What cations are responsible for water hardness?
The cations responsible for water hardness are Ca2+ and Mg2+.
Experimental Procedure, Part A.1. Calculate the mass of disodium ethylenediaminetetraacetate (molar mass = 372.24 g/mol) required to prepare 250 mL of 0.010 M solution. Show the calculation. Express the mass to the correct number of significant figures.
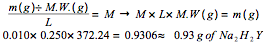
Experimental Procedure, Part A.3. A 25.7-mL volume of prepared Na2H2Y solution titrates 25.0 mL of standard 0.0107 M Ca2+ solution to Eriochrome Black T endpoint. What is the molar concentration of the Na2H2Y solution?
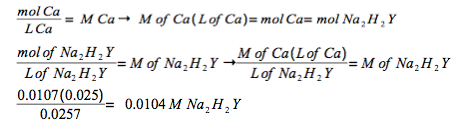
Which hardening ion Ca2+ or Mg2+ , binds more tightly to (forms a stronger complex ion with) the Eriochrome Black T indicator used for today's analysis?
Periodic trends show that as you move down the rows the lattice energy decrease. Therefore, Mg should have a higher lattice energy then Ca and because of this Mg will form a stronger complex ion with the Black T indicator.
What is the color change at the endpoint?
Sky Blue.
Calculate the moles of hardening ions in the water sample.
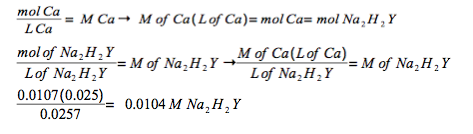
Assuming the hardness is due exclusively to CaCO3, express the hardness in mg CaCO3/L sample.
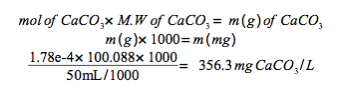
What is the hardness concentration expressed in ppm CaCO3
1 mg CaCO3/L = 1 ppm
356.3 ppm of CaCO3
Classify the hardness of this water according to Table 9.1.
Very hard water
Determine the number of moles of hardening ions present in a 100-mL volume sample that has a hardness of 58 ppm CaCO3.
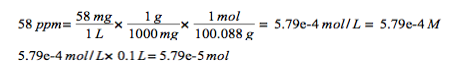
Determine the number of moles of hardening ions present in a 100-mL volume sample that has a hardness of 58 ppm CaCO3.
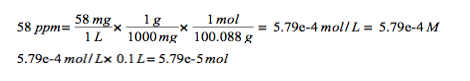
What volume of the 0.100 M Na2H2Y is need to reach the Eriochorme Black T endpoint for the analysis of the solution.
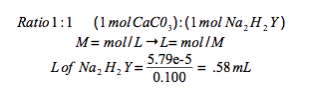
Water hardness is also commonly expressed in units of grains/gallons, where 1 grain/gallon equals 17.1 ppm CaCO3. Express the hardness of this "slightly hard" water sample in grains/gallon.
58 ppm/ 17.1 ppm = 3.39 gpg