Which of the following statements about gases is false?
A) Gases
are highly compressible.
B) Distances between molecules of gas
are very large compared to bond distances within molecules.
C)
Non-reacting gas mixtures are homogeneous.
D) Gases expand
spontaneously to fill the container they are placed in.
E) All
gases are colorless and odorless at room temperature.
E
Of the following, ________ has a strong acrid odor.
A)
N2O
B) CH4
C) CO
D) NH3
E) HCN
D
Of the following, ________ has the odor of rotten eggs.
A)
NH3
B) H2S
C) CO
D) NO2
E) HCN
B
One significant difference between gases and liquids is that
________.
A) a gas is made up of molecules
B) a gas expands
to fill its container
C) a gas may consist of both elements and
compounds
D) gases are always mixtures
E) All of the above
answers are correct.
B
Gaseous mixtures ________.
A) can only contain molecules
B)
are all heterogeneous
C) can only contain isolated atoms
D)
are all homogeneous
E) must contain both isolated atoms and molecules
D
Which of the following equations shows an incorrect relationship
between pressures given in terms of different units?
A) 1.20 atm
= 122 kPa
B) 152 mm Hg = 2.03 × 104 Pa
C) 0.760
atm = 578 mm Hg
D) 1.0 torr = 2.00 mm Hg
E) 1.00 atm = 760 torr
D
Which of the following is not a unit of pressure?
A) mm
Hg
B) mm
C) atm
D) Pa
E) psi
B
The pressure exerted by a column of liquid is equal to the product of
the height of the column times the gravitational constant times the
density of the liquid, P = ghd. How high a column of methanol (d =
0.79 g/mL) would be supported by a pressure that supports a 713 mm
column of mercury (d = 13.6 G/mL)?
A) 713 mm
B) 41
mm
C) 1.2 × 104 mm
D) 9.7 × 103 mm
E) 17 mm
C
Which statement about atmospheric pressure is false?
A) As air
becomes thinner, its density decreases.
B) Air actually has
weight.
C) With an increase in altitude, atmospheric pressure
increases as well.
D) The warmer the air, the lower the
atmospheric pressure.
E) Atmospheric pressure prevents water in
lakes, rivers, and oceans from boiling away.
C
In ideal gas equation calculations, expressing pressure in Pascals
(Pa), necessitates the use of the gas constant, R, equal to
________.
A) 0.08206 atm L mol-1K-1
B) 8.314
-Pa/mol-K
C) 62.36 L torr mol-1K-1
D) 1.987 cal
mol-1K-1
E) none of the above
B
The first person to investigate the relationship between the pressure
of a gas and its volume was ________.
A) Amadeo Avogadro
B)
Lord Kelvin
C) Jacques Charles
D) Robert Boyle
E)
Joseph Louis Gay-Lussac
D
Of the following, ________ is a correct statement of Boyle's
law.
A) PV = constant
B) P/V = constant
C) V/P =
constant
D) V/T = constant
E) n/P = constant
A
"Isothermal" means ________.
A) at constant
pressure
B) at constant temperature
C) at variable
temperature and pressure conditions
D) at ideal temperature and
pressure conditions
E) that ΔHrxn = 0
B
Of the following, ________ is a valid statement of Charles'
law.
A) P/T = constant
B) V/T = constant
C) PV =
constant
D) V = constant × n
E) V = constant × P
B
Which one of the following is a valid statement of Avogadro's
law?
A) P/T = constant
B) V/T = constant
C) PV =
constant
D) V = constant × n
E) V = constant × P
D
The temperature of an ideal gas at STP is ________.
A) 0
°C
B) -32 °F
C) -273 K
D) 0 °F
E) 0 K
A
Which of the following is not possible for an ideal gas?
A)
V1/T1 = V2/T2
B) V1/V2 = T1/T2
C) V2 = T2/T1 V1
D) V1T1 = V2T2
E) V1/V2 = T1/T2 = 0
D
The molar volume of a gas at STP is ________ L.
A)
0.08206
B) 62.36
C) 1.00
D) 22.4
E) 14.7
D
Standard temperature and pressure (STP), in the context of gases,
refers to ________.
A) 298.15 K and 1 atm
B) 273.15 K and 1
atm
C) 298.15 K and 1 torr
D) 273.15 K and 1 pascal
E)
273.15 K and 1 torr
B
Sodium bicarbonate is reacted with concentrated hydrochloric acid at
37.0 °C and 1.00 atm. The reaction of 6.00 kg of bicarbonate with
excess hydrochloric acid under these conditions will produce ________
L of CO2.
A) 1.09 × 102
B) 2.85 × 104
C) 1.82 × 104
D) 8.70 × 102
E) 1.82 × 103
E
How much CO2 (L) is produced when 2.10 kg of sodium bicarbonate
reacts with excess hydrochloric acid at 25.0 °C and 1.23 atm?
A)
4.17 × 10-1
B) 4.98 × 102
C) 4.17 × 101
D) 3.50
E) 4.98 × 10-1
B
The volume of a sample of gas (2.49 g) was 752 mL at 1.98 atm and 62
°C. The gas is ________.
A) SO2
B) SO3
C) NH3
D)
NO2
E) Ne
D
The density of NO2 in a 4.50 L tank at 760.0 torr and 25.0 °C is
________ g/L.
A) 1.64
B) 9.30
C) 1.68
D)
1.88
E) 3.27
D
A 255 mL round-bottom flask is weighed and found to have a mass of
114.85 g. A few milliliters of an easily vaporized liquid are added to
the flask and the flask is immersed in a boiling water bath. All of
the liquid vaporizes at the boiling temperature of water, filling the
flask with vapor. When all of the liquid has vaporized, the flask is
removed from the bath, cooled, dried, and reweighed. The new mass of
the flask and the condensed vapor is 115.23 g. Which of the following
compounds could the liquid be? (Assume the ambient pressure is 1
atm.)
A) ethanol
B) propanol
C) butanol
D)
hexanol
E) methanol
A
A sample of an unknown volatile liquid was injected into a Dumas
flask (mflask = 27.0928g, Vflask = 0.1040 L) and
heated until no visible traces of the liquid could be found. The flask
and its contents were then rapidly cooled and reweighed
(mflask+vapor = 27.4593g). The atmospheric pressure and
temperature during the experiment were 0.976 atm and 18.0 °C,
respectively. The unknown volatile liquid was ________.
A)
C6H12
B) C6H14
C) C7H14
D) C7H16
E) C6H6
B
The density of air at STP is 1.285 g/L. Which of the following cannot
be used to fill a balloon that will float in air at STP?
A)
CH4
B) NO
C) Ne
D) NH3
E) HF
B
The average kinetic energy of the particles of a gas is ________
proportional to ________.
A) directly, the rms speed
B)
directly, the square root of the rms speed
C) inversely, the
square of the rms speed
D) directly, the square of the rms
speed
E) inversely, the particle mass
D
The kinetic-molecular theory predicts that pressure rises as the
temperature of a gas increases because ________.
A) the average
kinetic energy of the gas molecules decreases
B) the gas
molecules collide more frequently with the wall
C) the gas
molecules collide less frequently with the wall
D) the gas
molecules collide more energetically with the wall
E) B and D
E
According to kinetic-molecular theory, in which of the following
gases will the root-mean-square speed of the molecules be the highest
at 200 °C?
A) HCl
B) Cl2
C) H2O
D) SF6
E)
None. The molecules of all gases have the same root-mean-square speed
at any given temperature.
C
According to kinetic-molecular theory, if the temperature of a gas is
raised from 100 °C to 200 °C, the average kinetic energy of the gas
will ________.
A) double
B) increase by a factor of
1.27
C) increase by a factor of 100
D) decrease by
half
E) decrease by a factor of 100
B
Which of the following is not part of the kinetic-molecular
theory?
A) Atoms are neither created nor destroyed by ordinary
chemical reactions.
B) Attractive and repulsive forces between
gas molecules are negligible.
C) Gases consist of molecules in
continuous, random motion.
D) Collisions between gas molecules do
not result in the loss of energy.
E) The volume occupied by all
of the gas molecules in a container is negligible compared to the
volume of the container.
A
Of the following gases, ________ will have the greatest rate of
effusion at a given temperature.
A) NH3
B) CH4
C)
Ar
D) HBr
E) HCl
B
A tank containing both HF and HBr gases developed a leak. The ratio
of the rate of effusion of HF to the rate of effusion of HBr is
________.
A) 4.04
B) 0.247
C) 2.01
D)
0.497
E) 16.3
C
Which of the following gases would have a similar rate of effusion to
CO at 350 K?
A) H2
B) He
C) CO2
D) N2
E) NO2
D
What is the temperature at STP?
A) 0 °C
B) 25 °C
C)
-32 °F
D) 298 k
E) 0 k
A
Arrange the following gases in order of increasing average molecular speed at 25 °C.
He, O2, CO2, N2
A) He < N2 < O2 < CO2
B) He < O2 < N2 <
CO2
C) CO2 < O2 < N2 < He
D) CO2 < N2 < O2
< He
E) CO2 < He < N2 < O2
C
Arrange the following gases in order of increasing average molecular speed at 25 °C.
Cl2, O2, F2, N2
A) Cl2 < F2 < O2 < N2
B) Cl2 < O2 < F2 <
N2
C) N2 < F2 < Cl2 < O2
D) Cl2 < F2 < N2
< O2
E) F2 < O2 < N2 < Cl2
A
Which one of the following gases would have the highest average
molecular speed at 25 °C?
A) O2
B) N2
C) CO2
D)
CH4
E) SF6
D
A sample of oxygen gas (O2) was found to effuse at a rate equal to
three times that of an unknown gas. The molecular weight of the
unknown gas is ________ g/mol.
A) 288
B) 96
C)
55
D) 4
E) 10.7
A
A sample of oxygen gas was found to effuse at a rate equal to two
times that of an unknown gas. The molecular weight of the unknown gas
is ________ g/mol.
A) 64
B) 128
C) 8
D) 16
E) 8.0
B
Two gases start to escape from a container and one of the gases
effuses 1.25 times as fast as the other one. The two gases could have
been ________.
A) NO and SF6
B) CO and CO2
C) CO2 and
Cl2
D) Cl2 and CO
E) CO2 and SF6
B
Carbon dioxide and an unknown gas start to effuse from a container
and the carbon dioxide takes 4.69 times as long to escape as the
unknown gas. What is the identity of the unknown gas?
A)
Br2
B) NO2
C) HCl
D) CO
E) H2
E
How much faster does 79Br2 effuse than
81Br2?
A) 0.975 times as fast
B) 1.025
times as fast
C) 1.013 times as fast
D) 0.988 times as
fast
E) Cannot be determined
C
An ideal gas differs from a real gas in that the molecules of an
ideal gas ________.
A) have no attraction for one another
B)
have appreciable molecular volumes
C) have a molecular weight of
zero
D) have no kinetic energy
E) have an average molecular mass
A
A real gas will behave most like an ideal gas under conditions of
________.
A) high temperature and high pressure
B) high
temperature and low pressure
C) low temperature and high
pressure
D) low temperature and low pressure
E) STP
B
Which one of the following gases would deviate the least from ideal
gas behavior?
A) Ne
B) CH3Cl
C) Kr
D) CO2
E) F2
A
Which noble gas is expected to show the largest deviations from the
ideal gas behavior?
A) helium
B) neon
C) argon
D)
krypton
E) xenon
E
The van der Waals equation for real gases recognizes that
________.
A) gas particles have non-zero volumes and interact
with each other
B) molar volumes of gases of different types are
different
C) the non-zero volumes of gas particles effectively
decrease the amount of "empty space" between them
D)
the molecular attractions between particles of gas decreases the
pressure exerted by the gas
E) all of the above statements are true
E
Real gases do not behave ideally at ________.
A) high pressure
and low temperature
B) low pressure and high temperature
C)
high pressure and high temperature
D) low pressure and low
temperature
E) high pressure only
A
A gas at a pressure of 10.0 Pa exerts a force of ________ N on an
area of 5.5 m2.
A) 55
B) 0.55
C) 5.5
D)
1.8
E) 18
A
A gas at a pressure of 325 torr exerts a force of ________ N on an
area of 5.5 m2.
A) 1.8 × 103
B) 59
C) 2.4 × 105
D) 0.018
E) 2.4
C
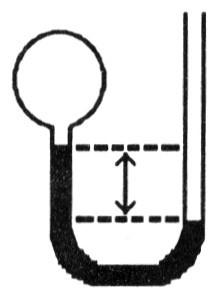
A gas vessel is attached to an open-end manometer containing a nonvolatile liquid of density 0.791 g/mL as shown below.
The difference in heights of the liquid in the two sides of the
manometer is 43.4 cm when the atmospheric pressure is 755 mm Hg. Given
that the density of mercury is 13.6 g/mL, the pressure of the enclosed
gas is ________ atm.
A) 1.03
B) 0.967
C) 0.993
D)
0.990
E) 0.987
B
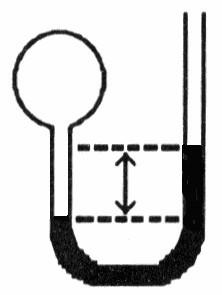
A gas vessel is attached to an open-end manometer filled with a nonvolatile liquid of density 0.993 g/mL as shown below.
The difference in heights of the liquid in the two sides of the
manometer is 32.3 mm when the atmospheric pressure is 765 mm Hg. Given
that the density of mercury is 13.6 g/mL, the pressure of the enclosed
gas is ________ atm.
A) 1.05
B) 1.01
C) 0.976
D)
0.993
E) 1.08
A
760 torr is equivalent to ________ mm Hg.
A) 380
B) 4.78 × 103
C) 1
D) 760
E) 121
D
A sample of a gas (5.0 mol) at 1.0 atm is expanded at constant
temperature from 10 L to 15 L. The final pressure is ________
atm.
A) 1.5
B) 7.5
C) 0.67
D) 3.3
E) 15
C
If 50.75 g of a gas occupies 10.0 L at STP, 129.3 g of the gas will
occupy ________ L at STP.
A) 3.92
B) 50.8
C)
12.9
D) 25.5
E) 5.08
D
A sample of H2 gas (12.28 g) occupies 100.0 L at 400.0 K and 2.00
atm. A sample weighing 9.49 g occupies ________ L at 353 K and 2.00
atm.
A) 109
B) 68.2
C) 54.7
D) 147
E) 77.3
B
A sample of a gas (1.50 mol) is contained in a 15.0 L cylinder. The
temperature is increased from 100 °C to 150 °C. The ratio of final
pressure to initial pressure [P2/P1] is
________.
A) 1.50
B) 0.667
C) 0.882
D)
1.13
E) 1.00
D
A sample of a gas originally at 25 °C and 1.00 atm pressure in a 2.5
L container is subject to a pressure of 0.85 atm and a temperature of
15 °C. The final volume of the gas is ________ L.
A) 3.0
B)
2.8
C) 2.6
D) 2.1
E) 0.38
B
A sample of a gas originally at 29 °C and 1.25 atm pressure in a 3.0
L container is allowed to contract until the volume is 2.2 L and the
temperature is 11 °C. The final pressure of the gas is ________
atm.
A) 2.9
B) 2.8
C) 1.6
D) 2.1
E) 0.38
C
If pressure and temperature are kept constant, the reaction of 38 mL of Cl2 gas with 22 mL of CH4 gas via the equation:
Cl2 (g) + CH4 (g) → HCl (g) + CH3Cl (g)
will produce a total of ________ mL of products.
A) 16
B)
30
C) 60
D) 38
E) 22
C
If pressure and temperature are kept constant, the reaction of 95 mL of Cl2 gas with 45 mL of CH4 gas via the equation:
Cl2 (g) + CH4 (g) → HCl (g) + CH3Cl (g)
will produce a total of ________ mL of products.
A) 95
B)
140
C) 45
D) 70
E) 50
B
If pressure and temperature are kept constant, the reaction of 29 mL
of N2 gas with 87 mL of H2 gas will form ________ mL of
ammonia.
A) 29
B) 43.5
C) 58
D) 174
E) 14.5
C
If pressure and temperature are kept constant, the reaction of 18 mL
of N2 gas with 54 mL of H2 gas will form ________ mL of
ammonia.
A) 108
B) 18
C) 9
D) 36
E) 27
D
The reaction of 50 mL of Cl2 gas with 50 mL of C2H4 gas via the equation:
Cl2 (g) + C2H4 (g) → C2H4Cl2 (g)
will produce a total of ________ mL of products if pressure and
temperature are kept constant.
A) 100
B) 50
C)
25
D) 125
E) 150
B
The amount of gas that occupies 60.82 L at 31.0 °C and 367 mm Hg is
________ mol.
A) 1.18
B) 0.850
C) 894
D)
11.6
E) 0.120
A
The amount of gas that occupies 36.52 L at 68.0 °C and 672 mm Hg is
________ mol.
A) 127
B) 1.15
C) 878
D)
24.4
E) 12.7
B
The pressure of a sample of CH4 gas (6.022 g) in a 30.0 L vessel at
402 K is ________ atm.
A) 2.42
B) 6.62
C) 0.413
D)
12.4
E) 22.4
C
The temperature of a sample of CH4 gas (10.34 g) in a 50.0 L vessel
at 1.33 atm is ________ °C.
A) 984
B) -195
C)
195
D) 1260
E) -1260
A
What is the volume (in m3) of a 0.25 mol of an unknown gas at a
pressure of 545.3 mm Hg and 15 °C?
A) 8.1 × 10-5
B) 8.2 × 10-3
C) 4.3 × 10-4
D) 2.2 × 10-1
E) 1.2 × 10-4
B
How many moles of an unknown gas are in a 325 mL container at a
pressure of 695 torr and 19 °C?
A) 9.42
B) 1.48 × 10-2
C) 12.4
D) 1.24 × 10-2
E) 80.6
D
A 0.133 mol sample of gas in a 525 mL container has a pressure of 312
torr. The temperature of the gas is ________ °C.
A) 20.3
B)
-253
C) -20.3
D) 203
E) 22.4
B
What mass (g) of carbon dioxide is contained in a 5.33 L vessel at 48
°C and 1.44 atm?
A) 12.8
B) 6.62 × 10-3
C) 0.291
D) 44.0
E) 151
A
The density of ammonia gas in a 4.32 L container at 837 torr and 45.0
°C is ________ g/L.
A) 3.86
B) 0.719
C) 0.432
D)
0.194
E) 4.22 × 10-2
B
What is the density of (g/L) CO2 at 1.53 atm and 45.2 °C?
A)
18.2
B) 2.58
C) 0.388
D) 9.99
E) 1.76
B
The molecular weight of a gas is ________ g/mol if 3.5 g of the gas
occupies 2.1 L at STP.
A) 41
B) 5.5 × 103
C) 37
D) 4.6 × 102
E) 2.7 × 10-2
C
The molecular weight of a gas is ________ g/mol if 6.7 g of the gas
occupies 6.3 L at STP.
A) 24
B) 3.6 × 103
C) 27
D) 3.0 × 102
E) 1.8 × 10-2
A
What is the molecular weight (g/mol) of an unknown gas that has a
density of 4.90 g/L at STP?
A) 1.10 × 10-2
B) 1.20 × 102
C) 1.10 × 102
D) 1.20 × 10-2
E) 1.10
C
The molecular weight of a gas that has a density of 7.10 g/L at 25.0
°C and 1.00 atm pressure is ________ g/mol.
A) 174
B)
14.6
C) 28.0
D) 5.75 × 10-3
E) 6.85 × 10-2
A
What is the molecular weight (g/mol) of an unknown gas that has a
density of 3.59 g/L at STP?
A) 7.37
B) 87.9
C)
0.0879
D) 80.5
E) 8.15 × 103
D
What is the density (g/L) of bromine gas at STP?
A) 6.53
B)
3.26
C) 3.56
D) 0.140
E) 7.13
E
) 5.25 g of zinc metal reacts with excess sulfuric acid to produce
hydrogen gas. What volume (L) of hydrogen is generated at STP?
A)
1.97
B) 118
C) 0.136
D) 1.80
E) 129
D
1.92 g of zinc metal reacts with excess hydrochloric acid to produce
hydrogen gas. What volume (L) of hydrogen is generated at STP?
A)
43.1
B) 0.656
C) 6.03 × 10-2
D) 0.719
E) 47.0
B
The volume of HCl gas required to react with excess magnesium metal
to produce 6.82 L of hydrogen gas at 2.19 atm and 35.0 °C is ________
L.
A) 6.82
B) 2.19
C) 13.6
D) 4.38
E) 3.41
C
What volume (mL) of fluorine gas is required to react with 1.28 g of
calcium bromide to form calcium fluoride and bromine gas at
STP?
A) 28.7
B) 144
C) 31.3
D) 157
E) 13.1
B
What volume (L) of fluorine gas is required to react with 2.31 g of
calcium bromide to form calcium fluoride and bromine gas at 8.19 atm
and 35.0 °C?
A) 3.57
B) 7.14
C) 357
D) 3.57 × 10-3
E) 4.05 × 10-3
D
What volume (mL) of sulfur dioxide can be produced by the complete
reaction of 3.82 g of calcium sulfite with excess HCl (aq), when the
final SO2 pressure is 827 torr at 44.0 °C?
A) 7.60 × 102
B) 1.39 × 10-4
C) 1.00 × 10-3
D) 0.106
E) 5.78 × 102
A
Automobile air bags use the decomposition of sodium azide as their source of gas for rapid inflation:
2NaN3 (s) → 2Na (s) + 3N2 (g).
What mass (g) of NaN3 is required to provide 40.0 L of N2 at 25.0 °C
and 763 torr?
A) 1.64
B) 1.09
C) 160
D)
71.1
E) 107
D
Automobile air bags use the decomposition of sodium azide as their source of gas for rapid inflation:
2NaN3 (s) → 2Na (s) + 3N2 (g).
What mass (g) of NaN3 is required to provide 26.5 L of N2 at 22.0 °C
and 1.10 atm?
A) 52.2
B) 700.
C) 0.807
D)
1.21
E) 1.10
A
The Mond process produces pure nickel metal via the thermal decomposition of nickel tetracarbonyl:
Ni(CO)4 (l) → Ni (s) + 4CO (g).
What volume (L) of CO is formed from the complete decomposition of
444 g of Ni(CO)4 at 752 torr and 22.0 °C?
A) 0.356
B)
63.7
C) 255
D) 20.2
E) 11.0
C
What volume (L) of NH3 gas at STP is produced by the complete
reaction of 7.5 g of H2O according to the following reaction?
Mg3N2 (s) + 6H2O (l) → 3Mg(OH)2 (aq) + 2NH3 (g)
A) 3.1
B) 9.3
C) 19
D) 28
E) 0.32
A
Ammonium nitrite undergoes thermal decomposition to produce only
gases:
NH4NO2 (s) → N2 (g) + 2H2O (g)
What volume (L) of
gas is produced by the decomposition of 35.0 g of NH4NO2(s) at 525 °C
and 1.5 atm?
A) 48
B) 160
C) 15
D) 72
E) 24
A
The thermal decomposition of potassium chlorate can be used to
produce oxygen in the laboratory.
2KClO3 (s) → 2KCl (s) + 3O2
(g)
What volume (L) of O2 gas at 25 °C and 1.00 atm pressure is
produced by the decomposition of 7.5 g of KClO3 (s)?
A)
4.5
B) 7.5
C) 2.3
D) 3.7
E) 11
C
Since air is a mixture, it does not have a "molar mass."
However, for calculation purposes, it is possible to speak of its
"effective molar mass." (An effective molar mass is a
weighted average of the molar masses of a mixture's components.) If
air at STP has a density of 1.285 g/L, its effective molar mass is
________ g/mol.
A) 26.94
B) 31.49
C) 30.00
D)
34.42
E) 28.80
E
A vessel contained N2, Ar, He, and Ne. The total pressure in the
vessel was 987 torr. The partial pressures of nitrogen, argon, and
helium were 44.0, 486, and 218 torr, respectively. The partial
pressure of neon in the vessel was ________ torr.
A) 42.4
B)
521
C) 19.4
D) 239
E) 760
D
What is the total pressure (atm) in a 10.0 L vessel that contains
2.34 mol of carbon dioxide, 1.73 mol of sulfur dioxide, and 4.50 mol
of argon at standard temperature?
A) 192
B) 1.92
C)
0.192
D) 19.2
E) 0.0192
D
A sample of Ne gas (2.5 L) at 4.5 atm and 25 °C was combined with 2.2
L of Ar gas at 6.3 atm and 25 °C at constant temperature in a 8.0 L
flask. Assuming the initial pressure in the flask was 0.00 atm and the
temperature upon mixing was 25 °C, what is the total pressure (atm) in
the flask?
A) 11.4
B) 10.0
C) 5.34
D) 10.8
E) 3.14
E
A sample of hydrogen gas (3.2 L) at 3.5 atm and 25 °C was combined
with 5.2 L of nitrogen gas at 7.3 atm and 25 °C at constant
temperature in a 15.0 L flask. Assuming the initial pressure in the
flask was 0.00 atm and the temperature upon mixing was 25 °C, what is
the total pressure (atm) in the flask?
A) 3.28
B)
5.85
C) 9.45
D) 15.4
E) 10.8
A
A gas mixture of Xe, Ne, and Ar has a total pressure of 12.20 atm.
What is the mole fraction of Xe if the partial pressures of Ne and Ar
are 2.10 and 4.50 atm, respectively?
A) 0.848
B)
0.459
C) 5.60
D) 2.18
E) 1.18
B
A gas mixture of He and Ar has a total pressure of 4.10 atm. What is
the mole fraction of Ar if the partial pressure of Ar is 1.50
atm?
A) 0.634
B) 2.6
C) 36.6
D) 0.366
E) 63.4
D
A gas mixture of N2 and H2 has a total pressure of 9.40 atm and
contains 11.3 mol of gas. If the partial pressure of N2 is 4.89 atm,
how many moles of H2 are in the mixture?
A) 4.51
B)
5.65
C) 5.42
D) 11.3
E) 5.88
C
A mixture of He and Ne at a total pressure of 0.95 atm is found to
contain 0.32 mol of He and 0.56 mol of Ne. The partial pressure of Ne
is ________ atm.
A) 1.7
B) 1.5
C) 0.60
D)
0.35
E) 1.0
C
A flask contains a mixture of N2 and H2 at a total pressure of 5.20
atm. If there are 1.00 mol of H2 and 8.00 mol of N2 in the flask, what
is the partial pressure (atm) of N2?
A) 2.60
B) 4.62
C)
5.20
D) 8.00
E) 0.578
B
Sodium hydride reacts with excess water to produce aqueous sodium
hydroxide and hydrogen gas:
NaH (s) + H2O (l) → NaOH (aq) + H2
(g)
A sample of NaH weighing ________ g will produce 982 mL of
gas at 28.0 °C and 765 torr, when the hydrogen is collected over
water. The vapor pressure of water at this temperature is 28
torr.
A) 2.93
B) 0.960
C) 0.925
D) 0.0388
E) 925
C
SO2 (5.00 g) and CO2 (5.00 g) were placed in a 750.0 mL container at
50.0 °C. The total pressure in the container was ________ atm.
A)
0.192
B) 4.02
C) 2.76
D) 6.78
E) 1.60
D
Sulfur dioxide (12.4 g) and carbon dioxide (12.4 g) are placed in a
500.0 mL container at 45.0 °C. What is the partial pressure (atm) of
sulfur dioxide in the container?
A) 0.0101
B) 5.06
C)
10.1
D) 648
E) 9.48
C
SO2 (5.00 g) and CO2 (5.00 g) were placed in a 750.0 mL container at
50.0 °C. The partial pressure of CO2 in the container was ________
atm.
A) 6.78
B) 2.76
C) 1.60
D) 0.192
E) 4.02
E
CO (5.00 g) and CO2 (5.00 g) were placed in a 750.0 mL container at
50.0 °C. The total pressure in the container was ________ atm.
A)
10.3
B) 4.02
C) 6.31
D) 0.292
E) 1.60
A
CO (5.00 g) and CO2 (5.00 g) were placed in a 750.0 mL container at
50.0 °C. The partial pressure of CO in the container was ________
atm.
A) 6.29
B) 4.02
C) 10.3
D) 0.292
E) 1.60
A
CO (5.00 g) and CO2 (5.00 g) were placed in a 750.0 mL container at
50.0 °C. The partial pressure of CO2 in the container was ________
atm.
A) 4.02
B) 10.3
C) 1.60
D) 0.292
E) 6.31
A
The root-mean-square speed of CO at 113 °C is ________ m/s.
A)
317
B) 58.3
C) 586
D) 993
E) 31.5
C
A sample of H2 gas (5.0 mol) effused through a pinhole in 10.5 s. It
will take ________ s for the same amount of O2 to effuse under the
same conditions.
A) 5.25
B) 42.0
C) 2.63
D)
168
E) 21.0
B
A sample of CO2 gas (3.0 mol) effused through a pinhole in 18.0 s. It
will take ________ s for the same amount of H2 to effuse under the
same conditions.
A) 396
B) 3.84
C) 0.818
D)
84.4
E) 1.92
B
A sample of He gas (2.0 mmol) effused through a pinhole in 53 s. The
same amount of an unknown gas, under the same conditions, effused
through the pinhole in 248 s. The molecular mass of the unknown gas is
________ g/mol.
A) 0.19
B) 5.5
C) 88
D) 19
E) 350
C
Using the van der Waals equation, the pressure in a 22.4 L vessel
containing 1.00 mol of neon gas at 100.0 °C is ________ atm. (a =
0.211 L2-atm/mol2, b = 0.0171 L/mol)
A) 0.730
B)
1.00
C) 1.21
D) 1.37
E) 0.367
D
Using the van der Waals equation, the pressure in a 22.4 L vessel
containing 1.50 mol of chlorine gas at 0.00 °C is ________ atm. (a =
6.49 L2-atm/mol2, b = 0.0562 L/mol)
A) 0.993
B) 1.50
C)
0.676
D) 1.91
E) 1.48
E
Molecular compounds of low molecular weight tend to be gases at room
temperature. Which of the following is most likely not a gas at room
temperature?
A) KBr
B) F2
C) HCN
D) SO2
E) CH4
A
A pressure of 0.500 atm is the same as a pressure of ________ of mm
Hg.
A) 193
B) 760.
C) 380.
D) 29.9
E) 33.0
C
A closed-end manometer was attached to a vessel containing argon. The
difference in the mercury levels in the two arms of the manometer was
9.60 cm. Atmospheric pressure was 783 mm Hg. The pressure of the argon
in the container was ________ mm Hg.
A) 96.0
B) 661
C)
773
D) 793
E) 882
A
A fixed amount of gas at 25.0 °C occupies a volume of 10.0 L when the
pressure is 751 torr. Use Boyle's law to calculate the pressure (torr)
when the volume is reduced to 7.25 L at a constant temperature of 25.0
°C.
A) 1.04 × 103
B) 0.097
C) 5.44 × 104
D) 544
E) 1.36
A
A fixed amount of gas at 25.0 °C occupies a volume of 8.66 L when the
pressure is 629 torr. Use Charles's law to calculate the volume (L)
the gas will occupy when the temperature is increased to while
maintaining the pressure at 629 torr.
A) 9.26
B)
11.2
C) 1.93
D) 6.70
E) 38.8
B
A sample of gas (24.2 g) initially at 4.00 atm was compressed from
8.00 L to 2.00 L at constant temperature. After the compression, the
gas pressure was ________ atm.
A) 4.00
B) 2.00
C)
1.00
D) 8.00
E) 16.0
E
A balloon originally had a volume of 4.39 L at 44 °C and a pressure
of 729 torr. The balloon must be cooled to ________ °C to reduce its
volume to 3.99 L (at constant pressure).
A) 0.00
B)
15.1
C) -288
D) 288
E) 546
B
If 3.21 mol of a gas occupies 56.2 L at 44 °C and 793 torr, 4.73 mol
of this gas occupies ________ L under these conditions.
A)
14.7
B) 61.7
C) 30.9
D) 82.8
E) 478
D
A gas originally at 27 °C and 1.00 atm pressure in a 3.3 L flask is
cooled at constant pressure until the temperature is 11 °C. The new
volume of the gas is ________ L.
A) 0.27
B) 3.1
C)
3.3
D) 3.5
E) 0.24
B
A sample of He gas (2.35 mol) occupies 57.9 L at 300.0 K and 1.00
atm. The volume of this sample is ________ L at 469 K and 1.00
atm.
A) 0.709
B) 41.1
C) 90.5
D) 1.41
E) 57.9
C
A sample of an ideal gas (3.00 L) in a closed container at 25.0 °C
and 76.0 torr is heated to 270 °C. The pressure of the gas at this
temperature is ________ torr.
A) 912
B) 138
C)
76.5
D) 39.5
E) 0.0253
B
A helium balloon is filled to a volume of 27.7 L at 300 K. What will
the volume of the balloon become if the balloon is heated to raise the
temperature to 392 K?
A) 36.2
B) 21.2
C) 0.0276
D)
0.0472
E) 3,260,000
A
At a temperature of ________ °C, 0.444 mol of CO gas occupies 11.8 L
at 839 torr.
A) 379
B) 73.0
C) 14.0
D)
32.0
E) 83.5
E
The pressure exerted by 1.0 mol of gas in a 13 L flask at 22 °C is
________ kPa.
A) 560
B) 190
C) 18
D) 2.4
E) 1.0
B
A 0.325 L flask filled with gas at 0.851 atm and 19 °C contains
________ mol of gas.
A) 0.0116
B) 0.0148
C)
9.42
D) 12.4
E) 80.7
A
A sample of gas (1.9 mol) is in a flask at 21 °C and 697 mm Hg. The
flask is opened and more gas is added to the flask. The new pressure
is 841 mm Hg and the temperature is now 26 °C. There are now ________
mol of gas in the flask.
A) 1.6
B) 2.3
C) 2.9
D)
3.5
E) 0.28
B
A sample of gas (1.3 mol) occupies ________ L at 22 °C and 4.0
atm.
A) 0.079
B) 0.59
C) 7.9
D) 13
E) 0.032
C
The volume of 0.15 mol of an ideal gas at 365 torr and 97 °C is
________ L.
A) 0.054
B) 2.5
C) 11
D) 9.5
E) 0.024
D
The volume occupied by 1.5 mol of gas at 35 °C and 2.0 atm pressure
is ________ L.
A) 38
B) 19
C) 2.2
D) 0.053
E) 0.026
B
How many moles of gas are there in a 36.3 L container at 25.2 °C and
570.3 mm Hg?
A) 1.11
B) 13.2
C) 0.863
D)
0.0110
E) 16,700
A
A gas in a 57.1 L pressure container at 24.9 °C and 889.8 mm Hg
contains ________ moles.
A) 32.7
B) 2080
C)
0.366
D) 0.0270
E) 2.73
E
The volume of 1.26 mol of gas at 69.4 kPa and 25.30 °C is ________
L.
A) 45.1
B) 633
C) 3.82
D) 0.445
E) 0.439
A
The density of HCN is ________ g/L at STP.
A) 1.21
B)
329
C) 605
D) 0.829
E) 2.21
A
The density of nitric oxide (NO) gas at 0.970 atm and 34.9 °C is
________ g/L.
A) 0.0384
B) 0.869
C) 1.15
D)
0.339
E) 10.2
C
The density of krypton gas at 0.866 atm and 44.4 °C is ________
g/L.
A) 0.0332
B) 0.359
C) 2.78
D) 5.57
E) 0.238
C
What is the density of chlorine gas (g/L) at 1.01 atm and 54.1
°C?
A) 2.67
B) 0.0376
C) 0.375
D) 0.228
E) 1.33
A
At STP, the density of ________ is 1.70 g/L.
A) NO
B)
Ne
C) CH4
D) F2
E) CO
D
What is the density of nitrogen dioxide gas (g/L) contained in a 4.77
L pressure tank at 738.4 mm Hg and 45.0 °C?
A) 1.71
B)
1300
C) 12.1
D) 0.0169
E) 8.17
A
What is the density of carbon dioxide gas (g/L) at 1106.2 mm Hg and
56.3 °C?
A) 13.9
B) 0.0234
C) 2.37
D) 1800
E) 0.0538
C
At at 315 K and 1.16 atm, the density of ________ is 0.906
g/L.
A) Ar
B) Kr
C) Xe
D) Ne
E) He
D
At standard pressure and 373 K, a 0.333-g sample of an unknown pure
gas occupies a volume of 0.255 L. What is the identity of the
gas?
A) xenon
B) argon
C) neon
D) helium
E) krypton
B
Calcium hydride ( ) reacts with water to form hydrogen gas:
(s) + O (l) → (aq) + (g)
How many grams of are needed to generate 48.0 L of gas at a pressure
of 0.995 atm and a temperature of 32 °C?
A) 56.8
B)
0.954
C) 161
D) 40.1
E) 80.3
D
How many liters of CO2 are formed during the combustion of 120.0 grams of ethane at STP in the open atmosphere?
179
How many liters of O2 are consumed during the combustion of 60.0 grams of ethane at STP in the open atmosphere?
157
A pressure vessel contains CO2 (PCO2 = 3.78 atm) and O2 (PO2 = 6 atm)
gases at a total pressure of 9.78 atm. What is the mole-fraction of
CO2 and O2 gases, respectively?
A) 37, 58.7
B) 0.61,
0.39
C) 2.59, 1.63
D) 0.39, 0.61
E) 0.63, 1.59
D
34.9 grams of hydrogen gas and 17.7 grams of methane gas are combined
in a reaction vessel with a total pressure at 2.92 atm. What is the
partial pressure (atm) of hydrogen gas?
A) 0.175
B)
50.5
C) 53.8
D) 2.75
E) 5.53
D
10.0 grams of argon and 20.0 grams of neon are placed in a 1216.1 ml
container at 25.1 °C. The partial pressure of neon is ________
atm.
A) 19.9
B) 5.03
C) 1.68
D) 20.0
E) 49.1
A
0.7515 moles of nitrogen gas and 0.1135 moles of methane gas are
placed in a 171.6 ml container at 20.8 °C. What is the partial
pressure (atm) of methane gas?
A) 1.14
B) 0.473
C)
16.0
D) 106
E) 226
C
A mixture of Xe, Kr, and Ar has a total pressure of 6.69 atm. What is
the mole fraction of Kr if the partial pressures of Xe and Ar are 1.92
atm and 2.75 atm, respectively.
A) 0.302
B) 0.287
C)
0.411
D) 0.259
E) 0.698
A
Zinc reacts with aqueous sulfuric acid to form hydrogen gas:
Zn (s) + H2SO4(aq) → ZnSO4 (aq) + H2(g)
In an experiment, 201 mL of wet is collected over water at 27 °C and
a barometric pressure of 765 torr. How many grams of Zn have been
consumed? The vapor pressure of water at 27 °C is 26.74 torr.
A)
4.38 ×106
B) 0.519
C) 519
D) 394
E) 3.94 × 105
B
Zinc reacts with aqueous sulfuric acid to form hydrogen gas:
Zn (s) + H2SO4(aq) → ZnSO4 (aq) + H2(g)
In an experiment, 177 mL of wet is collected over water at 27 °C and
a barometric pressure of 766 torr. The vapor pressure of water at 27
°C is 26.74 torr. The partial pressure of hydrogen in this experiment
is ________ atm.
A) 0.972
B) 739
C) 1.01
D)
793
E) 1.04
A
What is the rms speed (m/s) of N2 at 33.5 °C?
A) 173
B)
51.9
C) 302
D) 522
E) 16.5
D
What is the rms speed (m/s) of NO2 at 28.4 °C?
A) 40.2
B)
124
C) 233
D) 12.8
E) 404
E
At 500 °C, which of the following gases will have the greatest
root-mean-square speed?
A) N2O
B) UF6
C) O2
D)
NH3
E) None. The molecules of all gases have the same
root-mean-square speed at any given temperature.
D
The temperature and pressure specified by STP are ________ °C and ________ atm.
0, 1
How many molecules are there in 4.00 L of oxygen gas at 500 °C and 50.0 torr?
2.50 × 1021
What volume (L) of HCl gas is required to react with an excess of sodium to generate 19.5 L of hydrogen gas at 1.31 atm and 51.0 °C?
39.0
Calculate the density of hydrogen gas (in g/L) at 43.0 °C and 700.0 torr.
0.0717
A 5.50 L vessel contains 0.348 mol of methane, 0.311 mol of propane, and 0.445 mol of neon at a total pressure of 882 mm Hg. What is the partial pressure (in mm Hg) of methane?
278
What is the rms speed (m/s) of oxygen molecules at 36.0 °C?
491
The deviation from ideal behavior of a gas is most evident at ________ and/or low temperature.
high pressure
The ________ corrects the ideal gas law for the finite volume and attractive forces of gas molecules.
van der Waals equation
The main component of air is oxygen.
false
If the temperature is lowered from 60 °C to 30 °C, the volume of a fixed amount of gas will be one half the original volume.
false
A gas is considered "ideal" if one mole of it in a one-liter container exerts a pressure of exactly 1 atm at room temperature.
false
Kinetic-molecular theory assumes that attractive and repulsive forces between gas particles are stronger than those between gas particles and container walls.
false
According to the kinetic-molecular theory, molecules of different gases at the same temperature always have the same average kinetic energy.
true
The effusion rate of a gas is proportional to the square root of its molar mass.
false
Two deviations of real gases from ideal gases which are treated in the van der Waals equation are finite molecular volume and non-zero molecular attractions.
true