1) About 25 of the 92 natural elements are known to be essential to life. Which four of these 25 elements make up approximately 96% of living matter?
A) carbon, sodium, hydrogen, nitrogen
B) carbon, oxygen, phosphorus, hydrogen
C) oxygen, hydrogen, calcium, nitrogen
D) carbon, hydrogen, nitrogen, oxygen
E) carbon, oxygen, nitrogen, calcium
D
Why is each element unique and different from other elements in chemical properties?
A) Each element has a unique atomic mass.
B) Each element has a unique atomic weight.
C) Each element has a unique number of protons in its nucleus.
D) Each element has a unique number of neutrons in its nucleus.
E) Each element has different radioactive properties.
C
Knowing just the atomic mass of an element allows inferences about which of the following?
A) the chemical properties of the element
B) the number of protons in the element
C) the number of neutrons in the element
D) the number of protons plus neutrons in the element
E) both the number of protons and the chemical properties of the element
D
If a salamander relied on hydrogen bonds to cling to surfaces, what type of surface would cause the most problems for this animal?
A) a surface coated with a thin film of water
B) a surface made with carbon and hydrogen atoms covalently bonded together
C) a surface made with carbon, hydrogen, and oxygen atoms covalently bonded together
D) a surface made with carbon, hydrogen, nitrogen, and oxygen atoms covalently bonded together
E) a surface made with silicon and oxygen atoms covalently bonded together
B
A covalent chemical bond is one in which
A) electrons are removed from one atom and transferred to another atom so that the two atoms become oppositely charged.
B) protons and neutrons are shared by two atoms so as to satisfy the requirements of both atoms.
C) outer-shell electrons of two atoms are shared so as to satisfactorily fill the outer electron shells of both atoms.
D) outer-shell electrons of one atom are transferred to fill the inner electron shell of another atom.
E) an electron occupies a hybrid orbital located between the nuclei of two atoms.
C
Nitrogen (N) is much more electronegative than hydrogen (H). Which of the following statements is correct about the atoms in ammonia (NH3)?
A) Each hydrogen atom has a partial positive charge; the nitrogen atom has a partial negative charge.
B) The nitrogen atom has a strong positive charge; each hydrogen atom has a strong positive charge.
C) Each hydrogen atom has a slight negative charge; the nitrogen atom has a strong positive charge.
D) The nitrogen atom has a slight positive charge; each hydrogen atom has a slight negative charge.
E) There are covalent bonds between the hydrogen atoms and polar bonds between each hydrogen atom and the nitrogen atom.
A
7) When two atoms are equally electronegative, they will interact to form
A) hydrogen bonds.
B) van der Waals interactions.
C) polar covalent bonds.
D) nonpolar covalent bonds.
E) ionic bonds.
D
Which of the following explains most specifically the attraction of water molecules to one another?
A) nonpolar covalent bond
B) polar covalent bond
C) ionic bond
D) hydrogen bond
E) hydrophobic interaction
D
Geckos are lizards with “sticky” digits that result from:
A) a slimy substance secreted by glands on the digits
B) hydrogen bonds
C) Van der Waals forces
D) microscopic reaction cups
E) biological magnetic forces
C
What is the maximum number of hydrogen atoms that can be covalently bonded in a molecule containing two carbon atoms?
A) 2
B) 3
C) 4
D) 6
E) 8
D
Which of the following would be regarded as compounds?
A) H2O, O2, and CH4
B) H2O and O2
C) O2 and CH4
D) CH4 and O2, but not H2O
E) H2O and CH4, but not O2
E
The partial negative charge in a molecule of water occurs because
A) the oxygen atom acquires an additional electron.
B) the electrons shared between the oxygen and hydrogen atoms spend more time around the oxygen atom nucleus than around the hydrogen atom nucleus.
C) the oxygen atom has two pairs of electrons in its valence shell that are not neutralized by hydrogen atoms.
D) the oxygen atom forms hybrid orbitals that distribute electrons unequally around the oxygen nucleus.
E) one of the hydrogen atoms donates an electron to the oxygen atom.
B
Which of the following takes place as an ice cube cools a drink?
A) Molecular collisions in the drink increase.
B) Kinetic energy in the drink decreases.
C) A calorie of heat energy is transferred from the ice to the water of the drink.
D) The specific heat of the water in the drink decreases.
E) Evaporation of the water in the drink increases.
B
Liquid water's high specific heat is mainly a consequence of the
A) small size of the water molecules.
B) high specific heat of oxygen and hydrogen atoms.
C) absorption and release of heat when hydrogen bonds break and form.
D) fact that water is a poor heat conductor.
E) higher density of liquid water than solid water (ice).
C
Why does evaporation of water from a surface cause cooling of the surface?
A) The breaking of bonds between water molecules absorbs heat.
B) The water molecules with the most heat energy evaporate more readily.
C) The solute molecules left behind absorb heat.
D) Water molecules absorb heat from the surface in order to acquire enough energy to evaporate.
E) The expansion of water vapor extracts heat from the surface.
B
Why does ice float in liquid water?
A) The high surface tension of liquid water keeps the ice on top.
B) The ionic bonds between the molecules in ice prevent the ice from sinking.
C) Ice always has air bubbles that keep it afloat.
D) Hydrogen bonds stabilize and keep the molecules of ice farther apart than the water molecules of liquid water.
E) The crystalline lattice of ice causes it to be denser than liquid water.
D
Hydrophobic substances such as vegetable oil are
A) nonpolar substances that repel water molecules.
B) nonpolar substances that have an attraction for water molecules.
C) polar substances that repel water molecules.
D) polar substances that have an affinity for water.
E) charged molecules that hydrogen-bond with water molecules.
A
Which of the following solutions would require the greatest amount of base to be added to bring the solution to neutral pH?
A) gastric juice at pH 2
B) vinegar at pH 3
C) tomato juice at pH 4
D) black coffee at pH 5
E) household bleach at pH 12
A
Carbon dioxide produced from burning fossil fuels dissolves in sea water forming:
A) calcium carbonate
B) organic compounds
C) phospholipids
D) curbllipids
E) carbonic acid
E
The element present in all organic molecules is
A) hydrogen.
B) oxygen.
C) carbon.
D) nitrogen.
E) phosphorus.
C
Stanley Miller proved that organic molecules can be synthesized in the lab by recreating early earth conditions, most notable he was able to synthesize what?
Amino Acids
How many electron pairs does carbon share in order to complete its valence shell?
A) 1
B) 2
C) 3
D) 4
E) 8
D
Why are hydrocarbons insoluble in water?
A) The majority of their bonds are polar covalent carbon-to-hydrogen linkages.
B) The majority of their bonds are nonpolar covalent carbon-to-hydrogen linkages.
C) They are hydrophilic.
D) They exhibit considerable molecular complexity and diversity.
E) They are lighter than water.
B
Research indicates that ibuprofen, a drug used to relieve inflammation and pain, is a mixture of two enantiomers; that is, molecules that
A) have identical chemical formulas but differ in the branching of their carbon skeletons.
B) are mirror images of one another.
C) exist in either linear chain or ring forms.
D) differ in the location of their double bonds.
E) differ in the arrangement of atoms around their double bonds
B
A compound contains hydroxyl groups as its predominant functional group. Which of the following statements is true concerning this compound?
A) It lacks an asymmetric carbon, and it is probably a fat or lipid.
B) It should dissolve in water.
C) It should dissolve in a nonpolar solvent.
D) It won't form hydrogen bonds with water.
E) It is hydrophobic.
B
Which two functional groups are always found in amino acids?
A) ketone and methyl
B) carbonyl and amino
C) carboxyl and amino
D) amino and sulfhydryl
E) hydroxyl and carboxyl
C
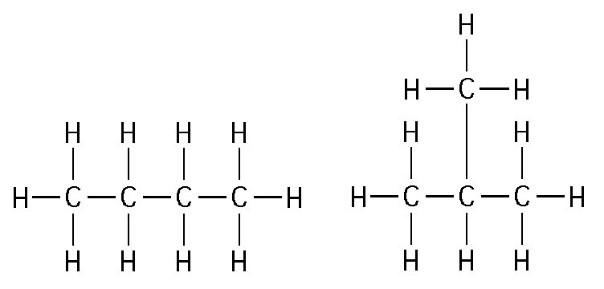
27) The two molecules shown in the figure above are best described as
A) optical isomers.
B) enantiomers.
C) structural isomers.
D) cis-trans isomers.
E) chain length isomers.
C
Which of these classes of biological molecules consist of both small molecules and macromolecular polymers?
A) lipids
B) carbohydrates
C) proteins
D) nucleic acids
E) lipids, carbohydrates, proteins, and nucleic acids all consist of only macromolecular polymers
B
Which of the following is not a polymer?
A) glucose
B) starch
C) cellulose
D) chitin
E) DNA
A
What is the chemical reaction mechanism by which cells make polymers from monomers?
A) phosphodiester linkages
B) hydrolysis
C) dehydration reactions
D) ionic bonding of monomers
E) the formation of disulfide bridges between monomers
C
On food packages, to what does the term insoluble fiber refer?
A) cellulose
B) polypeptides
C) starch
D) amylopectin
E) chitin
A
Which of the following is true of both starch and cellulose?
A) They are both polymers of glucose.
B) They are cis-trans isomers of each other.
C) They can both be digested by humans.
D) They are both used for energy storage in plants.
E) They are both structural components of the plant cell wall.
A
Humans can digest starch but not cellulose because
A) the monomer of starch is glucose, while the monomer of cellulose is galactose.
B) humans have enzymes that can hydrolyze the β glycosidic linkages of starch but not the α glycosidic linkages of cellulose.
C) humans have enzymes that can hydrolyze the α glycosidic linkages of starch but not the β glycosidic linkages of cellulose.
D) humans harbor starch-digesting bacteria in the digestive tract.
E) the monomer of starch is glucose, while the monomer of cellulose is glucose with a nitrogen-containing group.
C
Which of the following statements concerning saturated fats is not true?
A) They are more common in animals than in plants.
B) They have multiple double bonds in the carbon chains of their fatty acids.
C) They generally solidify at room temperature.
D) They contain more hydrogen than unsaturated fats having the same number of carbon atoms.
E) They are one of several factors that contribute to atherosclerosis.
B
There are 20 different amino acids. What makes one amino acid different from another?
A) different side chains (R groups) attached to a carboxyl carbon
B) different side chains (R groups) attached to the amino groups
C) different side chains (R groups) attached to an α carbon
D) different structural and optical isomers
E) different asymmetric carbons
C
Which type of interaction stabilizes the α helix and the β pleated sheet structures of proteins?
A) hydrophobic interactions
B) disulfide bonds
C) ionic bonds
D) hydrogen bonds
E) peptide bonds
D
The tertiary structure of a protein is the
A) bonding together of several polypeptide chains by weak bonds.
B) order in which amino acids are joined in a polypeptide chain.
C) unique three-dimensional shape of the fully folded polypeptide.
D) organization of a polypeptide chain into an α helix or β pleated sheet.
E) overall protein structure resulting from the aggregation of two or more polypeptide subunits.
C
What is the term used for a protein molecule that assists in the proper folding of other proteins?
A) tertiary protein
B) chaperonin
C) enzyme protein
D) renaturing protein
E) denaturing protein
B
Which of the following descriptions best fits the class of molecules known as nucleotides?
A) a nitrogenous base and a phosphate group
B) a nitrogenous base and a pentose sugar
C) a nitrogenous base, a phosphate group, and a pentose sugar
D) a phosphate group and an adenine or uracil
E) a pentose sugar and a purine or pyrimidine
C
If a DNA sample were composed of 10% thymine, what would be the percentage of guanine?
A) 10
B) 20
C) 40
D) 80
E) impossible to tell from the information given
C
When biologists wish to study the internal ultrastructure of cells, they can achieve the finest resolution by using
A) a phase-contrast light microscope.
B) a scanning electron microscope.
C) a transmission electronic microscope.
D) a confocal fluorescence microscope.
E) a super-resolution fluorescence microscope.
C
A primary objective of cell fractionation is to
A) view the structure of cell membranes.
B) sort cells based on their size and weight.
C) determine the size of various organelles.
D) separate the major organelles so that their particular functions can be determined.
E) separate lipid-soluble from water-soluble molecules.
D
All of the following are part of a prokaryotic cell except
A) DNA.
B) a cell wall.
C) a plasma membrane.
D) ribosomes.
E) an endoplasmic reticulum.
E
Which of the following is a major cause of the size limits for certain types of cells?
A) limitation on the strength and integrity of the plasma membrane as cell size increases
B) the difference in plasma membranes between prokaryotes and eukaryotes
C) evolutionary progression in cell size; more primitive cells have smaller sizes
D) the need for a surface area of sufficient area to support the cell's metabolic needs
E) rigid cell walls that limit cell size expansion
D
Large numbers of ribosomes are present in cells that specialize in producing which of the following molecules?
A) lipids
B) glycogen
C) proteins
D) cellulose
E) nucleic acids
C
Which structure is the site of the synthesis of proteins that may be exported from the cell?
A) rough ER
B) lysosomes
C) plasmodesmata
D) Golgi vesicles
E) free cytoplasmic ribosomes
A
Hydrolytic enzymes must be segregated and packaged to prevent general destruction of cellular components. Which of the following organelles contains these hydrolytic enzymes in animal cells?
A) chloroplast
B) lysosome
C) central vacuole
D) peroxisome
E) glyoxysome
B
The liver is involved in detoxification of many poisons and drugs. Which of the following structures is primarily involved in this process and therefore abundant in liver cells?
A) rough ER
B) smooth ER
C) Golgi apparatus
D) nuclear envelope
E) transport vesicles
B
Which organelle is the primary site of ATP synthesis in eukaryotic cells?
A) lysosome
B) vacuole
C) mitochondrion
D) Golgi apparatus
E) peroxisome
C
Movement of vesicles within the cell depends on what cellular structures?
A) microtubules and motor proteins
B) actin filaments and microtubules
C) actin filaments and ribosomes
D) centrioles and motor proteins
E) actin filaments and motor proteins
A
Singer and Nicolson's fluid mosaic model of the membrane proposed that
A) membranes are a phospholipid bilayer.
B) membranes are a phospholipid bilayer between two layers of hydrophilic proteins.
C) membranes are a single layer of phospholipids and proteins.
D) membranes consist of protein molecules embedded in a fluid bilayer of phospholipids.
E) membranes consist of a mosaic of polysaccharides and proteins.
D
The presence of cholesterol in the plasma membranes of some animals
A) enables the membrane to stay fluid more easily when cell temperature drops.
B) enables the animal to remove hydrogen atoms from saturated phospholipids.
C) enables the animal to add hydrogen atoms to unsaturated phospholipids.
D) makes the membrane less flexible, allowing it to sustain greater pressure from within the cell.
E) makes the animal more susceptible to circulatory disorders.
A
According to the fluid mosaic model of cell membranes, which of the following is a true statement about membrane phospholipids?
A) They can move laterally along the plane of the membrane.
B) They frequently flip-flop from one side of the membrane to the other.
C) They occur in an uninterrupted bilayer, with membrane proteins restricted to the surface of the membrane.
D) They are free to depart from the membrane and dissolve in the surrounding solution.
E) They have hydrophilic tails in the interior of the membrane.
A
Which of the following is a reasonable explanation for why unsaturated fatty acids help keep any membrane more fluid at lower temperatures?
A) The double bonds form kinks in the fatty acid tails, preventing adjacent lipids from packing tightly.
B) Unsaturated fatty acids have a higher cholesterol content and therefore more cholesterol in membranes.
C) Unsaturated fatty acids are more polar than saturated fatty acids.
D) The double bonds block interaction among the hydrophilic head groups of the lipids.
E) The double bonds result in shorter fatty acid tails and thinner membranes.
A
The primary function of polysaccharides attached to the glycoproteins and glycolipids of animal cell membranes is
A) to facilitate diffusion of molecules down their concentration gradients.
B) to actively transport molecules against their concentration gradients.
C) to maintain the integrity of a fluid mosaic membrane.
D) to maintain membrane fluidity at low temperatures.
E) to mediate cell-to-cell recognition.
E
A protein that spans the phospholipid bilayer one or more times is
A) a transmembrane protein.
B) an integral protein.
C) a peripheral protein.
D) an integrin.
E) a glycoprotein.
A
What kinds of molecules pass through a cell membrane most easily?
A) large and hydrophobic
B) small and hydrophobic
C) large polar
D) ionic
E) monosaccharides such as glucose
B
Which of the following statements is correct about diffusion?
A) It is very rapid over long distances.
B) It requires an expenditure of energy by the cell.
C) It is a passive process in which molecules move from a region of higher concentration to a region of lower concentration.
D) It is an active process in which molecules move from a region of lower concentration to one of higher concentration.
E) It requires integral proteins in the cell membrane.
C
Water passes quickly through cell membranes because
A) the bilayer is hydrophilic.
B) it moves through hydrophobic channels.
C) water movement is tied to ATP hydrolysis.
D) it is a small, polar, charged molecule.
E) it moves through aquaporins in the membrane.
E
Which of the following statements correctly describes the normal tonicity conditions for typical plant and animal cells?
A) The animal cell is in a hypotonic solution, and the plant cell is in an isotonic solution.
B) The animal cell is in an isotonic solution, and the plant cell is in a hypertonic solution.
C) The animal cell is in a hypertonic solution, and the plant cell is in an isotonic solution.
D) The animal cell is in an isotonic solution, and the plant cell is in a hypotonic solution.
E) The animal cell is in a hypertonic solution, and the plant cell is in a hypotonic solution.
D
What is the voltage across a membrane called?
A) water potential
B) chemical gradient
C) membrane potential
D) osmotic potential
E) electrochemical gradient
C
An organism with a cell wall would most likely be unable to take in materials through
A) diffusion.
B) osmosis.
C) active transport.
D) phagocytosis.
E) facilitated diffusion.
D
Which of the following is (are) true for anabolic pathways?
A) They do not depend on enzymes.
B) They are usually highly spontaneous chemical reactions.
C) They consume energy to build up polymers from monomers.
D) They release energy as they degrade polymers to monomers.
E) They consume energy to decrease the entropy of the organism and its environment.
C
For living organisms, which of the following is an important consequence of the first law of thermodynamics?
A) The energy content of an organism is constant.
B) The organism ultimately must obtain all of the necessary energy for life from its environment.
C) The entropy of an organism decreases with time as the organism grows in complexity.
D) Organisms grow by converting energy into organic matter.
E) Life does not obey the first law of thermodynamics.
B
Whenever energy is transformed, there is always an increase in the
A) free energy of the system.
B) free energy of the universe.
C) entropy of the system.
D) entropy of the universe.
E) enthalpy of the universe.
D
Which of the following is true for all exergonic reactions?
A) The products have more total energy than the reactants.
B) The reaction proceeds with a net release of free energy.
C) The reaction goes only in a forward direction: all reactants will be converted to products, but no products will be converted to reactants.
D) A net input of energy from the surroundings is required for the reactions to proceed.
E) The reactions are rapid.
B
When chemical, transport, or mechanical work is done by an organism, what happens to the heat generated?
A) It is used to power yet more cellular work.
B) It is used to store energy as more ATP.
C) It is used to generate ADP from nucleotide precursors.
D) It is lost to the environment.
E) It is transported to specific organs such as the brain.
D
Reactants capable of interacting to form products in a chemical reaction must first overcome a thermodynamic barrier known as the reaction's
A) entropy.
B) activation energy.
C) endothermic level.
D) equilibrium point.
E) free-energy content.
B
Which of the following statements regarding enzymes is true?
A) Enzymes increase the rate of a reaction by making the reaction more exergonic.
B) Enzymes increase the rate of a reaction by lowering the activation energy barrier.
C) Enzymes increase the rate of a reaction by reducing the rate of reverse reactions.
D) Enzymes change the equilibrium point of the reactions they catalyze.
E) Enzymes make the rate of a reaction independent of substrate concentrations.
B
The active site of an enzyme is the region that
A) binds allosteric regulators of the enzyme.
B) is involved in the catalytic reaction of the enzyme.
C) binds noncompetitive inhibitors of the enzyme.
D) is inhibited by the presence of a coenzyme or a cofactor.
B
How does a noncompetitive inhibitor decrease the rate of an enzyme reaction?
A) by binding at the active site of the enzyme
B) by changing the shape of the enzyme's active site
C) by changing the free energy change of the reaction
D) by acting as a coenzyme for the reaction
E) by decreasing the activation energy of the reaction
B
Which of the following statements describes enzyme cooperativity?
A) A multienzyme complex contains all the enzymes of a metabolic pathway.
B) A product of a pathway serves as a competitive inhibitor of an early enzyme in the pathway.
C) A substrate molecule bound to an active site of one subunit promotes substrate binding to the active site of other subunits.
D) Several substrate molecules can be catalyzed by the same enzyme.
E) A substrate binds to an active site and inhibits cooperation between enzymes in a pathway.
C
Where does glycolysis take place in eukaryotic cells?
A) mitochondrial matrix
B) mitochondrial outer membrane
C) mitochondrial inner membrane
D) mitochondrial intermembrane space
E) cytosol
E
Which of the following statements describes NAD+?
A) NAD+ is reduced to NADH during glycolysis, pyruvate oxidation, and the citric acid cycle.
B) NAD+ has more chemical energy than NADH.
C) NAD+ is oxidized by the action of hydrogenases.
D) NAD+ can donate electrons for use in oxidative phosphorylation.
E) In the absence of NAD+, glycolysis can still function.
A
The ATP made during glycolysis is generated by
A) substrate-level phosphorylation.
B) electron transport.
C) photophosphorylation.
D) chemiosmosis.
E) oxidation of NADH to NAD+.
A
The oxygen consumed during cellular respiration is involved directly in which process or event?
A) glycolysis
B) accepting electrons at the end of the electron transport chain
C) the citric acid cycle
D) the oxidation of pyruvate to acetyl CoA
E) the phosphorylation of ADP to form ATP
B
Which process in eukaryotic cells will proceed normally whether oxygen (O2) is present or absent?
A) electron transport
B) glycolysis
C) the citric acid cycle
D) oxidative phosphorylation
E) chemiosmosis
B
An electron loses potential energy when it
A) shifts to a less electronegative atom.
B) shifts to a more electronegative atom.
C) increases its kinetic energy.
D) increases its activity as an oxidizing agent.
E) moves further away from the nucleus of the atom.
B
Carbon dioxide (CO2) is released during which of the following stages of cellular respiration?
A) glycolysis and the oxidation of pyruvate to acetyl CoA
B) oxidation of pyruvate to acetyl CoA and the citric acid cycle
C) the citric acid cycle and oxidative phosphorylation
D) oxidative phosphorylation and fermentation
E) fermentation and glycolysis
B
During aerobic respiration, electrons travel downhill in which sequence?
A) food → citric acid cycle → ATP → NAD+
B) food → NADH → electron transport chain → oxygen
C) glucose → pyruvate → ATP → oxygen
D) glucose → ATP → electron transport chain → NADH
E) food → glycolysis → citric acid cycle → NADH → ATP
B
Where are the proteins of the electron transport chain located?
A) cytosol
B) mitochondrial outer membrane
C) mitochondrial inner membrane
D) mitochondrial intermembrane space
E) mitochondrial matrix
C
Energy released by the electron transport chain is used to pump H+ into which location in eukaryotic cells?
A) cytosol
B) mitochondrial outer membrane
C) mitochondrial inner membrane
D) mitochondrial intermembrane space
E) mitochondrial matrix
D
One function of both alcohol fermentation and lactic acid fermentation is to
A) reduce NAD+ to NADH.
B) reduce FAD+ to FADH2.
C) oxidize NADH to NAD+.
D) reduce FADH2 to FAD+.
E) do none of the above.
C
Which metabolic pathway is common to both fermentation and cellular respiration of a glucose molecule?
A) the citric acid cycle
B) the electron transport chain
C) glycolysis
D) synthesis of acetyl CoA from pyruvate
E) reduction of pyruvate to lactate
C
Which of the following are products of the light reactions of photosynthesis that are utilized in the Calvin cycle?
A) CO2 and glucose
B) H2O and O2
C) ADP, i, and NADP+
D) electrons and H+
E) ATP and NADPH
E
Where does the Calvin cycle take place?
A) stroma of the chloroplast
B) thylakoid membrane
C) cytoplasm surrounding the chloroplast
D) interior of the thylakoid (thylakoid space)
E) outer membrane of the chloroplast
A
When oxygen is released as a result of photosynthesis, it is a direct by-product of
A) reducing NADP+.
B) splitting water molecules.
C) chemiosmosis.
D) the electron transfer system of photosystem I.
E) the electron transfer system of photosystem II.
B
Which of the following are directly associated with photosystem I?
A) harvesting of light energy by ATP
B) receiving electrons from the thylakoid membrane electron transport chain
C) generation of molecular oxygen
D) extraction of hydrogen electrons from the splitting of water
E) passing electrons to the thylakoid membrane electron transport chain
B
What is the primary function of the Calvin cycle?
A) use ATP to release carbon dioxide
B) use NADPH to release carbon dioxide
C) split water and release oxygen
D) transport RuBP out of the chloroplast
E) synthesize simple sugars from carbon dioxide
E
Assume a thylakoid is somehow punctured so that the interior of the thylakoid is no longer separated from the stroma. This damage will have the most direct effect on which of the following processes?
A) the splitting of water
B) the absorption of light energy by chlorophyll
C) the flow of electrons from photosystem II to photosystem I
D) the synthesis of ATP
E) the reduction of NADP+
D
In a plant cell, where are the ATP synthase complexes located?
A) thylakoid membrane only
B) plasma membrane only
C) inner mitochondrial membrane only
D) thylakoid membrane and inner mitochondrial membrane
E) thylakoid membrane and plasma membrane
D
Which of the following statements best describes the relationship between photosynthesis and respiration?
A) Respiration runs the biochemical pathways of photosynthesis in reverse.
B) Photosynthesis stores energy in complex organic molecules, whereas respiration releases it.
C) Photosynthesis occurs only in plants and respiration occurs only in animals.
D) ATP molecules are produced in photosynthesis and used up in respiration.
E) Respiration is anabolic and photosynthesis is catabolic.
B
Generation of proton gradients across membranes occurs during
A) photosynthesis.
B) respiration.
C) both photosynthesis and respiration.
D) neither photosynthesis nor respiration.
E) photorespiration.
C
Carotenoids are often found in foods that are considered to have antioxidant properties in human nutrition. What related function do they have in plants?
A) They serve as accessory pigments to increase light absorption.
B) They protect against oxidative damage from excessive light energy.
C) They shield the sensitive chromosomes of the plant from harmful ultraviolet radiation.
D) They reflect orange light and enhance red light absorption by chlorophyll.
E) They take up and remove toxins from the groundwater.
B
CAM plants keep stomata closed in daytime, thus reducing loss of water. They can do this because they
A) fix CO2 into organic acids during the night.
B) fix CO2 into sugars in the bundle-sheath cells.
C) fix CO2 into pyruvate in the mesophyll cells.
D) use the enzyme phosphofructokinase, which outcompetes rubisco for CO2.
E) use photosystem I and photosystem II at night
A
The centromere is a region in which
A) chromatids remain attached to one another until anaphase.
B) metaphase chromosomes become aligned at the metaphase plate.
C) chromosomes are grouped during telophase.
D) the nucleus is located prior to mitosis.
E) new spindle microtubules form at either end.
A
Where do the microtubules of the spindle originate during mitosis in both plant and animal cells?
A) centromere
B) centrosome
C) centriole
D) chromatid
E) kinetochore
B
The stage of mitosis in which sister chromatids separate and move to opposite poles of the dividing cell
A) telophase
B) anaphase
C) prometaphase
D) metaphase
E) prophase
E
What is a cleavage furrow?
A) a ring of vesicles forming a cell plate
B) the separation of divided prokaryotes
C) a groove in the plasma membrane between daughter nuclei
D) the metaphase plate where chromosomes attach to the spindle
E) the space that is created between two chromatids during anaphase
C
Which of the following is a protein maintained at constant levels throughout the cell cycle that requires cyclin to become catalytically active?
A) PDGF
B) MPF
C) protein kinase
D) cyclin
E) Cdk
E
After which checkpoint is the cell first committed to continue the cell cycle through M?
A) G0
B) G1
C) G2
D) S
E) previous M
B
Which of the following most accurately describes a cyclin?
A) It is present in similar concentrations throughout the cell cycle.
B) It is activated to phosphorylate by complexing with a Cdk.
C) It decreases in concentration when MPF activity increases.
D) It activates a Cdk molecule when it is in sufficient concentration.
E) It activates a Cdk when its concentration is decreased.
D
Which of the following best describes a karyotype?
A) a pictorial representation of all the genes for a species
B) a display of each of the chromosomes of a single cell
C) the combination of all the maternal and paternal chromosomes of a species
D) the collection of all the chromosomes in an individual organism
E) a photograph of all the cells with missing or extra chromosomes
B
After telophase I of meiosis, the chromosomal makeup of each daughter cell is
A) diploid, and the chromosomes are each composed of a single chromatid.
B) diploid, and the chromosomes are each composed of two chromatids.
C) haploid, and the chromosomes are each composed of a single chromatid.
D) haploid, and the chromosomes are each composed of two chromatids.
E) tetraploid, and the chromosomes are each composed of two chromatids.
D
Sister chromatids are separated from each other.
A) The statement is true for mitosis only.
B) The statement is true for meiosis I only.
C) The statement is true for meiosis II only.
D) The statement is true for mitosis and meiosis I.
E) The statement is true for mitosis and meiosis II.
E
Independent assortment of chromosomes is a result of
A) the random and independent way in which each pair of homologous chromosomes lines up at the metaphase plate during meiosis I.
B) the random nature of the fertilization of ova by sperm.
C) the random distribution of the sister chromatids to the two daughter cells during anaphase II.
D) the relatively small degree of homology shared by the X and Y chromosomes.
E) the random and independent way in which each pair of homologous chromosomes lines up at the metaphase plate during meiosis I, the random nature of the fertilization of ova by sperm, the random distribution of the sister chromatids to the two daughter cells during anaphase II, and the relatively small degree of homology shared by the X and Y chromosomes.
A
A human cell containing 22 autosomes and a Y chromosome is
A) a sperm.
B) an egg.
C) a zygote.
D) a somatic cell of a male.
E) a somatic cell of a female.
A
Why did the F1 offspring of Mendel's classic pea cross always look like one of the two parental varieties?
A) No genes interacted to produce the parental phenotype.
B) Each allele affected phenotypic expression.
C) The traits blended together during fertilization.
D) One phenotype was completely dominant over another.
E) Different genes interacted to produce the parental phenotype
D
What was the most significant conclusion that Gregor Mendel drew from his experiments with pea plants?
A) There is considerable genetic variation in garden peas.
B) Traits are inherited in discrete units, and are not the results of "blending."
C) Recessive genes occur more frequently in the F1 generation than do dominant ones.
D) Genes are composed of DNA.
E) An organism that is homozygous for many recessive traits is at a disadvantage.
B
Two plants are crossed, resulting in offspring with a 3:1 ratio for a particular trait. What does this suggest?
A) that the parents were true-breeding for contrasting traits
B) that the trait shows incomplete dominance
C) that a blending of traits has occurred
D) that the parents were both heterozygous for a single trait
E) that each offspring has the same alleles for each of two traits
D
A sexually reproducing animal has two unlinked genes, one for head shape (H) and one for tail length (T). Its genotype is HhTt. Which of the following genotypes is possible in a gamete from this organism?
A) tt
B) Hh
C) HhTt
D) T
E) HT
E
Mendel accounted for the observation that traits which had disappeared in the F1 generation reappeared in the F2 generation by proposing that
A) new mutations were frequently generated in the F2 progeny, "reinventing" traits that had been lost in the F1.
B) the mechanism controlling the appearance of traits was different between the F1 and the F2 plants.
C) traits can be dominant or recessive, and the recessive traits were obscured by the dominant ones in the F1.
D) the traits were lost in the F1 due to dominance of the parental traits.
E) members of the F1 generation had only one allele for each trait, but members of the F2 had two alleles for each trait.
C
The fact that all seven of the pea plant traits studied by Mendel obeyed the principle of independent assortment most probably indicates which of the following?
A) None of the traits obeyed the law of segregation.
B) The diploid number of chromosomes in the pea plants was 7.
C) All of the genes controlling the traits were located on the same chromosome.
D) All of the genes controlling the traits behaved as if they were on different chromosomes.
E) The formation of gametes in plants occurs by mitosis only.
D
Mendel's observation of the segregation of alleles in gamete formation has its basis in which of the following phases of cell division?
A) prophase I of meiosis
B) anaphase II of meiosis
C) metaphase I of meiosis
D) anaphase I of meiosis
E) anaphase of mitosis
D
Black fur in mice (B) is dominant to brown fur (b). Short tails (T) are dominant to long tails (t). What fraction of the progeny of crosses BbTt × BBtt will be expected to have black fur and long tails?
A) 1/16
B) 3/16
C) 3/8
D) 1/2
E) 9/16
D
In certain plants, tall is dominant to short. If a heterozygous plant is crossed with a homozygous tall plant, what is the probability that the offspring will be short?
A) 1
B) 1/2
C) 1/4
D) 1/6
E) 0
E
Which of the following describes the ability of a single gene to have multiple phenotypic effects?
A) incomplete dominance
B) multiple alleles
C) pleiotropy
D) epistasis
C
Which of the following is an example of polygenic inheritance?
A) pink flowers in snapdragons
B) the ABO blood group in humans
C) Huntington's disease in humans
D) white and purple flower color in peas
E) skin pigmentation in humans
E
Which of the following provides an example of epistasis?
A) Recessive genotypes for each of two genes (aabb) results in an albino corn snake.
B) The allele b17 produces a dominant phenotype, although b1 through b16 do not.
C) In rabbits and many other mammals, one genotype (cc) prevents any fur color from developing.
D) In Drosophila (fruit flies), white eyes can be due to an X-linked gene or to a combination of other genes.
E) In cacti, there are several genes for the type of spines.
C
Why are dominantly inherited fatal disorders extremely rare?
A) Because they will get silenced by other genes to save the life of the individual that carries them.
B) Because an individual that inherits a copy of the dominant allele will always suffer from the condition and will likely die before passing the gene to future generations.
C) Because heterozygous will just be carriers and not suffer the condition.
D) Because they get treated early in life.
E) Because the dominant alleles responsible for fatal disorders will typically mutate before they can have an effect.
B
When Thomas Hunt Morgan crossed his red-eyed F1 generation flies to each other, the F2 generation included both red- and white-eyed flies. Remarkably, all the white-eyed flies were male. What was the explanation for this result?
A) The gene involved is on the Y chromosome.
B) The gene involved is on the X chromosome.
C) The gene involved is on an autosome, but only in males.
D) Other male-specific factors influence eye color in flies.
E) Other female-specific factors influence eye color in flies.
B
Which of the following is the meaning of the chromosome theory of inheritance as expressed in the early 20th century?
A) Individuals inherit particular chromosomes attached to genes.
B) Mendelian genes are at specific loci on the chromosome and in turn segregate during meiosis.
C) Homologous chromosomes give rise to some genes and crossover chromosomes to other genes.
D) No more than a single pair of chromosomes can be found in a healthy normal cell.
E) Natural selection acts on certain chromosome arrays rather than on genes.
B
Calico cats are female because
A) the males die during embryonic development.
B) a male inherits only one of the two X-linked genes controlling hair color.
C) the Y chromosome has a gene blocking orange coloration.
D) only females can have Barr bodies.
E) multiple crossovers on the Y chromosome prevent orange pigment production.
B
Which of the following statements is true of linkage?
A) The closer two genes are on a chromosome, the lower the probability that a crossover will occur between them.
B) The observed frequency of recombination of two genes that are far apart from each other has a maximum value of 100%.
C) All of the traits that Mendel studied–seed color, pod shape, flower color, and others–are due to genes linked on the same chromosome.
D) Linked genes are found on different chromosomes.
E) Crossing over occurs during prophase II of meiosis.
A
What does a frequency of recombination of 50% indicate?
A) The two genes are likely to be located on different chromosomes.
B) All of the offspring have combinations of traits that match one of the two parents.
C) The genes are located on sex chromosomes.
D) Abnormal meiosis has occurred.
E) Independent assortment is hindered.
A
What is the reason that linked genes are inherited together?
A) They are located close together on the same chromosome.
B) The number of genes in a cell is greater than the number of chromosomes.
C) Chromosomes are unbreakable.
D) Alleles are paired together during meiosis.
E) Genes align that way during metaphase I of meiosis.
A
The centimorgan (cM) is a unit named in honor of Thomas Hunt Morgan. To what is it equal?
A) the physical distance between two linked genes
B) 1% frequency of recombination between two genes
C) 1 nanometer of distance between two genes
D) the distance between a pair of homologous chromosomes
E) the recombination frequency between two genes assorting independently
B
Recombination between linked genes comes about for what reason?
A) Mutation on one homolog is different from that on the other homolog.
B) Independent assortment sometimes fails because Mendel had not calculated appropriately.
C) When genes are linked they always "travel" together at anaphase.
D) Crossovers between these genes result in chromosomal exchange.
E) Nonrecombinant chromosomes break and then re-join with one another.
D
One possible result of chromosomal breakage is for a fragment to join a nonhomologous chromosome. What is this alteration called?
A) deletion
B) transversion
C) inversion
D) translocation
E) duplication
D