Instructions for Side by Side Printing
- Print the notecards
- Fold each page in half along the solid vertical line
- Cut out the notecards by cutting along each horizontal dotted line
- Optional: Glue, tape or staple the ends of each notecard together
BMD 330 Chapter 10 - Microbial Metabolism
front 1 What is metabolism? | back 1 All chemical reactions and physical workings of the cell |
front 2 _____ requires energy input whereas ___ releases energy | back 2 Anabolism requires energy input whereas catabolsim releases energy |
front 3 What is anabolism? | back 3 The process of biosynthesis (synthesis of cell molecules and structures) |
front 4 What is catabolism? | back 4 Break the bonds of larger molecules |
front 5 Both ___ and ___ make up the metabolism. | back 5 Both catabolism and anabolism make up the metabolism. |
front 6 What is purpose of metabolism> | back 6
|
front 7 What speeds up the rate of a chemical reaction without becoming part of the products or being consumed? | back 7 Catalysts speeds up the rate of a chemical reaction without becoming part of the products or being consumed |
front 8 Enzymes overcome the ____________ allowing the reaction to occur faster. | back 8 Enzymes overcome the activation energy allowing the reaction to occur faster. |
front 9 In what three ways do enzymes overcome the activation energy? | back 9
|
front 10 Characteristics of Enzymes include: | back 10
|
front 11 What do enzymes work on? | back 11 Enzymes work on substrates.
|
front 12 How do enzymes work? | back 12 Enzymes bind to substrates and participate directly in changes to the substrate
|
front 13 What are simple enzymes? | back 13 Simple enzymes consist of protein alone and that protein does everything |
front 14 What is a conjugated enzyme? | back 14 Conjugated enzymes contain protein and some other nonprotein molecules |
front 15 Conjugated enzymes as a whole are called ___________. | back 15 Conjugated enzymes as a whole are called holoenzymes. |
front 16 Holoenzymes consists of ____________ and _____________. | back 16 Holoenzymes consists of apoenzyme and cofactor. |
front 17 Apoenzyme | back 17 Protein portion of the holoenzyme |
front 18 Cofactor | back 18 Nonprotein portion of the holoenzyme
|
front 19 What the active site/catalytic site on apoenzymes? | back 19
|
front 20 How do the active site and specificity of the apoenzyme arise? | back 20
|
front 21 How do enzyme substrate reactions work? | back 21
|
front 22 What are the metallic cofactors? | back 22
|
front 23 What is the purpose of the metallic cofactors? | back 23 Assist with precise functions between enzyme and substrate
|
front 24 What is the importances of coenzymes? | back 24
|
front 25 What are the 6 classes of enzymes? | back 25
|
front 26 Oxidoreductase | back 26 Transfer electrons from one substrate to another |
front 27 Dehydrogenase | back 27 Transfer a hydrogen from one compound to another |
front 28 Transferases | back 28 Transfer functional groups from one substrate to another |
front 29 Hydrolases | back 29 Cleave bonds on molecules with the addition of water |
front 30 Lyases | back 30 Add groups to or remove groups from double-bonded substrates |
front 31 Isomerases | back 31 Change a substrate to its isomeric form |
front 32 Ligases | back 32 Catalyze the formation of bonds with the input of ATP and the removal of water |
front 33 Transfer reactions are accomplished by: | back 33 Oxidoreductases |
front 34 Compare oxidation vs. reduction | back 34 Oxidation:
Reduction:
|
front 35 ____ and ___ are coenzyme carriers. | back 35 NAD and FAD are coenzyme carriers. |
front 36 Exoenzymes | back 36
|
front 37 Endoenzymes | back 37
|
front 38 Constitutive Enzymes | back 38 Always present in relatively constant amounts, regardless of the cellular environment
|
front 39 Regulated Enzymes | back 39
|
front 40 True or False: When you add more substrate, the constitutive enzyme increases. | back 40 False
|
front 41 What is the role of microbial enzymes in disease? | back 41
|
front 42 Exoenzymes | back 42
|
front 43 How are microbial enzymes transferred? | back 43 Via horizontal gene transfer |
front 44 Changes from normal conditions make enzymes to be chemically _______, also known as ________ | back 44 Changes from normal conditions make enzymes to be chemically unstable, also known as labile |
front 45 Denaturation of enzymes: | back 45
|
front 46 Metabolic pathways most often occur in... | back 46 Multistep series or pathway |
front 47 Metabolic Pathways | back 47
|
front 48 What are different patterns of metabolism? Include examples of each. | back 48
|
front 49 Example of divergent pathway | back 49 Protein catabolism |
front 50 Example of convergent pathway | back 50 Amino acid synthesis |
front 51 Competitive Inhibition | back 51
|
front 52 Noncompetitive Inhibition | back 52
|
front 53 What are the two direct controls on the action of enzymes? | back 53 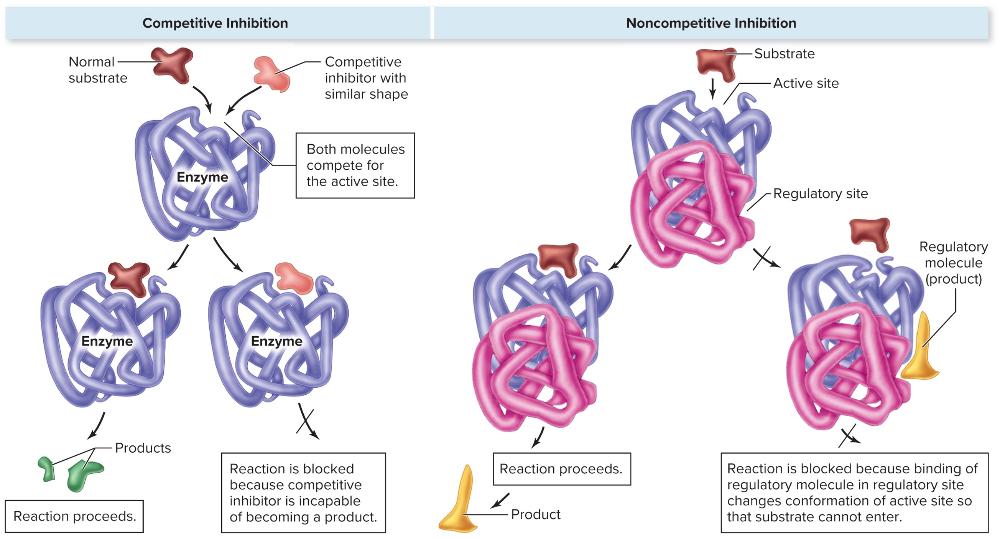
|
front 54 Another control on enzyme synthesis is ____________ | back 54 Another control on enzyme synthesis is enzyme repression. |
front 55 How does enzyme repression work? | back 55 Enzyme repression: stops further synthesis of an enzyme somewhere along its pathway
|
front 56 Genetic control of enzyme synthesis: Enzyme Repression Steps | back 56 1. DNA is transcribed into RNA 2. RNA translated into protein 3. Protein 4. Folds to form function enzyme structure 5. Substrate + Enzyme = Product 6. Excess product binds to DNA and shuts down further enzyme production |
front 57 Enzyme Induction | back 57
|
front 58 Exergonic Reactions | back 58
|
front 59 Endergonic Reactions | back 59
|
front 60 True or False: Exergonic and endergonic reactions are often coupled. | back 60 True
|
front 61 What is the simplified model of energy production? | back 61
|
front 62 What are the coenzyme carriers? | back 62 NAD and FAD |
front 63 Redox reactions always occur in pairs called _______ pairs. The enzymes important in cellular redox reactions are... | back 63 Redox reactions always occur in pairs called redox pairs. The enzymes important in cellular redox reactions are oxidoreductase. |
front 64 ________ agent will accept electrons. _______ agent will give up the electrons. | back 64 Oxidizing agent will accept electrons. Reducing agent will give up electrons. |
front 65 NAD Reduction | back 65 NAD+ --> NADH + H+ |
front 66 The reduced form is ______ | back 66 NADH |
front 67 The oxidized form is | back 67 NAD+ |
front 68 In aerobic metabolism, this acceptor is molecular ______ ; in anaerobic metabolism, it is ________. | back 68 In aerobic metabolism, this acceptor is molecular oxygen; in anaerobic metabolism, it is some other inorganic or organic compound. |
front 69 Newly ___________ compounds (the ones that gain electrons) have more energy than they did in their _________ state | back 69 Newly reduced compounds (the ones that gain electrons) have more energy than they did in their oxidized state |
front 70 The energy now present in the electron acceptor can be captured to ____________ (add an inorganic phosphate) to ADP or some other compound | back 70 The energy now present in the electron acceptor can be captured to phosphorylate (add an inorganic phosphate) to ADP or some other compound
|
front 71 True or False: The electrons and atoms are separate entities in the cell. | back 71 False |
front 72 Electron Transfer | back 72
|
front 73 NAD | back 73
|
front 74 FAD | back 74
|
front 75 NADP | back 75
|
front 76 The electron carriers are: | back 76
|
front 77 In catabolic pathways, electrons are ______ and carried through a series of redox reactions to a final electron acceptor. | back 77 In catabolic pathways, electrons are extracted and carried through a series of redox reactions to a final electron acceptor. |
front 78 What is the final electron acceptor in aerobic metabolism? | back 78 Oxygen |
front 79 What is the final electron acceptor in anaerobic metabolism? | back 79 Other inorganic or organic compound |
front 80 The adenosine triphosphate is a 3-part molecule that consists of: | back 80
|
front 81 Describe the role of the three phosphate groups bonded to the ribose | back 81
|
front 82 Adenosine consists of: | back 82
|
front 83 The primary energy currency of the cell is | back 83 ATP |
front 84 ATP must be ___________ when used in a chemical reaction. | back 84 ATP must be replaced when used in a chemical reaction.
|
front 85 What is substrate-level phosphorylation? | back 85 Generation of ATP through a transfer of a phosphate group from a phosphorylated compound directly to ADP |
front 86 What is oxidative phosphorylation? | back 86 A series of redox reactions occurring during the final phase of the respiratory pathway |
front 87 What is photophosphorylation? | back 87 ATP formed through a series of sunlight-driven reactions in phototrophs |
front 88 What are the three basic catabolic pathways? | back 88
|
front 89 Glycolysis is the most common pathway used to break down ___________. | back 89 Glycolysis is the most common pathway used to break down glucose. |
front 90 For aerobic respiration, glycolysis yields ______, Kreb's cycle yields ________, and the ETC yields ___________. It also uses _____ as the electron acceptor. Overall, the maximum net yield is ________. | back 90 For aerobic respiration, glycolysis yields 2 ATPs, Kreb's cycle yields 2 ATPs/GTPs, and the ETC yields 36-38 ATPs. It also uses O2 as the electron acceptor. Overall, the maximum net yield is 36-38 ATPs. |
front 91 For anaerobic respiration, glycolysis yields ______, Kreb's cycle yields ________, and the ETC yields ___________. It also uses _____ as the electron acceptor. Overall, the maximum net yield is ________. | back 91 For anaerobic respiration, glycolysis yields 2 ATP, Kreb's cycle yields 2 ATP, and the ETC yields 2-36 ATPs. It also uses non-O2 compound as the electron acceptor. Overall, the maximum net yield is 2-36 ATP. |
front 92 Fermentation produces the ________ amount of ATP (__ ATP). The process uses ______ as electron acceptor. It produces _____, ______. | back 92 Fermentation produces the least amount of ATP (2 ATP). The process uses organic compounds as electron acceptor. It produces alcohol, acids.
|
front 93 Both aerobic respiration and anaerobic respiration convert ______ to _____. | back 93 Both aerobic respiration and anaerobic respiration convert glucose to CO2. |
front 94 Fermentation pathways are used for: | back 94 Facultative and aerotolerant anaerobes
|
front 95 Fermentation uses only _______ | back 95 Fermentation uses only glycolysis |
front 96 The final electron acceptor for fermentation is... | back 96 Organic Compounds |
front 97 Aerobic heterotrophs use _______ respiration. | back 97 Aerobic |
front 98 Aerobic Respiration | back 98
|
front 99 What is the starting compound? And why? | back 99
|
front 100 The first step in all pathways is ________. | back 100 The first step in all pathways is glycolysis. |
front 101 Glucose is enzymatically converted to _____ ______ which is an essential intermediary metabolite. | back 101 Glucose is enzymatically converted to pyruvic acid which is an essential intermediary metabolite. |
front 102 Glycolysis may be the _____ phase of aerobic respiration or the primary metabolite pathway. | back 102 Glycolysis may be the first phase of aerobic respiration or the primary metabolite pathway.
|
front 103 The central metabolite is... | back 103 Pyruvic Acid |
front 104 How is pyruvic acid used in each pathway? | back 104
|
front 105 In anabolic pathways, pyruvic acid produces ____, ____, and _____. | back 105 In anabolic pathways, pyruvic acid produces amino acids, sugars, and fat metabolites. |
front 106 In fermentation, pyruvic acid produces... | back 106
|
front 107 For the Krebs cycle, _______ ____ is first converted to ______ _______ ____ before it enters the Krebs cycle. | back 107 For the Krebs cycle, pyruvic acid is first converted to acetyl coenzyme A before it enters the Krebs cycle. |
front 108 ________ reaction releases the first ____ molecule for the Krebs cycle. | back 108 Oxidation reaction releases the first CO2 molecule for the Krebs cycle. |
front 109 Describe the Krebs cycle | back 109
|
front 110 All reactions that occur in the Krebs cycle happen ______ for each glucose because ____ pyruvates are formed during glycolysis. | back 110 All reactions that occur in the Krebs cycle happen twice for each glucose molecule because two pyruvates are formed during glycolysis |
front 111 The Krebs cycle serves to... | back 111 Serves to transfer the energy stored in acetyl CoA to NAD+ and FAD by reducing them |
front 112 What are the main products of the Krebs cycle: | back 112
|
front 113 A single spin around the Krebs cycle has eight steps, beginning with _____ _____ formation and ending with _____ acid | back 113 A single spin around the Krebs cycle has eight steps, beginning with citric acid formation and ending with oxaloacetic acid |
front 114 What is the electron transport system (ETS)? | back 114
|
front 115 The sequence of electron carriers in the respiratory chain of most aerobic organisms is: | back 115
|
front 116 Where is the electron transport system located? | back 116 On the inner membrane of the mitochondrial cristae |
front 117 What are the elements of the electron transport? | back 117
|
front 118 Role of ATP Synthase | back 118
|
front 119 Role of Oxidative Phosphorylation | back 119
|
front 120 As the electron transport carriers shuttle electrons, hydrogen ions are _____________ pumped into the periplasmic space or the space between the cell wall and the cytoplasmic membrane. This sets up a concentration gradient of hydrogen ions called _____ _____ ______. | back 120 As the electron transport carriers shuttle electrons, hydrogen ions are actively pumped into the periplasmic space or the space between the cell wall and the cytoplasmic membrane. This sets up a concentration gradient of hydrogen ions called proton motive force. |
front 121 What is the proton motive force? | back 121
|
front 122 Is ATP synthesis the same in eukaryotes? | back 122 ATP synthesis in eukaryotes occurs through the same overall process.
|
front 123 What is the theoretical yield from aerobic respiration? | back 123 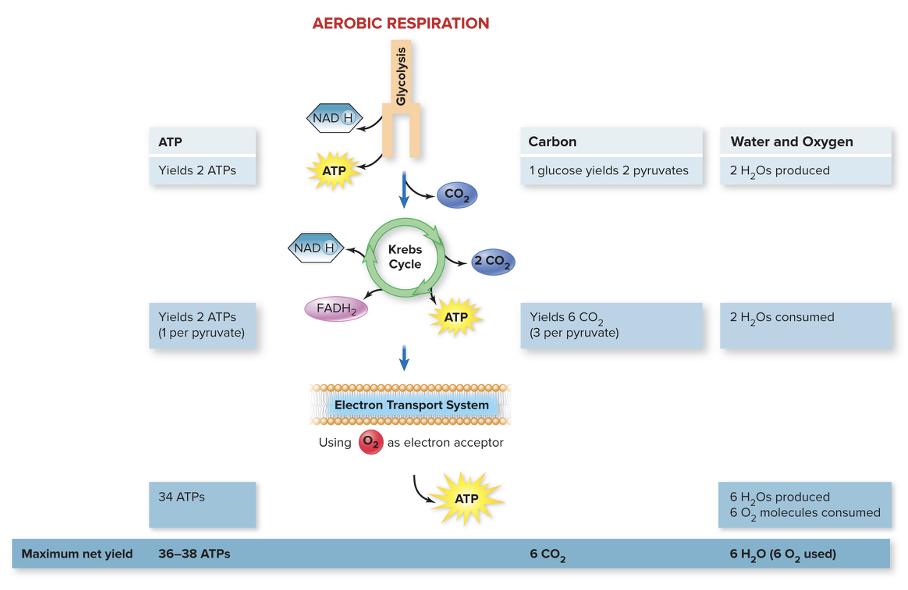
|
front 124 Summary of Aerobic Respiration (ATP generation) | back 124 Total possible production of ATP is 40:
Actual total ATPs produced may be lower in certain eukaryotic cells
|
front 125 Summary of Aerobic Respiration (Other Products) | back 125 Six CO2 molecules generated during the Krebs cycle Six O2 molecules consumed during electron transport Six H2O molecules produced in electron transport and two in glycolysis
|
front 126 The terminal step is catalyzed by the enzyme __, also called ___________. | back 126 The terminal step is catalyzed by the enzyme aa3, also called cytochrome oxidase. |
front 127 What is formed by the terminal step? | back 127 Adapted to receive electrons from cytochrome c, pick up hydrogens from solution, and react with oxygen to form water 2H+ + 2e- +1/2 O2 --> H2O |
front 128 E. coli undergoes __________ respiration. | back 128 E. coli undergoes anaerobic respiration. |
front 129 The nitrate and nitrite reduction system is a test that can used for... | back 129 identification of bacteria |
front 130 Nitrate reductase catalyzes the removal of _______ from nitrate (NO3) reducing it to nitrite (NO2) and water | back 130 Nitrate reductase catalyzes the removal of _______ from nitrate (NO3) reducing it to nitrite (NO2) and water |
front 131 What is fermentation? | back 131
|
front 132 _____ is constantly recycled in fermentation and reused for continuation of glycolysis | back 132 NAD |
front 133 The two last products of fermentation are: | back 133
|
front 134 What are the products of alcoholic fermentation? | back 134
|
front 135 What are the products of acidic fermentation? | back 135
|
front 136 What are the products of mixed fermentation? | back 136
|
front 137 What is the role of lipases in lipid catabolism? | back 137 Break apart fatty acids joined to glycerol
|
front 138 What is beta oxidation? | back 138
|
front 139 What is the role of proteases in protein catabolism? | back 139
|
front 140 What is amphibolism? | back 140
|
front 141 What is precursor molecule? | back 141 A compound that is the source of another compound
|
front 142 Gluconeogenesis is.... | back 142 Pyruvate is a starting point glucose synthesis in the event of inadequate glucose supply |
front 143 Pyruvate as a precursor: | back 143
|
front 144 Acetyl CoA as a precursor: | back 144
|
front 145 Precursors to DNA and RNA: | back 145
|
front 146 What is a crucial role of glucose in metabolism and energy utilization? | back 146
|
front 147 Role of Proteins | back 147
|
front 148 To assemble the cell, cell division must take place, and therefore: | back 148
|
front 149 Light-Dependent Reactions | back 149
|
front 150 Light-Independent Reactions | back 150
|
front 151 Photophosphorylation is a.... | back 151 Light-Dependent Reaction
|
front 152 The calvin cycle is... | back 152 Light-Independent Reaction
|
front 153 Oxygenic (oxygen-releasing) photosynthesis: | back 153
|
front 154 Anoxygenic photosynthesis: | back 154
|