1) The smallest cell structure that would most likely be visible with
a standard (not super- resolution) research-grade light microscope is
a _____.
A) mitochondrion
B) microtubule
C) ribosome
D) microfilament
A
2) The advantage of light microscopy over electron microscopy is that
_____.
A) light microscopy provides for higher magnification than
electron microscopy
B) light microscopy provides for higher resolving power than electron microscopy
C) light microscopy allows one to view dynamic processes in living
cells
D) light microscopy provides higher contrast than electron microscopy
C
3) In the fractionation of homogenized cells using centrifugation,
the primary factor that determines whether a specific cellular
component ends up in the supernatant or the pellet is the
_____.
A) relative solubility of the component
B) size and
weight of the component
C) percentage of carbohydrates in the
component
D) presence or absence of lipids in the component
B
5) What technique would be most appropriate to use to observe the
movements of condensed chromosomes during cell division?
A)
standard light microscopy
B) scanning electron microscopy
C) transmission electron microscopy
A
7) All of the following are part of a prokaryotic cell EXCEPT _____.
A) a cell wall
B) a plasma membrane
C) ribosomes
D) an endoplasmic reticulum
D
9) Which of the following is a major difference between prokaryotic cells and eukaryotic cells?
A) Prokaryotes have cells while eukaryotes do not.
B)
Eukaryotic cells have more intracellular organelles than
prokaryotes.
C) Prokaryotes are not able to carry out aerobic
respiration, relying instead on anaerobic metabolism.
D) Prokaryotes are generally larger than eukaryotes.
B
11) Prokaryotes are classified as belonging to two different domains. What are the domains?
A) Bacteria and Eukarya
B) Bacteria and Archaea
C)
Archaea and Protista
D) Bacteria and Protista
B
12) Which structure is common to plant and animal cells?
A) chloroplast
B) central vacuole
C) mitochondrion
D) centriole
C
13) Which of the following is present in a prokaryotic cell?
A) mitochondrion
B) ribosome
C) chloroplast
D) ER
B
15) Which organelle or structure is absent in plant cells?
A) mitochondria
B) microtubules
C) centrosomes
D) peroxisomes
C
16) What is the function of the nuclear pore complex found in
eukaryotes?
A) It regulates the movement of proteins and RNAs
into and out of the nucleus.
B) It synthesizes the proteins
required to copy DNA and make mRNA.
C) It selectively transports
molecules out of the nucleus, but prevents all inbound molecules from
entering the nucleus.
D) It assembles ribosomes from raw
materials that are synthesized in the nucleus.
A
17) Which of the following macromolecules leaves the nucleus of a
eukaryotic cell through pores in the nuclear membrane?
A)
DNA
B) amino acids
C) mRNA
D) phospholipids
C
18) Which of the following statements correctly describes some aspect
of protein secretion from prokaryotic cells?
A) Prokaryotes
cannot secrete proteins because they lack an endomembrane
system.
B) The mechanism of protein secretion in prokaryotes is
probably the same as that in eukaryotes.
C) Proteins secreted by prokaryotes are synthesized on ribosomes bound to the cytoplasmic surface of the plasma membrane.
D) Prokaryotes cannot secrete proteins because they lack ribosomes.
C
19) Large numbers of ribosomes are present in cells that specialize
in producing which of the following molecules?
A) lipids
B) glycogen
C) proteins
D) nucleic acids
C
21) A cell with a predominance of free ribosomes is most likely _____.
A) primarily producing proteins for secretion
B) primarily
producing proteins in the cytosol
C) constructing an extensive
cell wall or extracellular matrix
D) enlarging its vacuole
B
23) A cell with an extensive area of smooth endoplasmic reticulum is specialized to _____.
A) play a role in storage
B) synthesize large quantities of
lipids
C) actively export protein molecules
D) import and export protein molecules
B
24) Which structure is NOT part of the endomembrane system?
A) nuclear envelope
B) chloroplast
C) Golgi apparatus
D) plasma membrane
B
27) Which structure is the site of the synthesis of proteins that may be exported from the cell?
A) rough ER
B) plasmodesmata
C) Golgi vesicles
D) free cytoplasmic ribosomes
A
29) The liver is involved in detoxification of many poisons and
drugs. Which of the following structures is primarily involved in this
process and, therefore, abundant in liver cells?
A) rough
ER
B) smooth ER
C) Golgi apparatus
D) nuclear envelope
B
30) Which of the following produces and modifies polysaccharides that will be secreted?
A) lysosome
B) mitochondrion
C) Golgi apparatus
D) peroxisome
C
31) What is the most likely pathway taken by a newly synthesized
protein that will be secreted by a cell?
A) ER → Golgi →
nucleus
B) Golgi → ER → lysosome
C) ER → Golgi → vesicles that fuse with plasma
membrane
D) ER → lysosomes → vesicles that fuse with plasma membrane
C
32) Asbestos is a material that was once used extensively in construction. One risk from working in a building that contains asbestos is the development of asbestosis caused by the inhalation of asbestos fibers. Cells will phagocytize asbestos, but are not able to degrade it. As a result, asbestos fibers accumulate in _____.
A) mitochondria
B) ribosomes
C) peroxisomes
D) lysosomes
D
33) Which of the following is NOT true? Both chloroplasts and mitochondria _____.
A) have their own DNA
B) have multiple membranes
C)
are part of the endomembrane system
D) are capable of reproducing themselves
C
34) Which organelle is the primary site of ATP synthesis in eukaryotic cells?
A) lysosome
B) mitochondrion
C) Golgi apparatus
D) peroxisome
B
35) Thylakoids, DNA, and ribosomes are all components found in _____.
A) chloroplasts
B) mitochondria
C) lysosomes
D) nuclei
A
36) In a plant cell, DNA may be found _____.
A) only in the
nucleus
B) only in the nucleus and chloroplasts
C) in the
nucleus, mitochondria, and chloroplasts
D) in the nucleus,
mitochondria, chloroplasts, and peroxisomes
C
37) In a liver cell detoxifying alcohol and some other poisons, the
enzymes of the peroxisome remove hydrogen from these molecules and
_____.
A) combine the hydrogen with water molecules to generate
hydrogen peroxide
B) use the hydrogen to break down hydrogen peroxide
C) transfer the hydrogen to the mitochondria
D) transfer
the hydrogen to oxygen molecules to generate hydrogen peroxide
D
38) The evolution of eukaryotic cells most likely involved
_____.
A) endosymbiosis of an aerobic bacterium in a larger host
cell—the endosymbiont evolved into mitochondria
B) anaerobic
archaea taking up residence inside a larger bacterial host cell to
escape toxic oxygen—the anaerobic bacterium evolved into
chloroplasts
C) an endosymbiotic fungal cell evolving into the
nucleus
D) acquisition of an endomembrane system and subsequent
evolution of mitochondria from a portion of the Golgi
A
39) Where are proteins produced other than on ribosomes free in the
cytosol or ribosomes attached to the ER?
A) in the extracellular
matrix
B) in the Golgi apparatus
C) in mitochondria
D) in the nucleolus
C
40) Suppose a cell has the following molecules and structures:
enzymes, DNA, ribosomes, plasma membrane, and mitochondria. It could
be a cell from _____.
A) a bacterium
B) an animal but not a plant
C) nearly any eukaryotic organism
D) a plant but not an animal
C
41) Cyanide binds with at least one molecule involved in producing
ATP. If a cell is exposed to cyanide, most of the cyanide will be
found within the _____.
A) mitochondria
B) peroxisomes
C) lysosomes
D) endoplasmic reticulum
A
42) Suppose a young boy is always tired and fatigued, suffering from
a metabolic disease. Which of the following organelles is most likely
involved in this disease?
A) lysosomes
B) Golgi apparatus
C) ribosomes
D) mitochondria
D
43) Motor proteins provide for molecular motion in cells by
interacting with what types of cellular structures?
A) membrane
proteins of the inner nuclear envelope
B) free ribosomes and
ribosomes attached to the ER
C) components of the cytoskeleton
D) cellulose fibers in
the cell wall
C
45) Vinblastine, a drug that inhibits microtubule polymerization, is
used to treat some forms of cancer. Cancer cells given vinblastine
would be unable to _____.
A) form cleavage furrows during cell
division
B) migrate by amoeboid movement
C) separate chromosomes during cell division
D) maintain the shape of the nucleus
C
46) Amoebae move by crawling over a surface (cell crawling), which
involves _____.
A) growth of actin filaments to form bulges in
the plasma membrane
B) setting up microtubule extensions that
vesicles can follow in the movement of cytoplasm
C) reinforcing the pseudopod with intermediate filaments
D)
cytoplasmic streaming
A
49) Spherocytosis is a human blood disorder associated with a
defective cytoskeletal protein in the red blood cells (RBCs). What do
you suspect is the consequence of such a defect?
A) abnormally
shaped RBCs
B) an insufficient supply of ATP in the RBCs
C) an insufficient supply of oxygen-transporting proteins in the RBCs
D) adherence of RBCs to blood vessel walls, causing plaque formation
A
50) Cytochalasin D is a drug that prevents actin polymerization. A
cell treated with cytochalasin D will still be able to _____.
A)
divide in two
B) contract muscle fibers
C) extend pseudopodia
D) move vesicles within a cell
D
51) Cells require which of the following to form cilia or flagella?
A) tubulin
B) laminin
C) actin
D) intermediate filaments
A
52) Which of the following statements about the cytoskeleton is
true?
A) The cytoskeleton of eukaryotes is a static structure
most resembling scaffolding used at construction sites.
B)
Although microtubules are common within a cell, actin filaments are
rarely found outside of the nucleus.
C) Movement of cilia and
flagella is the result of motor proteins causing microtubules to move
relative to each other.
D) Chemicals that block the assembly of
the cytoskeleton would have little effect on a cell's response to
external stimuli.
C
53) The cell walls of bacteria, fungi, and plant cells and the
extracellular matrix of animal cells are all external to the plasma
membrane. Which of the following is a characteristic common to all of
these extracellular structures?
A) They must block water and
small molecules to regulate the exchange of matter and energy with
their environment.
B) They must provide a rigid structure that maintains an
appropriate ratio of cell surface area to volume.
C) They are
constructed of polymers that are synthesized in the cytoplasm and
then transported out of the cell.
D) They are composed of a mixture of lipids and nucleotides.
C
54) A mutation that disrupts the ability of an animal cell to add
polysaccharide modifications to proteins would most likely cause
defects in its _____.
A) nuclear matrix and extracellular
matrix
B) mitochondria and Golgi apparatus
C) Golgi apparatus and extracellular matrix
D) nuclear pores and secretory vesicles
C
56) Plasmodesmata in plant cells are most similar in function to
which of the following structures in animal cells?
A)
desmosomes
B) gap junctions
C) extracellular matrix
D) tight junctions
B
57) Ions can travel directly from the cytoplasm of one animal cell to
the cytoplasm of an adjacent cell through _____.
A)
plasmodesmata
B) tight junctions
C) desmosomes
D) gap junctions
D
58) In plant cells, the middle lamella _____.
A) allows adjacent
cells to adhere to one another
B) prevents dehydration of
adjacent cells
C) maintains the plant's circulatory
system
D) allows for gas and nutrient exchange among adjacent cells
A
59) Where would you expect to find tight junctions?
A) in the
epithelium of an animal's stomach
B) between the smooth
endoplasmic reticulum and the rough endoplasmic reticulum
C) between plant cells in a woody plant
D) in the plasma
membrane of prokaryotes
A
61) Gaucher disease is the most common of lipid storage diseases in humans. It is caused by a deficiency of an enzyme necessary for lipid metabolism. This leads to a collection of fatty material in organs of the body including the spleen, liver, kidneys, lungs, brain, and bone marrow.
Using your knowledge of the structure of eukaryotic cells,
identify the statement below that best explains how internal
membranes and the organelles of cells would be involved in Gaucher
disease.
A) The mitochondria are most likely defective and do
not produce adequate amounts of ATP needed for cellular respiration.
B) The rough endoplasmic reticulum contains too many ribosomes
which results in an overproduction of the enzyme involved in
carbohydrate catalysis.
C) The lysosomes lack sufficient amounts
of enzymes necessary for the metabolism of lipids.
D) The Golgi apparatus produces vesicles with faulty membranes that leak their contents into the cytoplasm of the cell.
C
1) For a protein to be an integral membrane protein, it would have to be _____.
A) hydrophilic
B) hydrophobic
C) amphipathic, with at
least one hydrophobic region
D) exposed on only one surface of the membrane
C
2) You have a planar bilayer with equal amounts of saturated and unsaturated phospholipids. After testing the permeability of this membrane to glucose, you increase the proportion of unsaturated phospholipids in the bilayer. What will happen to the membrane's permeability to glucose?
A) Permeability to glucose will increase.
B) Permeability to
glucose will decrease.
C) Permeability to glucose will stay the
same.
D) You cannot predict the outcome. You simply have to make
the measurement.
A
3) According to the fluid mosaic model of cell membranes,
phospholipids _____.
A) can move laterally along the plane of the
membrane
B) frequently flip-flop from one side of the membrane to
the other
C) occur in an uninterrupted bilayer, with membrane
proteins restricted to the surface of the membrane
D) have hydrophilic tails in the interior of the membrane
A
4) The membranes of winter wheat are able to remain fluid when it is
extremely cold by _____. A) increasing the percentage of unsaturated
phospholipids in the membrane
B) increasing the percentage of
cholesterol molecules in the membrane
C) decreasing the number of
hydrophobic proteins in the membrane
D) cotransport of glucose and hydrogen
A
5) Some regions of the plasma membrane, called lipid rafts, have a
higher concentration of cholesterol molecules. At higher temperatures,
these regions _____.
A) are more fluid than the surrounding
membrane
B) are less fluid than the surrounding membrane
C) detach from the plasma membrane and clog arteries
D)
have higher rates of lateral diffusion of lipids and proteins into
and out of these regions
B
6) Singer and Nicolson's fluid mosaic model of the membrane proposed
that membranes_____. A) are a phospholipid bilayer between two layers
of hydrophilic proteins
B) are a single layer of phospholipids
and proteins
C) consist of protein molecules embedded in a fluid
bilayer of phospholipids
D) consist of a mosaic of polysaccharides and proteins
C
8) Which of these are NOT embedded in the hydrophobic portion of the
lipid bilayer at all? A) transmembrane proteins
B) integral
proteins
C) peripheral proteins
D) All of these are embedded in the hydrophobic portion of the lipid bilayer.
C
9) Why are lipids and proteins free to move laterally in
membranes?
A) The interior of the membrane is filled with liquid
water.
B) Lipids and proteins repulse each other in the
membrane.
C) Hydrophilic portions of the lipids are in the
interior of the membrane.
D) There are only weak hydrophobic
interactions in the interior of the membrane.
D
15) Cell membranes are asymmetrical. Which of the following
statements is the most likely explanation for the membrane's
asymmetrical nature?
A) Since the cell membrane forms a border
between one cell and another in tightly packed tissues such as
epithelium, the membrane must be asymmetrical
B) Since cell membranes communicate signals from one organism
to another, the cell membranes must be asymmetrical.
C) The two
sides of a cell membrane face different environments and carry out
different functions.
D) Proteins only function on the cytoplasmic side of the cell membrane, which results in the membrane's asymmetrical nature.
C
16) In what way do the membranes of a eukaryotic cell vary?
A)
Phospholipids are found only in certain membranes.
B) Certain
proteins are unique to each membrane.
C) Only certain membranes
of the cell are selectively permeable.
D) Some membranes have
hydrophobic surfaces exposed to the cytoplasm, while others have
hydrophilic surfaces facing the cytoplasm.
B
17) Which of the following is a reasonable explanation for why
unsaturated fatty acids help keep a membrane more fluid at lower
temperatures?
A) The double bonds form kinks in the fatty acid
tails, preventing adjacent lipids from packing tightly.
B) Unsaturated fatty acids have a higher cholesterol content
and, therefore, more cholesterol in membranes.
C) Unsaturated
fatty acids are more polar than saturated fatty acids.
D) The
double bonds block interaction among the hydrophilic head groups of
the lipids
A
18) What kinds of molecules pass through a cell membrane most easily?
A) large and hydrophobic
B) small and hydrophobic
C)
large polar
D) ionic
B
19) Which of the following most accurately describes selective
permeability?
A) An input of energy is required for
transport.
B) Lipid-soluble molecules pass through a
membrane.
C) There must be a concentration gradient for molecules
to pass through a membrane.
D) Only certain molecules can cross a cell membrane.
D
20) Which of the following is a characteristic feature of a carrier protein in a plasma membrane?
A) It exhibits a specificity for a particular type of
molecule.
B) It requires the expenditure of cellular energy to
function.
C) It works against diffusion.
D) It has no hydrophobic regions.
A
21) Which of the following would likely move through the lipid
bilayer of a plasma membrane most rapidly?
A) CO2
B) an amino acid
C) glucose
D) K+
A
22) Which of the following allows water to move much faster across cell membranes?
A) the sodium-potassium pump
B) ATP
C) peripheral proteins
D) aquaporins
D
24) Diffusion _____.
A) is very rapid over long
distances
B) requires an expenditure of energy by the
cell
C) is a passive process in which molecules move from a
region of higher concentration to a region of lower
concentration
D) requires integral proteins in the cell membrane
C
25) Which of the following processes includes all others?
A) osmosis
B) facilitated diffusion
C) passive transport
D) transport of an ion down its electrochemical gradient
C
26) When a cell is in equilibrium with its environment, which of the
following occurs for substances that can diffuse through the
cell?
A) There is random movement of substances into and out of
the cell.
B) There is directed movement of substances into and
out of the cell.
C) There is no movement of substances into and out of the cell.
D) All movement of molecules is directed by active transport.
A
27) Which of the following is true of osmosis?
A) Osmosis only
takes place in red blood cells.
B) Osmosis is an energy-demanding
or "active" process.
C) In osmosis, water moves across
a membrane from areas of lower solute concentration to areas of higher
solute concentration.
D) In osmosis, solutes move across a
membrane from areas of lower water concentration to areas of higher
water concentration.
C
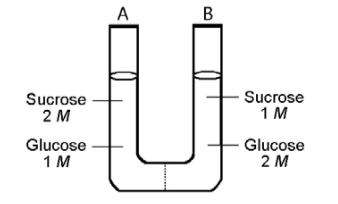
The solutions in the two arms of this U-tube are separated by a membrane that is permeable to water and glucose but not to sucrose. Side A is half-filled with a solution of 2 M sucrose and 1 M glucose. Side B is half-filled with 1 M sucrose and 2 M glucose. Initially, the liquid levels on both sides are equal.
28) Refer t o the figure. Initially, in terms of tonicity, the solution in side A with respect to the solution in side B is _____.
A) hypotonic
B) isotonic
C) saturated
D) hypertonic
B

The solutions in the two arms of this U-tube are separated by a membrane that is permeable to water and glucose but not to sucrose. Side A is half-filled with a solution of 2 M sucrose and 1 M glucose. Side B is half-filled with 1 M sucrose and 2 M glucose. Initially, the liquid levels on both sides are equal.
29) Refer to the figure. After the system reaches equilibrium, what changes are observed?
A) The molarity of sucrose is higher than that of glucose on side
A.
B) The water level is higher in side A than in side B.
C)
The water level is unchanged.
D) The water level is higher in side B than in side A.
B
30) A patient was involved a serious accident and lost a large
quantity of blood. In an attempt to replenish body fluids, distilled
water—equal to the volume of blood lost—is added to the blood directly
via one of his veins. What will be the most probable result of this
transfusion?
A) The patient's red blood cells will shrivel up
because the blood has become hypotonic compared to the cells.
B) The patient's red blood cells will swell and possibly burst
because the blood has become hypotonic compared to the cells.
C)
The patient's red blood cells will shrivel up because the blood has
become hypertonic compared to the cells.
D) The patient's red blood cells will burst because the blood has become hypertonic compared to the cells.
B
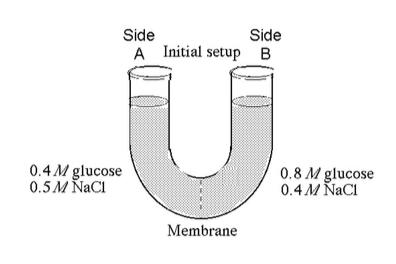
The solutions in the arms of a U-tube are separated at the bottom of the tube by a selectively permeable membrane. The membrane is permeable to sodium chloride but not to glucose. Side A is filled with a solution of 0.4 M glucose and 0.5 M sodium chloride (NaCl), and side B is filled with a solution containing 0.8 M glucose and 0.4 M sodium chloride. Initially, the volume in both arms is the same..
31) Refer to the figure. At the beginning of the experiment,
A) side A is hypertonic to side B.
B) side A is hypotonic
to side B.
C) side A is hypertonic to side B with respect to glucose.
D) side A is hypotonic to side B with respect to NaCl.
B

32) Refer to the figure. If you examine side A after three days, you
should find _____.
A) a decrease in the concentration of NaCl and
glucose and an increase in the water level
B) a decrease in the concentration of NaCl, an increase in water
level, and no change in the concentration of glucose
C) a
decrease in the concentration of NaCl and a decrease in the water
level
D) no change in the concentration of NaCl and glucose and
an increase in the water level
C
36) Celery stalks that are immersed in fresh water for several hours become stiff. Similar stalks left in a 0.15 M salt solution become limp. From this we can deduce that the fresh water_____.
A) and the salt solution are both hypertonic to the cells of the
celery stalks
B) is hypotonic and the salt solution is hypertonic
to the cells of the celery stalks
C) is hypertonic and the salt solution is hypotonic to the cells of the celery stalks
D) is isotonic and the salt solution is hypertonic to the cells of the celery stalks
B
37) What will happen to a red blood cell (RBC), which has an internal
ion concentration of about 0.9 percent, if it is placed into a beaker
of pure water?
A) The cell would shrink because the water in the
beaker is hypotonic relative to the cytoplasm of the RBC.
B) The cell would shrink because the water in the beaker is
hypertonic relative to the cytoplasm of the RBC.
C) The cell
would swell because the water in the beaker is hypotonic relative to
the cytoplasm of the RBC.
D) The cell will remain the same size because the solution outside the cell is isotonic.
C
38) Which of the following statements correctly describes the normal
tonicity conditions for typical plant and animal cells? The animal
cell is in _____.
A) a hypotonic solution, and the plant cell is
in an isotonic solution
B) an isotonic solution, and the plant
cell is in a hypertonic solution
C) a hypertonic solution, and the plant cell is in an isotonic solution
D) an isotonic solution, and the plant cell is in a hypotonic solution
D
39) In which of the following would there be the greatest need for osmoregulation?
A) an animal connective tissue cell bathed in isotonic body
fluid
B) a salmon moving from a river into an ocean
C) a red
blood cell surrounded by plasma
D) a plant being grown hydroponically in a watery mixture of designated nutrients
B
41) A sodium-potassium pump _____.
A) moves three potassium ions
out of a cell and two sodium ions into a cell while producing an ATP
for each cycle
B) move three sodium ions out of a cell and two
potassium ions into a cell while consuming an ATP for each
cycle
C) moves three potassium ions out of a cell and two sodium
ions into a cell while consuming 2 ATP in each cycle
D) move
three sodium ions out of a cell and two potassium ions into a cell and
generates an ATP in each cycle
B
43) Which of the following membrane activities requires energy from
ATP?
A) facilitated diffusion of chloride ions across the
membrane through a chloride channel
B) movement of Na+ ions from a lower concentration in a mammalian
cell to a higher concentration in the extracellular fluid
C)
movement of glucose molecules into a bacterial cell from a medium
containing a higher concentration of glucose than inside the
cell
D) movement of carbon dioxide out of a paramecium
B
44) The voltage across a membrane is called the _____.
A) chemical gradient
B) membrane potential
C) osmotic potential
D) electrochemical gradient
B
45) Ions diffuse across membranes through specific ion channels down _____.
A) their chemical gradients
B) their concentration
gradients
C) the electrical gradients
D) their electrochemical gradients
D
1) Which of the following is true of metabolism in its entirety in
all organisms? A) Metabolism depends on a constant supply of energy
from food.
B) Metabolism uses all of an organism's
resources.
C) Metabolism consists of all the energy
transformation reactions in an organism.
D) Metabolism manages the increase of entropy in an organism.
C
2) Which of the following is an example of potential rather than kinetic energy?
A) water rushing over Niagara Falls
B) light flashes emitted
by a firefly
C) a molecule of glucose
D) a crawling beetle foraging for food
C
3) Most cells cannot harness heat to perform work because _____.
A) heat is not a form of energy
B) temperature is usually
uniform throughout a cell
C) heat can never be used to do work
D) heat must remain constant during work
B
4) Which of the following involves a decrease in entropy?
A) condensation reactions
B) reactions that separate
monomers
C) depolymerization reactions
D) hydrolysis reactions
A
5) Which term most precisely describes the cellular process of
breaking down large molecules into smaller ones?
A) catabolism
(catabolic pathways)
B) metabolism
C) anabolism (anabolic pathways)
D) dehydration
A
6) Anabolic pathways _____.
A) are usually highly spontaneous
chemical reactions
B) consume energy to build up polymers from
monomers
C) release energy as they degrade polymers to
monomers
D) consume energy to decrease the entropy of the
organism and its environment
B
8) For living organisms, which of the following is an important
consequence of the first law of thermodynamics?
A) The energy
content of an organism is constant.
B) The organism ultimately
must obtain all of the necessary energy for life from its environment.
C) The entropy of an organism decreases with time as the organism grows in complexity.
D) Organisms grow by converting energy into organic matter.
B
9) Living organisms increase in complexity as they grow, resulting in
a decrease in the entropy of an organism. How does this relate to the
second law of thermodynamics?
A) Living organisms do not obey the
second law of thermodynamics, which states that entropy must increase
with time.
B) Life obeys the second law of thermodynamics because the decrease
in entropy as the organism grows is exactly balanced by an increase in
the entropy of the universe.
C) As a consequence of growing,
organisms cause a greater increase in entropy in their environment
than the decrease in entropy associated with their growth.
D) Living organisms are able to transform energy into entropy.
C
10) Which of the following statements is a logical consequence of the
second law of thermodynamics?
A) If the entropy of a system
increases, there must be a corresponding decrease in the entropy of
the universe.
B) If there is an increase in the energy of a
system, there must be a corresponding decrease in the energy of the
rest of the universe.
C) Every chemical reaction must increase
the total entropy of the universe.
D) Energy can be transferred
or transformed, but it cannot be created or destroyed.
C
12) Which of the following types of reactions would decrease the entropy within a cell?
A) anabolic reactions
B) hydrolysis
C) digestion
D) catabolic reactions
A
15) A system at chemical equilibrium _____. A) consumes energy at a
steady rate
B) releases energy at a steady rate
C) has zero
kinetic energy
D) can do no work
D
16) Which of the following is true for all exergonic
reactions?
A) The products have more total energy than the
reactants.
B) The reaction proceeds with a net release of free
energy.
C) The reaction goes only in a forward direction: all
reactants will be converted to products, but no products will be
converted to reactants.
D) A net input of energy from the surroundings is required for the reactions to proceed.
B
19) Choose the pair of terms that correctly completes this sentence:
Catabolism is to anabolism as _____ is to _____.
A) exergonic;
spontaneous
B) exergonic; endergonic
C) free energy; entropy
D) work; energy
B
21) Why is ATP an important molecule in metabolism?
A) Its
hydrolysis provides an input of free energy for exergonic
reactions.
B) It provides energy coupling between exergonic and
endergonic reactions.
C) Its terminal phosphate group contains a
strong covalent bond that, when hydrolyzed, releases free
energy.
D) Its terminal phosphate bond has higher energy than the
other two phosphate bonds.
B
24) Catabolic pathways _____.
A) combine molecules into more
energy-rich molecules
B) supply energy, primarily in the form of
ATP, for the cell's work
C) are endergonic
D) are spontaneous and do not need enzyme catalysis
B
25) When chemical, transport, or mechanical work is done by an
organism, what happens to the heat generated?
A) It is used to
power yet more cellular work.
B) It is used to store energy as
more ATP.
C) It is used to generate ADP from nucleotide precursors.
D) It is lost to the environment.
D
30) Which of the following is true of enzymes?
A) Enzyme
function is increased if the 3- D structure or conformation of an
enzyme is altered.
B) Enzyme function is independent of physical and chemical
environmental factors such as pH and temperature.
C) Enzymes
increase the rate of chemical reaction by lowering activation energy
barriers.
D) Enzymes increase the rate of chemical reaction by
providing activation energy to the substrate.
C
32) The lock-and-key analogy for enzymes applies to the specificity
of enzymes _____. A) as they form their tertiary and quaternary
structure
B) binding to their substrate
C) interacting with water
D) interacting with ions
B
33) You have discovered an enzyme that can catalyze two different
chemical reactions. Which of the following is most likely to be
correct?
A) The enzyme contains α-helices and β-pleated
sheets.
B) The enzyme is subject to competitive inhibition and
allosteric regulation.
C) Two types of allosteric regulation occur: The binding of one
molecule activates the enzyme, while the binding of a different
molecule inhibits it.
D) Either the enzyme has two distinct
active sites or the reactants involved in the two reactions are very
similar in size and shape.
D
34) Reactants capable of interacting to form products in a chemical
reaction must first overcome a thermodynamic barrier known as the
reaction's _____.
A) entropy
B) activation energy
C) equilibrium point
D) free-energy content
B
36) The active site of an enzyme is the region that _____.
A) binds allosteric regulators of the enzyme
B) is involved
in the catalytic reaction of the enzyme
C) binds noncompetitive
inhibitors of the enzyme
D) is inhibited by the presence of a coenzyme or a cofactor
B
37) According to the induced fit hypothesis of enzyme catalysis,
_____.
A) the binding of the substrate depends on the shape of
the active site
B) some enzymes change their structure when
activators bind to the enzyme
C) the binding of the substrate changes the shape of the enzyme's active site
D) the active site creates a microenvironment ideal for the reaction
C
38) Increasing the substrate concentration in an enzymatic reaction
could overcome which of the following?
A) the need for a
coenzyme
B) allosteric inhibition
C) competitive inhibition
D) insufficient cofactors
C
40) A noncompetitive inhibitor decreases the rate of an enzyme reaction by _____.
A) binding at the active site of the enzyme
B) changing the
shape of the enzyme's active site
C) changing the free energy
change of the reaction
D) acting as a coenzyme for the reaction
B
Succinate dehydrogenase catalyzes the conversion of succinate to fumarate. The reaction is inhibited by malonic acid, which resembles succinate but cannot be acted upon by succinate dehydrogenase. Increasing the ratio of succinate to malonic acid reduces the inhibitory effect of malonic acid.
51) What is malonic acid's role with respect to succinate dehydrogenase? Malonic acid _____.
A) is a competitive inhibitor
B) blocks the binding of
fumarate
C) is a noncompetitive inhibitor
D) is an allosteric regulator
A
56) You have isolated a previously unstudied protein, identified its complete structure in detail, and determined that it catalyzes the breakdown of a large substrate. You notice it has two binding sites. One of these is large, apparently the bonding site for the large substrate; the other is small, possibly a binding site for a regulatory molecule. What do these findings tell you about the mechanism of this protein?
A) It is probably a structural protein that is involved in cell-to-cell adhesion.
B) It is probably an enzyme that works through allosteric
regulation.
C) It is probably an enzyme that works through
competitive inhibition.
D) It is probably a cell membrane
transport protein–like an ion channel.
B
57) Allosteric enzyme regulation is usually associated with _____.
A) feedback inhibition
B) activating activity
C) an
enzyme with more than one subunit
D) the need for cofactors
C