BMD 330 Chapter 10 - Microbial Metabolism
What is metabolism?
All chemical reactions and physical workings of the cell
_____ requires energy input whereas ___ releases energy
Anabolism requires energy input whereas catabolsim releases energy
What is anabolism?
The process of biosynthesis (synthesis of cell molecules and structures)
What is catabolism?
Break the bonds of larger molecules
Both ___ and ___ make up the metabolism.
Both catabolism and anabolism make up the metabolism.
What is purpose of metabolism>
- Assembles smaller molecules into large macromolecules for the cell, utilizing ATP to form bonds (anabolism)
- Breaks down macromolecules into smaller molecules, a process that yields energy (catabolism)
- Collects and spends energy in the form of ATP or heat
What speeds up the rate of a chemical reaction without becoming part of the products or being consumed?
Catalysts speeds up the rate of a chemical reaction without becoming part of the products or being consumed
Enzymes overcome the ____________ allowing the reaction to occur faster.
Enzymes overcome the activation energy allowing the reaction to occur faster.
In what three ways do enzymes overcome the activation energy?
- Increasing thermal energy (heating) to increase the velocity of molecules
- Increasing the concentration of reactants to increase the rate of molecular collisions
- Adding a catalyst
Characteristics of Enzymes include:
- Most composed of protein and may require cofactors
- Act as organic catalysts to speed up the rate of cellular reactions
- Lower the activation every required for a chemical reaction proceed
- Have unique characteristics such as shape, specificity, and function
- Enable metabolic reactions to proceed at a speed compatible with life
- Have an active site for target molecules (substrates)
- Much larger in size than their substrates
- Associate closely with substrates but do not become integrated into the reaction products
- Not used up or permanently changed by reaction
- Can be recycles, thus function in extremely low concentrations
- Are greatly affected by temperature and pH
- Can be regulated by feedback and genetic mechanisms
What do enzymes work on?
Enzymes work on substrates.
- Substrates are reactant molecules upon which enzymes act.
How do enzymes work?
Enzymes bind to substrates and participate directly in changes to the substrate
- Never a part of the products; Does not become part of the products
- Is not used up by the reaction
- Can function over and over again
What are simple enzymes?
Simple enzymes consist of protein alone and that protein does everything
What is a conjugated enzyme?
Conjugated enzymes contain protein and some other nonprotein molecules
Conjugated enzymes as a whole are called ___________.
Conjugated enzymes as a whole are called holoenzymes.
Holoenzymes consists of ____________ and _____________.
Holoenzymes consists of apoenzyme and cofactor.
Apoenzyme
Protein portion of the holoenzyme
Cofactor
Nonprotein portion of the holoenzyme
- Organic molecule, also called coenzyme
- Inorganic (metal ions)
What the active site/catalytic site on apoenzymes?
- Actual site where the substrate binds
- Three-dimensional crevice or groove formed by the way amino acid chains are folded
- Each enzyme has a different: Primary structure, Variation in folding, Unique active site
How do the active site and specificity of the apoenzyme arise?
- As the polypeptide forms intrachain bonds, it folds in a three-dimensional (tertiary) state. Active sites (AS) are created by the 3D shape.
- More complex enzymes have a quaternary structure consisting of several polypeptides bound by weak forces. Often the active site is formed by the junction of two polypeptides.
How do enzyme substrate reactions work?
- When the enzyme and substrate come together, the substrate (S) must show the correct fit and position with respect to the enzyme (E).
- When the ES complex is formed, it enters a transition state. During this temporary but tight interlocking union, the enzyme participates directly in breaking or making bonds.
- Once the reaction is complete, the enzyme releases the products.
What are the metallic cofactors?
- Iron
- Copper
- Magnesium
- Manganese
- Zinc
- Cobalt
- Selenium
- And so on
What is the purpose of the metallic cofactors?
Assist with precise functions between enzyme and substrate
- Activate enzymes
- Help bring the active site and substrate close together
- Participate directly in chemical reactions
What is the importances of coenzymes?
- Organic compounds that work with the apoenzyme to alter the substrate
- Remove a chemical group from one substrate and add it to another substrate
- Carry and transfer hydrogen atoms, electrons, carbon dioxide, and amino groups
- Vitamins are an important component of coenzymes
What are the 6 classes of enzymes?
- Oxidoreductase and dehydrogenase
- Transferase
- Hydrolases
- Lyases
- Isomerases
- Ligases
Oxidoreductase
Transfer electrons from one substrate to another
Dehydrogenase
Transfer a hydrogen from one compound to another
Transferases
Transfer functional groups from one substrate to another
Hydrolases
Cleave bonds on molecules with the addition of water
Lyases
Add groups to or remove groups from double-bonded substrates
Isomerases
Change a substrate to its isomeric form
Ligases
Catalyze the formation of bonds with the input of ATP and the removal of water
Transfer reactions are accomplished by:
Oxidoreductases
Compare oxidation vs. reduction
Oxidation:
- Loss of electrons
- A compound that loses electrons is oxidized
Reduction:
- Gain of electrons
- A compound that gains electrons is reduced
____ and ___ are coenzyme carriers.
NAD and FAD are coenzyme carriers.
Exoenzymes
- Transported extracellularly
- Break down large food molecules or harmful chemicals
Endoenzymes
- Retained intracellularly and function inside the cell
- Most enzymes of metabolic pathways
Constitutive Enzymes
Always present in relatively constant amounts, regardless of the cellular environment
- Not dependent on the amount of substrate
- Protein amount never changes
Regulated Enzymes
- Production is turned on (induced) or turned off (repressed) in response to changes in concentration of substrate
- May not always be around at all times
- Dependent on the amount of substrate
- More substrate = increase the expression of that particular enzyme
- Decrease substrate = repress that enzyme, thus downregulated the gene expression
- Example: Repressible operon, structural operon
True or False: When you add more substrate, the constitutive enzyme increases.
False
- When you add more substrate, the regulated enzyme is induced.
- When you remove substrate, the regulated enzyme is repressed.
- There is no change in amount of constitutive enzyme when the amount of substrate changes.
What is the role of microbial enzymes in disease?
- Pathogens secrete unique exoenzymes that help them avoid host defenses or promote multiplication in tissues.
Exoenzymes
- Considered virulence factors because these enzymes contribute to pathogenicity (Usually trying to extract nutrients from their hosts)
- Some of these enzymes function as toxins
- Examples: streptokinase, streptolysin, elastase, collagenase, lipase, and penicillinase
How are microbial enzymes transferred?
Via horizontal gene transfer
Changes from normal conditions make enzymes to be chemically _______, also known as ________
Changes from normal conditions make enzymes to be chemically unstable, also known as labile
Denaturation of enzymes:
- Weak bonds that maintain the shape of an apoenzyme are broken by heat, low or high pH, or certain chemicals
- Disruption distorts the enzyme’s shape
- Prevents the substrate from attaching to the active site
- Nonfunctional enzymes block metabolic reactions and can lead to cell death
Metabolic pathways most often occur in...
Multistep series or pathway
Metabolic Pathways
- Each step is catalyzed by an enzyme
- The product of one reaction is often the reactant (substrate) for the next
- Many pathways have branches that have alternate methods for nutrient processing
- Some pathways take a cyclic form
- Pathways are interconnected and merge at many sites
What are different patterns of metabolism? Include examples of each.
- Linear (example: Glycolysis)
- Cyclic (example: Kreb's)
- Branched (Divergent or Convergent)
Example of divergent pathway
Protein catabolism
Example of convergent pathway
Amino acid synthesis
Competitive Inhibition
- A molecule that resembles the substrate (called the mimic) occupies the active site, preventing the substrate from binding
- Enzyme cannot act on the inhibitor and is effectively shut down
- Direct control on the action of enzymes
Noncompetitive Inhibition
- Some enzymes have two binding sites—the active site and the regulatory site
- Regulated by the binding of molecules other than the substrate to the regulatory site
- Often, the regulatory molecule is the product of the enzymatic reaction; Provides negative feedback that slows enzyme activity once a certain concentration of product is reached
- Negative Feedback
- Buildup of products turns off the enzyme for that particular reaction
- Usually, this is only if the enzyme has a regulatory domain
- Direct control on the action of enzymes
What are the two direct controls on the action of enzymes?
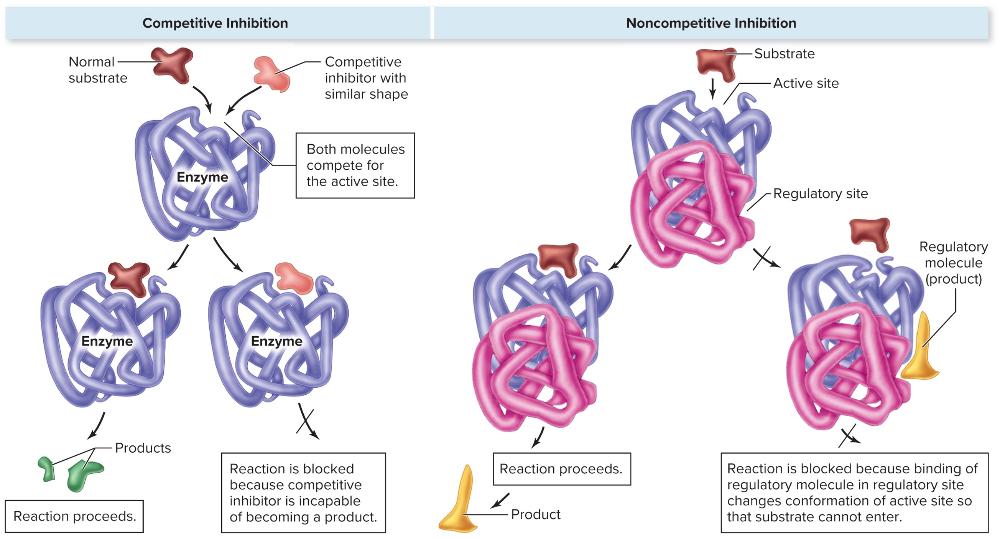
- Competitive Inhibition
- Noncompetitive Inhibition
Another control on enzyme synthesis is ____________
Another control on enzyme synthesis is enzyme repression.
How does enzyme repression work?
Enzyme repression: stops further synthesis of an enzyme somewhere along its pathway
- If the end product of an enzymatic reaction reaches excess, the genetic apparatus for replacing enzymes is suppressed
- Response time is longer than for feedback inhibition, effects more enduring
Genetic control of enzyme synthesis: Enzyme Repression Steps
1. DNA is transcribed into RNA
2. RNA translated into protein
3. Protein
4. Folds to form function enzyme structure
5. Substrate + Enzyme = Product
6. Excess product binds to DNA and shuts down further enzyme production
Enzyme Induction
- Enzymes appear (are induced) only when suitable substrates are present. Synthesis of an enzyme is induced by its substrate; inverse of enzyme repression
- The system is naturally off, but when a substrate becomes available, then that causes upregulation of that particular enzyme to be expressed.
- No substrate = Not induced
Exergonic Reactions
- Release energy as they go forward
- Energy is available for doing cellular work
Endergonic Reactions
- Require the addition of energy to move forward
True or False: Exergonic and endergonic reactions are often coupled.
True
- So, when we look at the breakdown of nutrients, we think about breaking the bonds and releasing electrons and molecules that can help generate energy in a stepwise fashion, where you release energy, and that released energy is being used to make precursor molecules and eventually building blocks of the cell.
- During exergonic reactions, energy released by bonds is stored in certain high-energy phosphate bonds, such as in ATP. The ability of ATP to temporarily store and release the energy of chemical bonds fuels the endergonic cell reactions.
What is the simplified model of energy production?
- Glucose is oxidized as it passes through sequential metabolic pathways, resulting in the removal of hydrogens and their accompanying electrons. The energy from the hydrogens and electrons is used to generate ATP. Eventually, all that is left of the carbon skeleton of glucose is the end product CO2. Another by-product of aerobic metabolism (due to electrons and hydrogen ions combining with oxygen) is H2O.
What are the coenzyme carriers?
NAD and FAD
Redox reactions always occur in pairs called _______ pairs. The enzymes important in cellular redox reactions are...
Redox reactions always occur in pairs called redox pairs. The enzymes important in cellular redox reactions are oxidoreductase.
________ agent will accept electrons. _______ agent will give up the electrons.
Oxidizing agent will accept electrons. Reducing agent will give up electrons.
NAD Reduction
NAD+ --> NADH + H+
The reduced form is ______
NADH
The oxidized form is
NAD+
In aerobic metabolism, this acceptor is molecular ______ ; in anaerobic metabolism, it is ________.
In aerobic metabolism, this acceptor is molecular oxygen; in anaerobic metabolism, it is some other inorganic or organic compound.
Newly ___________ compounds (the ones that gain electrons) have more energy than they did in their _________ state
Newly reduced compounds (the ones that gain electrons) have more energy than they did in their oxidized state
The energy now present in the electron acceptor can be captured to ____________ (add an inorganic phosphate) to ADP or some other compound
The energy now present in the electron acceptor can be captured to phosphorylate (add an inorganic phosphate) to ADP or some other compound
- This process stores the energy in a high-energy molecule
True or False: The electrons and atoms are separate entities in the cell.
False
Electron Transfer
- The cell handles electrons as parts of an atom and not as separate entities
- Hydrogens contain a proton and an electron and are often involved in the transfer of electrons processes
- The removal of hydrogens from a compound during a redox reaction is called dehydrogenation
- The job of handling these protons and electrons falls to one or more carriers
- Dehydrogenations are an essential supplier of electrons for the respiratory electron transport system
NAD
- Most common electron carrier
- Carries hydrogens and a pair of electrons from dehydrogenation reactions
- Reduced NAD is presented as NADH+ + H+ or NADH
FAD
- Reduced FAD is presented as FADH
NADP
- NAD phosphate
The electron carriers are:
- NAD
- FAD
- NADP
In catabolic pathways, electrons are ______ and carried through a series of redox reactions to a final electron acceptor.
In catabolic pathways, electrons are extracted and carried through a series of redox reactions to a final electron acceptor.
What is the final electron acceptor in aerobic metabolism?
Oxygen
What is the final electron acceptor in anaerobic metabolism?
Other inorganic or organic compound
The adenosine triphosphate is a 3-part molecule that consists of:
- Adenine - Nitrogen base
- Ribose - 5 carbon sugar
- Three phosphate groups bonded to the ribose
Describe the role of the three phosphate groups bonded to the ribose
- Bulky and have a negative charge
- Repelling electrostatic charges imposes a strain between the last two phosphate groups
- Removal of phosphates releases free energy
Adenosine consists of:
- Adenine
- Ribose
The primary energy currency of the cell is
ATP
ATP must be ___________ when used in a chemical reaction.
ATP must be replaced when used in a chemical reaction.
- ATP utilization and replenishment is an ongoing cycle
What is substrate-level phosphorylation?
Generation of ATP through a transfer of a phosphate group from a phosphorylated compound directly to ADP
What is oxidative phosphorylation?
A series of redox reactions occurring during the final phase of the respiratory pathway
What is photophosphorylation?
ATP formed through a series of sunlight-driven reactions in phototrophs
What are the three basic catabolic pathways?
- Aerobic Respiration
- Anaerobic Respiration
- Fermentation
Glycolysis is the most common pathway used to break down ___________.
Glycolysis is the most common pathway used to break down glucose.
For aerobic respiration, glycolysis yields ______, Kreb's cycle yields ________, and the ETC yields ___________. It also uses _____ as the electron acceptor. Overall, the maximum net yield is ________.
For aerobic respiration, glycolysis yields 2 ATPs, Kreb's cycle yields 2 ATPs/GTPs, and the ETC yields 36-38 ATPs. It also uses O2 as the electron acceptor. Overall, the maximum net yield is 36-38 ATPs.
For anaerobic respiration, glycolysis yields ______, Kreb's cycle yields ________, and the ETC yields ___________. It also uses _____ as the electron acceptor. Overall, the maximum net yield is ________.
For anaerobic respiration, glycolysis yields 2 ATP, Kreb's cycle yields 2 ATP, and the ETC yields 2-36 ATPs. It also uses non-O2 compound as the electron acceptor. Overall, the maximum net yield is 2-36 ATP.
Fermentation produces the ________ amount of ATP (__ ATP). The process uses ______ as electron acceptor. It produces _____, ______.
Fermentation produces the least amount of ATP (2 ATP). The process uses organic compounds as electron acceptor. It produces alcohol, acids.
- YOU ONLY DO GLYCOSIS
- THERE IS NO KREB OR ETC FOR FREMENTATION
Both aerobic respiration and anaerobic respiration convert ______ to _____.
Both aerobic respiration and anaerobic respiration convert glucose to CO2.
Fermentation pathways are used for:
Facultative and aerotolerant anaerobes
- Facultative anaerobes - Organisms that grow with or without oxygen, but grow better when oxygen is present.
- Aerotolerant - Do not use oxygen, but can tolerate its presence
Fermentation uses only _______
Fermentation uses only glycolysis
The final electron acceptor for fermentation is...
Organic Compounds
Aerobic heterotrophs use _______ respiration.
Aerobic
Aerobic Respiration
- A series of enzyme-catalyzed reactions (Electrons transferred from fuel molecules to oxygen as a final electron acceptor)
- Principal energy-yielding scheme for aerobic heterotrophs
- Provides ATP and metabolic intermediates for other pathways
What is the starting compound? And why?
- Glucose
- Carbohydrates such as glucose are good fuels because they are readily oxidized; They are excellent hydrogen and electron donors
- The enzymatic withdrawal of hydrogens from glucose removes electrons that can be used in energy transfers
- The end products of the conversion of these carbon compounds are energy-rich ATP and energy-poor carbon dioxide and water
The first step in all pathways is ________.
The first step in all pathways is glycolysis.
Glucose is enzymatically converted to _____ ______ which is an essential intermediary metabolite.
Glucose is enzymatically converted to pyruvic acid which is an essential intermediary metabolite.
Glycolysis may be the _____ phase of aerobic respiration or the primary metabolite pathway.
Glycolysis may be the first phase of aerobic respiration or the primary metabolite pathway.
- Synthesizes a small amount of ATP anaerobically
The central metabolite is...
Pyruvic Acid
How is pyruvic acid used in each pathway?
- Strict aerobes and some anaerobes send pyruvic acid to the Krebs cycle
- Facultative anaerobes re-reduce pyruvic acid into acids or other products
In anabolic pathways, pyruvic acid produces ____, ____, and _____.
In anabolic pathways, pyruvic acid produces amino acids, sugars, and fat metabolites.
In fermentation, pyruvic acid produces...
- Acids, gas
- Acetaldehyde which further makes alcohol, acetone, 2,3-butanediol
For the Krebs cycle, _______ ____ is first converted to ______ _______ ____ before it enters the Krebs cycle.
For the Krebs cycle, pyruvic acid is first converted to acetyl coenzyme A before it enters the Krebs cycle.
________ reaction releases the first ____ molecule for the Krebs cycle.
Oxidation reaction releases the first CO2 molecule for the Krebs cycle.
Describe the Krebs cycle
- Pyruvic acid is first converted to acetyl coenzyme A (acetyl CoA) before it enters the Krebs cycle
- Oxidation reaction releases the first C O2 molecule
- A cluster of enzymes and coenzyme A dehydrogenate pyruvic acid to a 2-carbon acetyl group
- NAD is reduced to NADH
- NADH formed is shuttled to the electron transport system to produce ATP
All reactions that occur in the Krebs cycle happen ______ for each glucose because ____ pyruvates are formed during glycolysis.
All reactions that occur in the Krebs cycle happen twice for each glucose molecule because two pyruvates are formed during glycolysis
The Krebs cycle serves to...
Serves to transfer the energy stored in acetyl CoA to NAD+ and FAD by reducing them
What are the main products of the Krebs cycle:
- Reduced NADH and FADH2
- Two ATP produced through substrate-level phosphorylation
A single spin around the Krebs cycle has eight steps, beginning with _____ _____ formation and ending with _____ acid
A single spin around the Krebs cycle has eight steps, beginning with citric acid formation and ending with oxaloacetic acid
What is the electron transport system (ETS)?
- Chain of special redox carriers that receives electrons from NADH and FADH2
- Electrons are passed sequentially from one redox molecule to the next
- Flow of electrons allows the active transport of hydrogens outside the cell membrane
- Oxygen receives hydrogens and electrons and produces water
The sequence of electron carriers in the respiratory chain of most aerobic organisms is:
- NADH dehydrogenase
- flavin mononucleotide (FMN)
- coenzyme Q
- cytochrome b
- cytochrome c1
- cytochrome c
- cytochromes a and a3 (which are complexed together)
Where is the electron transport system located?
On the inner membrane of the mitochondrial cristae
What are the elements of the electron transport?
- ATP Synthase
- Oxidative Phosphorylation
Role of ATP Synthase
- Stationed along the membrane in close association with the ETS carriers
- Captures released energy from the ETS carriers
Role of Oxidative Phosphorylation
- The coupling of ATP synthesis to electron transport
- Each NADH that enters the ETS gives rise to three ATP molecules
- NAD and FMN enter the ETS at a different point, so there is less energy released, and only give rise to two ATP molecules
As the electron transport carriers shuttle electrons, hydrogen ions are _____________ pumped into the periplasmic space or the space between the cell wall and the cytoplasmic membrane. This sets up a concentration gradient of hydrogen ions called _____ _____ ______.
As the electron transport carriers shuttle electrons, hydrogen ions are actively pumped into the periplasmic space or the space between the cell wall and the cytoplasmic membrane. This sets up a concentration gradient of hydrogen ions called proton motive force.
What is the proton motive force?
- Consists of a difference in charge between the outside of the membrane (+) and the inside (−)
- Separation of charges temporarily stores potential energy
- H+ can only diffuse into the membrane through ATP synthase, which sets the stage for ATP synthesis
Is ATP synthesis the same in eukaryotes?
ATP synthesis in eukaryotes occurs through the same overall process.
- ETS is station in mitochondrial membranes, between the mitochondrial matrix and the outer intermembrane space
- This difference affects the amount of ATP produced
What is the theoretical yield from aerobic respiration?
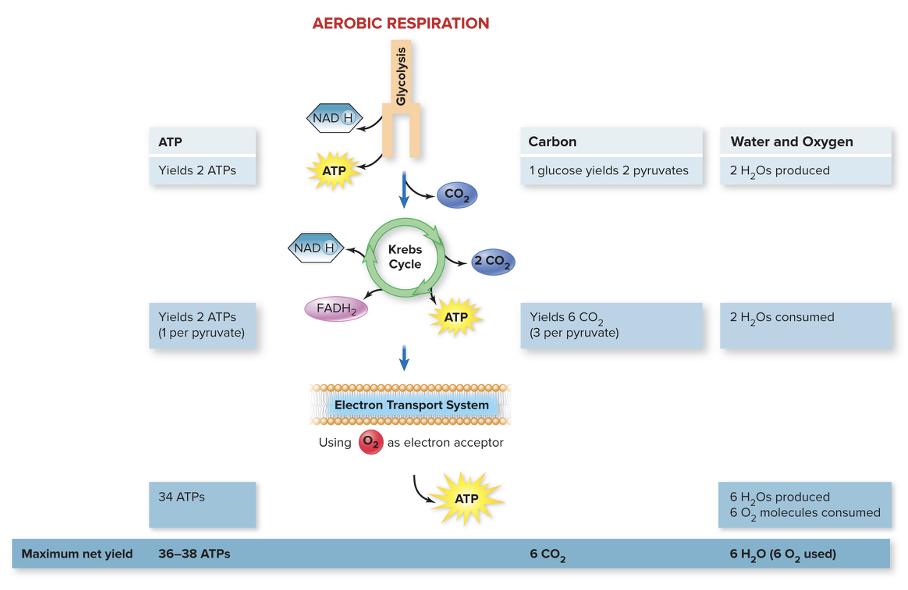
- 36-38 ATPs
- 6CO2
- 6 H2O (6O2 used)
Summary of Aerobic Respiration (ATP generation)
Total possible production of ATP is 40:
- Four from glycolysis
- Two from the Krebs cycle
- Thirty-four from electron transport
- Minus 2 ATP expended in early glycolysis equals a maximum of 38 ATP per molecule of glucose
Actual total ATPs produced may be lower in certain eukaryotic cells
- Energy is expended transporting NADH across the mitochondrial membrane during glycolysis
Summary of Aerobic Respiration (Other Products)
Six CO2 molecules generated during the Krebs cycle
Six O2 molecules consumed during electron transport
Six H2O molecules produced in electron transport and two in glycolysis
- Two used in the Krebs cycle
- Net total of 6H2O molecules
The terminal step is catalyzed by the enzyme __, also called ___________.
The terminal step is catalyzed by the enzyme aa3, also called cytochrome oxidase.
What is formed by the terminal step?
Adapted to receive electrons from cytochrome c, pick up hydrogens from solution, and react with oxygen to form water
2H+ + 2e- +1/2 O2 --> H2O
E. coli undergoes __________ respiration.
E. coli undergoes anaerobic respiration.
The nitrate and nitrite reduction system is a test that can used for...
identification of bacteria
Nitrate reductase catalyzes the removal of _______ from nitrate (NO3) reducing it to nitrite (NO2) and water
Nitrate reductase catalyzes the removal of _______ from nitrate (NO3) reducing it to nitrite (NO2) and water
What is fermentation?
- The incomplete oxidation of glucose or other carbohydrates in the absence of oxygen
- Uses organic compounds as the terminal electron acceptors
- Yields a small amount of ATP
_____ is constantly recycled in fermentation and reused for continuation of glycolysis
NAD
The two last products of fermentation are:
- Ethyl alcohol
- Lactic acid
What are the products of alcoholic fermentation?
- Occurs in yeast or bacterial species that have metabolic pathways for converting pyruvic acid to ethanol
- Decarboxylation of pyruvic acid to acetaldehyde
- Reduction of acetaldehyde to ethanol
- NADH formed during glycolysis is oxidized, regenerating NAD and allowing glycolysis to continue
What are the products of acidic fermentation?
- Pathways extremely varied
- Homolactic fermentation: Lactic acid bacteria reduce pyruvate to lactic acid mainly
- Heterolactic fermentation: glucose is fermented to a mixture of lactic acid, acetic acid, and carbon dioxide
What are the products of mixed fermentation?
- Members of the family Enterobacteriaceae possess enzyme systems for converting pyruvic acid to several acids simultaneously (Acetic, lactic, succinic, formic acids, as well as CO2)
- This fermentative activity accounts for the accumulation of some types of gas in the intestine
What is the role of lipases in lipid catabolism?
Break apart fatty acids joined to glycerol
- Glycerol converted to dihydroxyacetone phosphate which can enter Step 4 of glycolysis
What is beta oxidation?
- Oxidation of fatty acids
- 2-carbon units transferred to coenzyme A, creating acetyl CoA, which enters the Krebs cycle
- Oxidation of a 6-carbon fatty acid yields 50 ATP, compared to 38 for a 6-carbon sugar
What is the role of proteases in protein catabolism?
- Break down proteins to their amino acid components
- Amino groups removed through deamination
- Remaining carbon compound can be easily converted to one of several Krebs cycle intermediates
What is amphibolism?
- Most catabolic pathways contain strategic molecular intermediates that can be diverted into anabolic pathways
- A given molecule can serve multiple purposes to derive maximum benefit from all nutrients and metabolites
- Catabolic and anabolic pathways are integrated to improve cell efficiency
What is precursor molecule?
A compound that is the source of another compound
- Glyceraldehyde-3-phosphate can be diverted from glycolysis and converted into precursors for amino acid, carbohydrate, and triglyceride synthesis
Gluconeogenesis is....
Pyruvate is a starting point glucose synthesis in the event of inadequate glucose supply
Pyruvate as a precursor:
- Provides intermediates for amino acids
- Gluconeogenesis: pyruvate is a starting point for glucose synthesis in the event of inadequate glucose supply
Acetyl CoA as a precursor:
- Can be converted into one of several amino acids
- Can be condensed into hydrocarbon chains for fatty acid and lipid synthesis
Precursors to DNA and RNA:
- Pathways that synthesize purines and pyrimidines originate in amino acids
- Can be dependent on intermediates from the Krebs cycle
What is a crucial role of glucose in metabolism and energy utilization?
- Biosynthesis ensured by several alternative pathways
- Major component of cellulose cell walls and storage granules
- Glucose-6-P used to form glycogen
Role of Proteins
- Large proportion of a cell’s contents
- Essential components of enzymes, cytoplasmic membrane, cell wall, and cell appendages
- Twenty amino acids are needed to make these proteins
- Some organisms (for example, E. coli) have pathways to synthesize all 20 amino acids
To assemble the cell, cell division must take place, and therefore:
- Two cells need twice as many ribosomes, enzymes, and so on.
- Cell wall and membranes nearly double
- Phospholipid bilayer components assemble spontaneously with no energy input
- Proteins and other components must be added to the membranes
- Cell wall growth requires energy input
Light-Dependent Reactions
- Proceed only in the presence of sunlight
- Catabolic, energy-producing reactions
Light-Independent Reactions
- Proceed regardless of the lighting conditions
- Anabolic, synthetic reactions
- Carbon atoms from CO2 are added to the carbon backbones of organic molecules
Photophosphorylation is a....
Light-Dependent Reaction
- Channels in the thylakoids pump H+ into the inner chamber, producing a charge gradient
- ATP synthase phosphorylates ADP to ATP
- NADPH and ATP are released into the stroma of the chloroplast where they drive the reactions of the Calvin cycle
The calvin cycle is...
Light-Independent Reaction
- Occurs in the chloroplast stroma or the cytoplasm of cyanobacteria
- Use energy produced in the light phase to synthesize glucose
Oxygenic (oxygen-releasing) photosynthesis:
- Dominant type on earth
- Occurs in plants, algae, and cyanobacteria
Anoxygenic photosynthesis:
- Occurs in green and purple bacteria that utilize bacteriochlorophyll
- Have only cyclic photosystem I
- Generate a small amount of ATP
- Use H2, H2S, or other elemental sulfur as an electron source
- Many are strict anaerobes