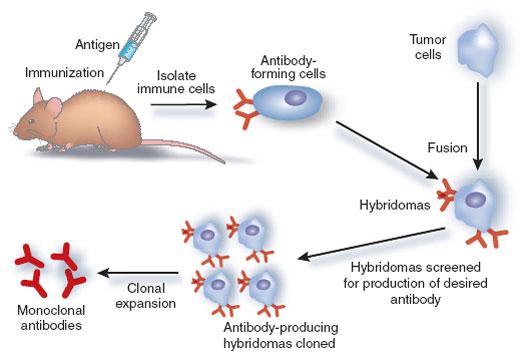
- hybridoma: the breakthrough of fusing an "immortal" cancerous B cell, myelomas, with an antibody-producing normal B cell.
- monoclonal antibodies: antibodies produces from a single hybridoma clone done in a cultured environment
- usefulness: they are uniform, they are highly specific, and they can be produced readily in large qualities
southern blotting
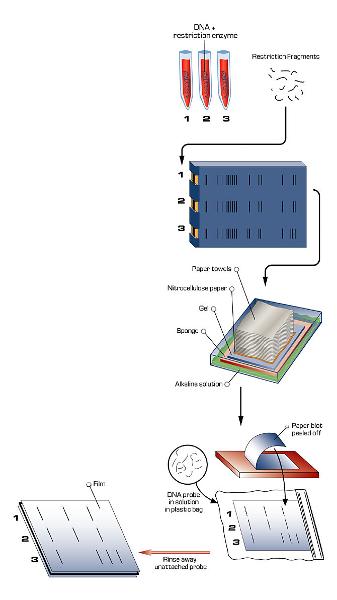
- a technique that uses DNA probes to detect the presence of specific DNA in restriction fragment separated by electrophoresis
- any person's DNA can be tested for the presence of the mutated gene
- DNA containing the gene of interest is extracted from human cells and cut into fragments by restriction enzymes
- the fragments are separated according to size by electrophoresis. Each band consists consist of many copies of particular DNA fragment. The bands are invisible but can be made visible by staining
- The DNA are transferred to a nitrocellulose filter by blotting. The solution passes through the gel and filters the paper towels by capillary action
- The produces a nitrocellulose filter with DNA fragments positioned exactly as on the gel
- The filter is exposed to a labeled probe for a specific gene. The probe will base-pair (hybridize) with a short sequence present on the gene
- The fragment containing the gene of interest is identified band on the filter
DNA probe
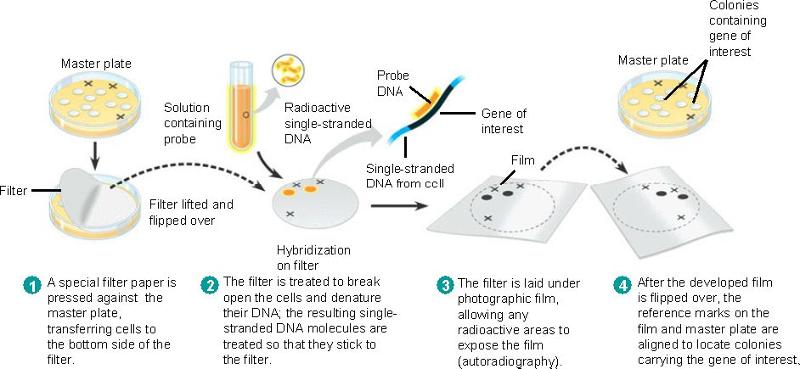
- DNA probe: short segments of single-stranded DNA that complementary to the desired gene are synthesized
How do microbiologists determine that a microbe causes a particular disease?
Koch's Postulates
- The same pathogen must be present in every cases of the disease
- The pathogen must be isolated from the diseased host and grown in a pure culture
- the pathogen from the pure culture must cause the disease when it is inoculated into a healthy, susceptible laboratory animal
- the pathogen must be isolated from the inoculated animal and must be shown to be the original microorganism
Exceptions to Koch's postulates
- some organism's can not be grown on an artificial media
- some diseases (tetanus) have unequivocal signs and symptoms
- some diseases (pneumonia and nephritis) may be caused by a variety of organisms
- some pathogens such as S. pyogenes cause several different diseases
- Certain pathogens causes diseases in humans only (ex. HIV)
Pros & cons of recombinant DNA technology & examples
- Pros- medically we can synthesize human insulin for diabetes patients etc. agriculturally we can engineer plants to resist bugs, or extreme weather.
- Cons- health concerns like recombinant DNA has resulted in the inadvertent production of toxic substances in genetically engineered bacteria, yeast, plants and animals it could similarly cause these problems in humans. Also ethical concerns arise if human DNA is inserted into tomato crops to hasten growth is eating these tomatoes, cannibalism. Similar ethical debates can be found on biotechnology.