Measurements
meter > millimeter (1/1000) > micrometer (um)(1/1,000,000) > nanometer (nm) (1/1,000,000,000)
1 millimeter is equal to 1,000 micrometers.
nanometer is what is used to measure most viruses
Temp Classification and limitations
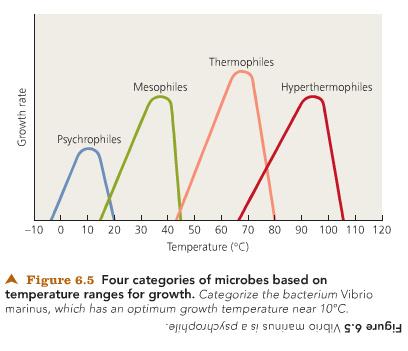
At temperatures higher than the maximum growth temperature for an organism, hydrogen bonds are broken, proteins are denatured, and membranes become too fluid.
microbes described in terms of temperature requirements, (from coldest to warmest) psychrophiles (below 20 C), mesophiles (humans- below 40 C), thermophiles (45C-75C), hyperthermophiles (80C-to about 130C recorded)
the well-defined upper and lower temperature limit within which it each species of Prokaryotes grows; Prokaryotes are divided into five groups based on their optimum growth temperatures. (thermo stability appears to be due to protein structure)
Types of microscopy and differences
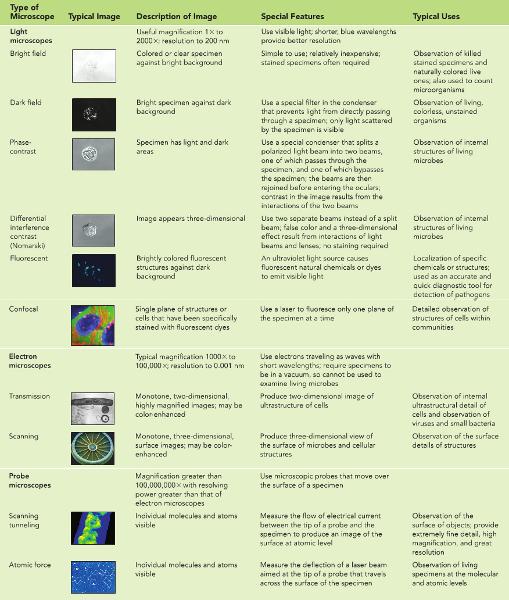
Confocal, phase-contrast, and dark-field microscopes are all types of light microscopes. As such, they can magnify only up to about 2,000×.
scanning electron micrographs typically appear two-dimensional.
Unlike transmission electron microscopy, in SEM specimens do not have to be thinly sliced before they can be visualized.
Taxonomy classification
Linnaeus system is the bases though it has been updated. Domain > Kingdom >Phylum > Class > Order > Family > Genus > Species (can mate)
Woese 3 Domains- Eukarya, Bacteria, Archaea
'Whittaker's 5 Kingdom taxonomic Scheme' - Plantae, Fungi, Animalia, Protista, Prokaryotae
Linnaeus
Swedish botanist, based on what they had in common. Only used two 2 kingdoms at the time, Plante and Animals
Fermentation
what happens when there is no oxygen to metabolize. Oxidizes NADH and can make either Lactic Acid (humans) or ethanol alcohol - keeps glycolosis going but does not make as much ATP
It is an alternative way to return electron carriers to their oxidized state.
Pyruvic Acid takes the electrons from NADH, oxidizing it back into NAD+.
the NAD+ newly regenerated by fermentation returns to glycolysis to pick up more electrons.
What is the intermediate product formed by pyruvic acid during alcoholic fermentation? Acetaldehyde
It regenerates NAD+ for glycolysis.
Glucose is partially oxidized during fermentation.
Culturing organisms
special culture techniques: animal and cell cultures, low oxygen (in a special jar), enrichment culture where it uses special things to help grow certain types.
also uses transport media to move around, Anaerobic Media that uses reducing media to actually remove oxygen from the jar
can be grown in liquid called broths, or on surface media.
Defined Media: compisition is known and exact for type of bacteria to grow- for fstidious organisioms that require an exact type of nutrition to grow
Selective: designed to help one type grow and inhibit another
Complex: more defined than defined- like MacConkey Agar, for when nutritional needs are not known. has good variety of nutrition options for the bacteria.
Differential: will grow both but the media changes to be able to tell them apart. like blood agar
chart in lab book is helpful
chlorophyll
pigment molecule that captures light energy for use in photosynthesis
has hydrocarbon tail attached to a light-absorbing 'active site'
Gram stain procedure
can be used to distinguish bacteria based on the structure of their cell walls. it detects peptidoglycan, which is present in a thick layer in Gram positive bacteria.[1] A Gram positive results in a purple/blue color while a Gram negative results in a pink/red color.
There are four basic steps of the Gram stain:
Applying a primary stain (crystal violet) to a heat-fixed smear of a bacterial culture. Heat fixing kills some bacteria but is mostly used to affix the bacteria to the slide so that they don't rinse out during the staining procedure.
The addition of a mordant, which binds to crystal violet and traps it in the cell (Gram's iodine)
Rapid decolorization with alcohol or acetone, and
Counterstaining with safranin.[9] Carbol fuchsin is sometimes substituted for safranin since it will more intensely stain anaerobic bacteria but it is much less commonly employed as a counterstain.
amination
Reaction involving the addition of an amine group to a metabolite to make an amino acid
Deamination
Process in which amine groups are split from amino acids
smear preparation
attaching the organism to a microscope slide.
thin film on the plate, then fixed to the slide with either heat or in come cases chemicals like methyl alcohol or formalin are applied to it for one minute
desiccation (drying) and fixation kill the microorganisms and generally preserve the shape and size
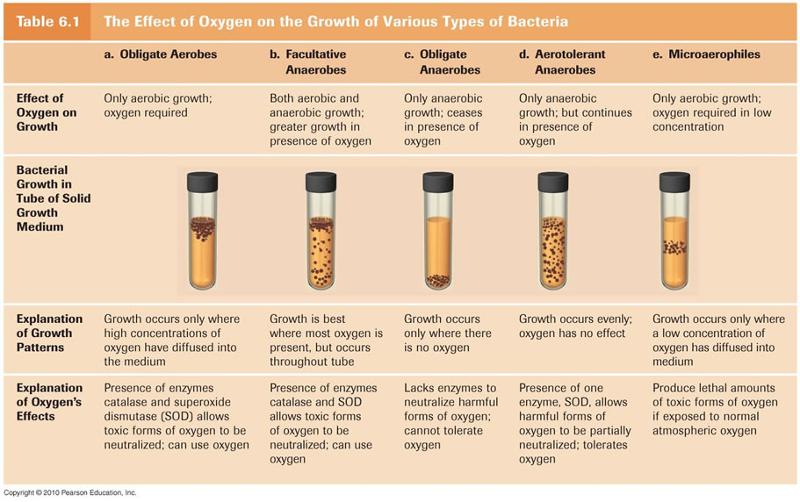
oxygen tolerance
On the basis of oxygen requirements, organisms are classified as obligate aerobes, facultative anaerobes, obligate anaerobes, aerotolerant anaerobes, and microaerophiles.
Facultative anaerobes can grow in aerobic or anaerobic conditions although tend to grow better in aerobic conditions.
Includes E. coli and some yeasts.
Aerotolerant anaerobes don’t use O2 but tolerate it – have SOD or some similar enzyme.
Many ferment carbohydrate to lactate, which inhibits growth of aerobes.
Microaerophilic aerobes require low O2 concentrations; they are sensitive to oxygen-derived free radicals.
decarboxylation
removal of CO2, requires a coenzyme derived from the vitamin thiamine.
Used in the Synthesis of Acetyl-CoA (for krebs cycle) after glycolosis and right before entering the krebs cycle (page 137)
a chemical reaction that removes a carboxyl group and releases carbon dioxide (CO2).
The reverse process, which is the first chemical step in photosynthesis, is called carboxylation, the addition of CO2 to a compound. Enzymes that catalyze decarboxylations are called decarboxylases or, the more formal term, carboxy-lyases
oxidation reactions
happens at the same time as reduction reactions- oxidation reactions involve the loss of a electron (usually donating to a oxygen atom)
can be oxidized in three ways- losing a hydrogen atom, losing a simple electron, or gaining a oxygen atom
used in Krebs Cycle and ETC.
Biological oxidations are often called dehydrogenation reactions because they involve the loss of a hydrogen atom.
reduction reactions
opposite of oxidation reaction, involves gaining of one electron
the gain in electrons reduces the overall electrical charge (hence the name reduction)
phosphorylation
To add a phosphate group to (an organic molecule).
Production of ATP by transferring phosphates directly from metabolic products to ADP (from homework answers)
the addition of a phosphate group to a protein or other organic molecule. Phosphorylation turns many protein enzymes on and off, thereby altering their function and activity.
ATP, the "high-energy" exchange medium in the cell, is synthesized in the mitochondrion by addition of a third phosphate group to ADP in a process referred to as oxidative phosphorylation. ATP is also synthesized by substrate-level phosphorylation during glycolysis. ATP is synthesized at the expense of solar energy by photophosphorylation in the chloroplasts of plant cells.
structure and function of enzymes
Enzymes are substrate specific which means that it has a specific shape that allows only the wanted substrate to bond and react with it. Because of its structure and shape it can carry out its function in speeding up the reaction by lowering the activation energy needed for the reaction to take place
They can be used to catalyze a chemical reaction over and over again.
They form a temporary intermediate compound with a substrate.
They are usually, but not always, proteins.
They can be denatured if the pH of their environment is too high or too low.
acid fast stain
differential stain that stains the cells of the genera Myobacterium and Nocardia that cause many human diseases.
Cells of these bacteria have large amounts of waxy lipids in the call walls so they don't stain well with the gram stain.
pink tells you it is positive for the acid-fast cells, blue cells are not positive.
capsule stain
also called a negative stain. Eosin and nigrosin are examples of acidic dyes used.
because you are using a acidic dye it is 'repulsed by the negative charge on the surface of cells and do not stain them- leaving a negative area around bacterial capsules. (looks like a halo around it)
endospore stain
endospores, which are dormant, highly resistant to heat, desiccation, and chemicals live inside other bacteria and can wake up to make people sick.
the Schaeffer-Fulton endospore stain uses- malachite green stain enters an endospore with heat then colled and decolored with water and counterstained with safranin.
Vegetative cells are pink, endospores are green.
microscope lens
three types: eyepiece lens, a condenser lens, and an objective lens.
Thermoduric mesophiles
mesophiles that can survive brief periods of higher temps. inadequate heating during pasteurization or canning can result in food spoilage by these.
chemoheterotroph
An organism deriving energy by ingesting intermediates or building blocks that it is incapable of creating on its own.
Most chemoheterotrophs obtain energy by ingesting organic molecules like glucose.
are unable to fix carbon and form their own organic compounds. Chemoheterotrophs can be chemolithoheterotrophs, utilizing inorganic energy sources such as sulfur or chemoorganoheterotrophs, utilizing organic energy sources such as carbohydrates, lipids, and proteins
photoheterotroph
An organism that depends on light for most of its energy and principally on organic compounds for its carbon.
'Photoheterotrophs': Bacteria that use sunlight as their energy source and organic compounds from the environment as their carbon source.
chemoautotroph
An organism (typically a bacterium or a protozoan) that obtains energy through chemical process, which is by the oxidation of electron donating molecules from the environment, rather than by photosynthesis.
in addition to deriving energy from chemical reactions, synthesize all necessary organic compounds from carbon dioxide. Chemoautotrophs use inorganic energy sources, such as hydrogen sulfide, elemental sulfur, ferrous iron, molecular hydrogen, and ammonia. Most are bacteria or archaea that live in hostile environments such as deep sea vents and are the primary producers in such ecosystems.
lithoautotroph
A microbe that takes energy from reduced compounds of minerals.
(need to find more info on this)
Photoautotroph
Photoautotrophs: Bacteria that use sunlight as their energy source and carbon dioxide (as part of photosynthesis) as their carbon source. These bacteria thus obtain all their nourishment through photosynthesis
glycolysis
produces energy in the form of NADH and ATP
exergonic pathway (gives energy)
more to be added...
Krebs cycle
more to be added- below info from the quiz and homework:
Oxaloacetic acid is regenerated
NAD+ and FAD are reduced as a result.
It is also known as the tricarboxylic acid cycle.
The ATP generated is initially GTP. (functions as a energy carrier)
the bridge step is Decarboxylation of pyruvic acid
4 electron carriers are reduced in the Krebs cycle only
metabolites during respiration are oxidized completely to carbon dioxide and water.
Entner-Doudoroff pathway
is an alternate series of reactions that catabolize glucose to pyruvate.
using a set of enzymes different from those used in either glycolysis or the pentose phosphate pathway. Most bacteria use glycolysis and the pentose phosphate pathway.
Has a net yield of 1 ATP for every glucose molecule processed, as well as 1 NADH and 1 NADPH.
Anabolism
The reactions involved in producing larger compounds from smaller compounds (endergonic- require more energy than they release)
example, The use of amino acids to make proteins / synthesis of lipids for cell membranes from glycerol and fatty acids.
another example is The Pentose Phosphate Pathway
the reactions that occur between glucose and pyruvic acid can either be anabolic or catabolic.
Catabolism
break large molecules into smaller ones and release energy (exergonic- give energy)
example: breakdown of lipids into glycerol and fatty acids.
another example is oxidative phosphorylation
Where does the energy required for anabolic reactions come from? (Catabolic reactions)
the reactions that occur between glucose and pyruvic acid can either be anabolic or catabolic.
Woese
sequenced nukleotides of smaller RNA to help classify bacteria and other cells.
proposed eukaria, bacteria, and archaea domains
toxic forms of oxygen
(Hydrogen peroxide:) is a by-product of aerobic respiration and can severely damage proteins.
(Singlet oxygen:) Extremely reactive form of oxygen, present in phagocytic cells. damage other molecules
(superoxide free radicals) :damage other molecules
Toxic forms of oxygen are produced in aerobic environments during photochemistry and is a result of aerobic respiration. Hydrogen peroxide is a by-product of aerobic respiration and can severely damage proteins. Singlet oxygen and anion superoxide are also forms of toxic oxygen and damage other molecules.
superoxide dismutase (SOD)
Catalase: Breaks hydrogen peroxide into water and O 2 Common. Produced by humans, as well as many bacteria.
Calvin-Benson cycle
Sunlight is not directly needed to carry out this process. (light independent reactions of photosynthesis)
It produces glyceraldehyde-3-phosphate by fixation of CO2 molecules.
anabolism beings in photosynthetic organisms with carbon fixation by the enzymes of this process.
the key product formed by the Calvin-Benson cycle is G3P- enzymes use G3P as the starting point for synthesizing sugars, complex polysaccharides such as starch, cellulose for cell walls in alge, and peptidoglycan for cell walls of bacteria.
ONE of the six G3P’s produced in one cycle are used to make one molecule of glucose.
Net Loss of 9 ATP after one cycle
Beta-oxidation
a catabolic process in which the enzymes split pairs of hydrogenated carbon atoms from a fatty acid and join them to coenzyme A to form acetyl-CoA
numerical aperture
ability of the lense to gather light
resolution distance is dependent on the wavelegnth of the radiation and the numerical aperture
Acidic dyes
Acidic dyes are negatively-charged dyes. Since bacteria are also negatively-charged, they will repel the acidic dyes. So, instead of staining the bacterium itself, it will be the background that will be colorized
Magnification
refraction of radiation
the apparent increase in the size of an object. indicated by a number and x - results when a beam of radiation refracts as it passes through a lense.
the degree a image is enlarged depends on the thickness of the lense, curvature, and speed of light through its substance
useful magnification is determined by the resolution and the contrast
immersion oil
used on lens of 100x - increases magnification and resolution by capturing the light that is normally refracted by air, increasing the numerical aperture
resolution
this and contrast help determine the useful magnification of a microscope.
AKA resolving power- the ability to distinguish between objects that are close together.
Leeuwenhoek's microscopes had power of 1um, so he could see things if they were more than about 1 micrometer apart. Objects closer than that appeared as only one object.
resolution distance is dependent on the wavelength radiation and the numerical aperture (ability of lens to gather light)
carrier molecules of ETC
they are membrane bound, in a series, and pass electrons from one to another and ultimately to a final electron acceptor molecule(like a fire brigade passing buckets to the last person who throw it in a fire)
the electrons energy is used to pump it across
four categories: flavoproteins (e.g. FAD), cytochromes, iron-sulphur proteins (e.g. ferredoxin), and ubiquinone.
Carrier molecules are usually proteins bound to a nonprotein group; they can undergo oxidation and reduction relatively easily, thus allowing electrons to flow through the system.
direct methods for measuring
can do with or without incubation:
WITH Incubation: microscopic counts and electronic counting
Serial Dilution and Viable plate counts, Membrane Filtration, Most Probable Number (uses statistics),
WITHOUT Incubation: Microscopic Counts (glass slide with a grid AKA Petroff-Hausser Counting Chamber), and electronic counters(Coulter Counter and Flow Cytometry)
(indirect methods use variables like metabolic activity, dry weight, turbitidy(light filtration), and even DNA sequences)
gluconeogenesis
process by which glucose is made, primarily in the liver, from non-carbohydrate sources. The body is able to make glucose from amino acids (protein), glycerol (the backbone of triglycerides, the primary fat storage molecule), and glucose metabolism intermediaries like lactate and pyruvate.
metabolic pathway that results in the generation of glucose from non-carbohydrate carbon substrates such as pyruvate, lactate, glycerol, and glucogenic amino acids.
It is one of the two main mechanisms humans and many other animals use to keep blood glucose levels from dropping too low (hypoglycemia).
streak plate method
technique used to isolate a pure strain from a single species of microorganism, often bacteria. Samples can then be taken from the resulting
colonies has 4 quadrants, each spread from the one before it
nomenclature
says all taxa have to have Latin or latinized names, linnaean system uses 'binomial nomenclature' because it assigns two names to each- the 'specific epithet' must be in lowercase
The procedure of assigning names to the kinds and groups of organisms listed in a taxonomic classification: 'the rules of nomenclature in botany.'
Ribozymes
process RNA molecules in eukaryotes
in eukaryotes, they process other RNA molecules by removing sections of RNA and splicing the remaining pieces together.
similar to action of protein enzymes
Classification methods
physical characteristics, biochemical tests, serological tests, phage typing, and analysys of nucleic acids (page 117)
Complete oxidation of glucose
nets 38 ATP after glycolosis, Krebs Cycle, and the ETC
Leeuwenhoek
simple microscope
did not invent the microscope- but was a fine lense crafter - other info about him from last exam- first person to see 'beasties' etc.
Growth
talking about growth of population not of growth of size
needs the right PH, Temp, Osmolartiy, pressure, and nutrients
What are the chemical requirements for microbial growth? sources of carbon, nitrogen, oxygen, hydrogen, sulfur, phosphorous and trace elements
what are the 5 phases of the bacteria growth curve?
Answer
1. lag phase
2. exponential phase (log phase)
3. stationary phase
4. death phase
5. phase of prolonged decline
Barophile
can survive under great pressures. They live deep under the surfaces of the earth or water. There are three kinds of these microorganisms: barotolerant, barophilic, and extreme barophiles.
Halophile
extremophile organisms that can thrive if the enzyme working in the organism reaches a temperature of over 30 degrees c while in environments with very high concentrations of salt.
can be found anywhere with a concentration of salt five times greater than the salt concentration of the ocean
often involved in formation of salty foods
cellular metabolism (from quiz 2)
Enzymes are used in both catabolic and anabolic reactions.
Energy obtained from nutrients or light is stored in the bonds of ATP.
ATP is used in the formation of macromolecules.
The goal of metabolism is reproduction of the organism.
(from quiz 2)common to chemiosmosis and the light-dependent reactions of photosynthesis?
both electron transport and a proton gradient
(from quiz 2) A fastidious organism might be grown on which of the following types of media
enriched media
A microbiologist inoculates a growth medium with 100 bacterial cells/ml. If the generation time of the species is 1 hour, how long will it be before the culture contains more than 10,000 cells/ml?
7 hours
electron beams
shorter wavelength
(from quiz 2) electron microscopes
a fluorescent screen
an electron beam
a vacuum
magnetic fields
Pyruvic acid
product of both glycolysis and the Entner-Doudoroff pathway.
What is the purpose of adding salt to mannitol salt agar?
High concentrations of salt inhibit the growth of non-halophilic organisms but allow for the growth of bacteria from the genus Staphylococci.
What is the purpose of adding eosin and methylene blue to eosin-methylene blue agar?
Eosin and methylene blue are dyes that precipitate on the surface of acid-producing colonies of enteric lactose fermenters, creating a metallic green sheen.
Which observation indicates a positive test for starch hydrolysis?
A clear zone in the media surrounding the bacterial culture following the addition of iodine
selective media
incorporate chemical substances that inhibit the growth of one type of bacteria while permitting the growth of another.
Crystal violet agar: inhibits the growth of gram-positive organisms, used to isolate gram-negative organisms
Phenylethyl alcohol agar: inhibits the growth of gram-negative organisms, used to isolate gram-positive organisms
NaCl (sodium chloride) agar: inhibits the growth of a non-halophilic organisms, often used to isolate Staphylococcus
You observe red colored colonies growing on MacConkey agar. What does this indicate about the organism?
The organism is able to ferment lactose.
differential media
incorporate chemical compounds that produce characteristic changes in the appearance of the bacterial colonies.
You observe a yellow zone surrounding growth on mannitol salt agar. What does this indicate about the organism?
The organism is able to ferment mannitol
hemolytic activity
Gamma hemolysis; no lysis of red blood cells
Beta hemolysis; lysis of red blood cells with complete destruction and use of hemoglobin by the organism
Alpha hemolysis; incomplete lysis of red blood cells, with reduction of hemoglobin
You inoculate an organism onto blood agar and observe a clear zone surrounding the bacterial growth. What type of hemolysis is indicated by this observation?
Beta hemolysis
example of a dye used in a simple stain?
Methylene blue
objective lens
where magnification begins in a typical brightfield microscope
Where would you expect to find electron transport chains in a prokaryote?
Along the plasma membrane
what does oxygen get reduced to at the end of the electron transport chain?
Water
what does the electron transport chain do to the concentration of hydrogen ions (protons)?
The concentration of protons is higher outside the membrane than inside.
The process of generating ATP using a proton gradient is referred to as
chemiosmosis
cellular respiration equation
1 Glucose + 6 O2 (Oxygen) ---Yields-- 6 CO2 + 6 H20 + 38 ATP
enzymes (reactions)
The shape, size, and electron configuration of the substrate can be accommodated by an enzyme's active site.
The amino acids in an enzyme can facilitate the reaction by accepting or donating electrons.
The first step of any enzymatic reaction isbinding of the substrate by the enzymes
Noncyclic photophosphorylation employs which photosystem(s)?
Both photosystem I and photosystem II
What is the fate of electrons in noncyclic photophosphorylation? they Reduce NADP+ to NADPH
What is the fate of the NADPH molecules created during noncyclic photophosphorylation? They are used in the Calvin-Benson cycle.
Kinky people can often find good sex
Kingdom
Phylum
Class
Order
Family
Genus
Species
light waves
Visible light has the longest wavelength of radiation, whereas gamma rays have the shortest wavelength.
visible > UV > Gamma
Basic Dyes
Basic dyes are attracted to the negatively charged surface of cells.
Basic dyes work best at a high pH (more basic pH).
what best describes the function of a photosystem?
It absorbs light energy and converts it to ATP and NADPH.