1) The basis of the VSEPR model of molecular bonding is ________.
A) regions of electron density on an atom will organize themselves so as to maximize s-character
B) regions of electron density in the valence shell of an atom will arrange themselves so as to maximize overlap
C) atomic orbitals of the bonding atoms must overlap for a bond to form
D) electron domains in the valence shell of an atom will arrange themselves so as to minimize repulsions
E) hybrid orbitals will form as necessary to, as closely as possible, achieve spherical symmetry
D) electron domains in the valence shell of an atom will arrange themselves so as to minimize repulsions
2) In counting the electron domains around the central atom in VSEPR theory, a ________ is not included.
A) nonbonding pair of electrons
B) single covalent bond
C) core level electron pair
D) double covalent bond
E) triple covalent bond
C) core level electron pair
3) The electron-domain geometry of ________ is tetrahedral.
A) CBr4
B) PH3
C) CCl2Br2
D) XeF4
E) all of the above except XeF4
E) all of the above except XeF4
4) Of the following species, ________ will have bond angles of 120°.
A) PH3
B) ClF3
C) NCl3
D) BCl3
E) All of these will have bond angles of 120°.
D) BCl3
5) The molecular geometry of the BrO3- ion is ________.
A) trigonal pyramidal
B) trigonal planar
C) bent
D) tetrahedral
E) T-shaped
A) trigonal pyramidal
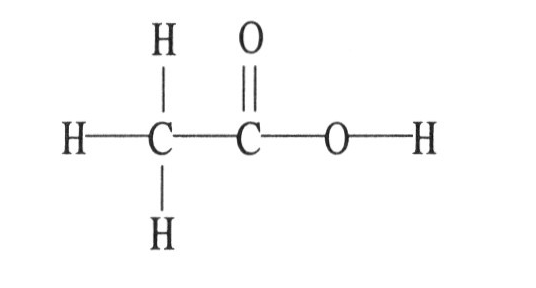
6) The molecular geometry of the left-most carbon atom in the molecule below is ________.
A) trigonal planar
B) trigonal bipyramidal
C) tetrahedral
D) octahedral
E) T-shaped
C) tetrahedral
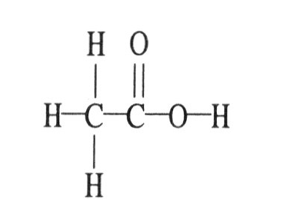
7) The molecular geometry of the right-most carbon in the molecule below is ________.
A) trigonal planar
B) trigonal bipyramidal
C) tetrahedral
D) octahedral
E) T-shaped
A) trigonal planar
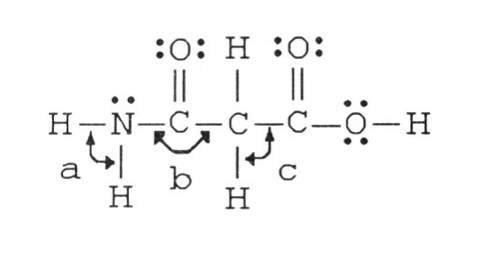
8) The bond angles marked a, b, and c in the molecule below are about ________, ________, and ________, respectively.
A) 90°, 90°, 90°
B) 120°, 120°, 90°
C) 120°, 120°, 109.5°
D) 109.5°, 120°, 109.5°
E) 109.5°, 90°, 120°
D) 109.5°, 120°, 109.5°
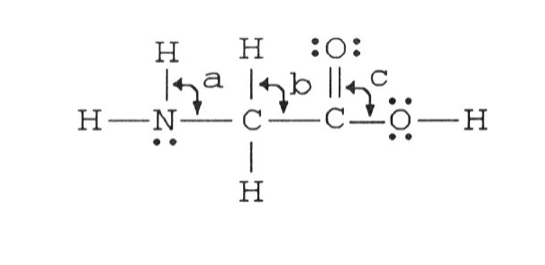
9) The bond angles marked a, b, and c in the molecule below are about ________, ________, and ________, respectively.
A) 109.5°, 109.5°, 109.5°
B) 120°, 109.5°, 120°
C) 109.5°, 109.5°, 120°
D) 90°, 180°, 90°
E) 109.5°, 109.5°, 90°
C) 109.5°, 109.5°, 120°
10) The central Xe atom in the XeF4 molecule has ________ unbonded electron pair(s) and ________ bonded electron pair(s) in its valence shell.
A) 1, 4
B) 2, 4
C) 4, 0
D) 4, 1
E) 4, 2
B) 2, 4
11) An electron domain consists of ________.
a) a nonbonding pair of electrons
b) a single bond
c) a multiple bond
A) a only
B) b only
C) c only
D) a, b, and c
E) b and c
D) a, b, and c
12) The molecular geometry consists of ________.
a) a nonbonding pair of electrons
b) a single bond
c) a multiple bond
A) a only
B) b only
C) c only
D) a, b, and c
E) b and c
E) b and c
13) The electron-domain geometry and the molecular geometry of a molecule of the general formula ABn are ________.
A) never the same
B) always the same
C) sometimes the same
D) not related
E) mirror images of one another
C) sometimes the same
14) The electron-domain geometry and the molecular geometry of a molecule of the general formula ABn will always be the same if ________.
A) there are no lone pairs on the central atom
B) there is more than one central atom
C) n is greater than four
D) n is less than four
E) the octet rule is obeyed
A) there are no lone pairs on the central atom
15) A molecule has the formula AB3 and the central atom is in a different plane from the surrounding three atoms. Its molecular shape is ________.
A) tetrahedral
B) trigonal pyramidal
C) linear
D) T-shaped
E) bent
B) trigonal pyramidal
16) PCl5 has ________ electron domains and a ________ molecular arrangement.
A) 6, trigonal bipyramidal
B) 6, tetrahedral
C) 5, square pyramidal
D) 5, trigonal bipyramidal
E) 6, seesaw
D) 5, trigonal bipyramidal
17) For molecules of the general formula ABn, n can be greater than four ________.
A) for any element A
B) only when A is an element from the third period or below the third period
C) only when A is boron or beryllium
D) only when A is carbon
E) only when A is Xe
B) only when A is an element from the third period or below the third period
Consider the following species when answering the following questions:
(i) PCl3(ii) CCl4(iii) TeCl4(iv) XeF4(v) SF6
18) For which of the molecules is the molecular geometry (shape) the same as the VSEPR electron domain arrangement (electron domain geometry)?
A) (i) and (ii)
B) (i) and (iii)
C) (ii) and (v)
D) (iv) and (v)
E) (v) only
C) (ii) and (v)
19) Of the molecules below, only ________ is polar.
A) CCl4
B) CH4
C) SeF4
D) SiCl4
C) SeF4
20) Of the molecules below, only ________ is nonpolar.
A) BF3
B) NF3
C) IF3
D) PBr3
E) BrCl3
A) BF3
21) The molecular geometry of the PF3 molecule is ________, and this molecule is ________.
A) trigonal planar, polar
B) trigonal planar, nonpolar
C) trigonal pyramidal, polar
D) trigonal pyramidal, nonpolar
E) tetrahedral, unipolar
C) trigonal pyramidal, polar
22) Of the following molecules, only ________ is polar.
A) BeCl2
B) BF3
C) CBr4
D) SiH2Cl2
E) Cl2
D) SiH2Cl2
23) For molecules with only one central atom, how many lone pairs on the central atom guarantees molecular polarity?
A) 1
B) 2
C) 1 or 2
D) 3
E) 1 or 3
A) 1
24) The molecular geometry of the CHF3 molecule is ________, and the molecule is ________.
A) trigonal pyramidal, polar
B) tetrahedral, nonpolar
C) seesaw, nonpolar
D) tetrahedral, polar
E) seesaw, polar
D) tetrahedral, polar
Consider the following species when answering the following questions:
(i) PCl3(ii) CCl4(iii) TeCl4(iv) XeF4(v) SF6
25) Which of the molecules has a see-saw shape?
A) (i)
B) (ii)
C) (iii)
D) (iv)
E) (v)
C) (iii)
Consider the following species when answering the following questions:
(i) PCl3(ii) CCl4(iii) TeCl4(iv) XeF4(v) SF6
26) In which of the molecules does the central atom utilize d orbitals to form hybrid orbitals?
A) (i) and (ii)
B) (iii) only
C) (i) and (v)
D) (iii), (iv), and (v)
E) (v) only
D) (iii), (iv), and (v)
27) The combination of two atomic orbitals results in the formation of ________ molecular orbitals.
A) 1
B) 2
C) 3
D) 4
E) 0
B) 2
28) The sp3d2 atomic hybrid orbital set accommodates ________ electron domains.
A) 2
B) 3
C) 4
D) 5
E) 6
E) 6
29) The sp2 atomic hybrid orbital set accommodates ________ electron domains.
A) 2
B) 3
C) 4
D) 5
E) 6
B) 3
30) The hybridizations of nitrogen in NF3 and NH3 are ________ and ________, respectively.
A) sp2, sp2
B) sp, sp3
C) sp3, sp
D) sp3, sp3
E) sp2, sp3
D) sp3, sp3
31) The hybridizations of iodine in IF3 and IF5 are ________ and ________, respectively.
A) sp3, sp3d
B) sp3d, sp3d2
C) sp3d, sp3
D) sp3d2, sp3d
E) sp3d2, sp3d2
B) sp3d, sp3d2
32) The hybrid orbitals used for bonding by the sulfur atom in the SF4 molecule are ________ orbitals.
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
D) sp3d
33) The hybrid orbitals used for bonding by Xe in the unstable XeF2 molecule are ________ orbitals.
A) sp2
B) sp3
C) sp3d
D) sp3d2
E) sp
C) sp3d
34) The hybridization scheme for BeF2 is ________.
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
A) sp
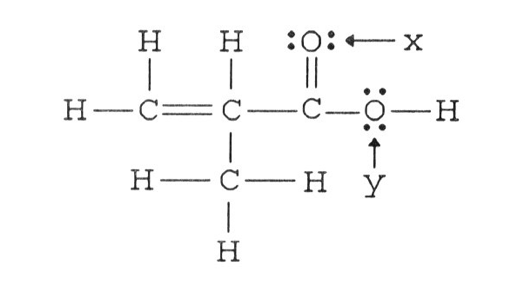
35) The hybridization of the oxygen atom labeled y in the structure below is ________. The C-O-H bond angle is ________.
A) sp, 180°
B) sp2, 109.5°
C) sp3, 109.5°
D) sp3d2, 90°
E) sp, 90°
C) sp3, 109.5°
36) The electron-domain geometry of the AsF5 molecule is trigonal bipyramidal. The hybrid orbitals used by the As atom for bonding are ________ orbitals.
A) sp2d2
B) sp3
C) sp3d2
D) sp3d
E) sp2
D) sp3d
37) ________ hybrid orbitals are used for bonding by Xe in the XeF4 molecule.
A) sp2
B) sp3
C) sp3d
D) sp3d2
E) sp
D) sp3d2
Consider the following species when answering the following questions:
(i) PCl3(ii) CCl4(iii) TeCl4(iv) XeF4(v) SF6
38) In which of the molecules is the central atom sp3d2 hybridized?
A) (i) and (ii)
B) (iii) only
C) (iii) and (iv)
D) (iv) and (v)
E) (v) only
D) (iv) and (v)
39) When four atomic orbitals are mixed to form hybrid orbitals, how many hybrid orbitals are formed?
A) one
B) six
C) three
D) four
E) five
D) four
40) A triatomic molecule cannot be linear if the hybridization of the central atoms is ________.
A) sp
B) sp2
C) sp3
D) sp2 or sp3
E) sp2d or sp3d2
D) sp2 or sp3
41) Valence bond theory does not address the issue of ________.
A) excited states of molecules
B) molecular shape
C) covalent bonding
D) hybridization
E) multiple bonds
A) excited states of molecules
42) A typical double bond ________.
A) is stronger and shorter than a single bond
B) consists of one σ bond and one π bond
C) imparts rigidity to a molecule
D) consists of two shared electron pairs
E) All of the above answers are correct.
E) All of the above answers are correct.
43) A typical triple bond ________.
A) consists of one σ bond and two π bonds
B) consists of three shared electrons
C) consists of two σ bonds and one π bond
D) consists of six shared electron pairs
E) is longer than a single bond
A) consists of one σ bond and two π bonds
44) In a polyatomic molecule, "localized" bonding electrons are associated with ________.
A) one particular atom
B) two particular atoms
C) all of the atoms in the molecule
D) all of the π bonds in the molecule
E) two or more σ bonds in the molecule
B) two particular atoms
45) There are ________ σ bonds and ________ π bonds in H3C–CH2–CH=CH–CH2–C CH.
A) 14, 2
B) 10, 3
C) 12, 2
D) 13, 2
E) 16, 3
E) 16, 3
46) Which of the following molecules or ions will exhibit delocalized bonding?
SO2 SO3 SO3 2-
A) SO2, SO3, and SO32-
B) SO32- only
C) SO2 and SO3
D) SO3 and SO32-
E) None of the above will exhibit delocalized bonding.
C) SO2 and SO3
47) Which of the following molecules or ions will exhibit delocalized bonding?
NO2- NH4+ N3-
A) NH4+ and N3-
B) NO2- only
C) NO2-, NH4+, and N3-
D) N3- only
E) NO2- and N3-
B) NO2- only
48) In order to exhibit delocalized π bonding, a molecule must have ________.
A) at least two π bonds
B) at least two resonance structures
C) at least three σ bonds
D) at least four atoms
E) trigonal planar electron domain geometry
B) at least two resonance structures
49) In a typical multiple bond, the σ bond results from overlap of ________ orbitals and the π bond(s) result from overlap of ________ orbitals.
A) hybrid, atomic
B) hybrid, hybrid
C) atomic, hybrid
D) hybrid, hybrid or atomic
E) hybrid or atomic, hybrid or atomic
A) hybrid, atomic
50) The carbon-carbon σ bond in ethylene, H2C =CH2, results from the overlap of ________.
A) sp hybrid orbitals
B) sp3 hybrid orbitals
C) sp2 hybrid orbitals
D) s atomic orbitals
E) p atomic orbitals
C) sp2 hybrid orbitals
51) The π bond in ethylene, H2C CH2, results from the overlap of ________.
A) sp3 hybrid orbitals
B) s atomic orbitals
C) sp hybrid orbitals
D) sp2 hybrid orbitals
E) p atomic orbitals
E) p atomic orbitals
52) A typical double bond consists of ________.
A) three sigma bonds
B) three pi bonds
C) one sigma and two pi bonds
D) one sigma and one pi bond
E) three ionic bonds
D) one sigma and one pi bond
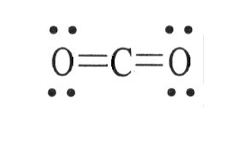
53) The Lewis structure of carbon dioxide is given below. The hybridization of the carbon atom in carbon dioxide is ________.
A) sp3
B) sp2
C) sp
D) sp2d
E) sp2d2
C) sp