Instructions for Side by Side Printing
- Print the notecards
- Fold each page in half along the solid vertical line
- Cut out the notecards by cutting along each horizontal dotted line
- Optional: Glue, tape or staple the ends of each notecard together
Chapters 19-20 Organic Chemistry II
front 1 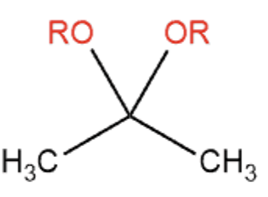 Name That Functional Group! | back 1 Acetal |
front 2 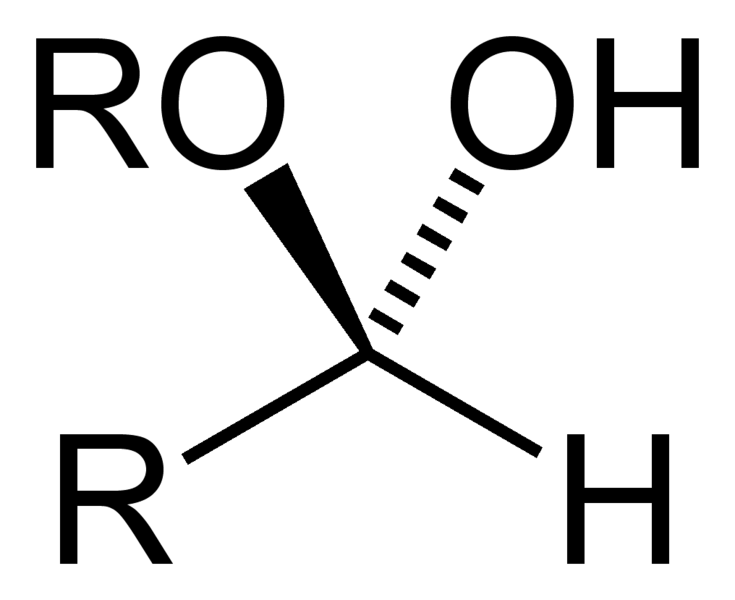 Name That Functional Group! | back 2 Hemiacetal |
front 3 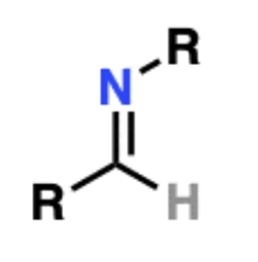 Name That Functional Group! | back 3 Imine |
front 4 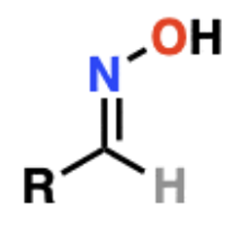 Name That Functional Group! | back 4 Oxime |
front 5 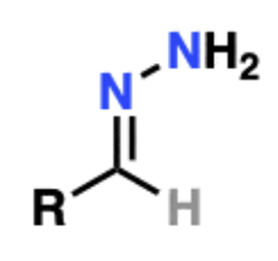 Name That Functional Group! | back 5 Hydrazone |
front 6 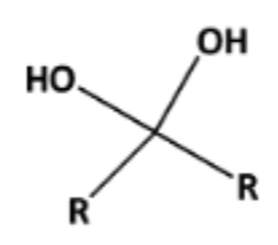 Name That Functional Group! | back 6 Gem-diol (hydroxyl groups are on the same carbon) |
front 7 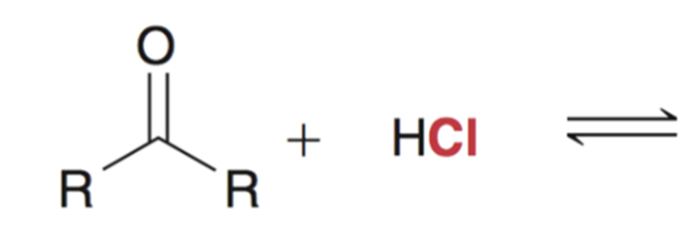 | back 7 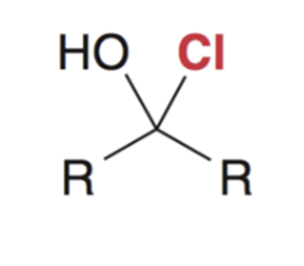 Reaction favors starting materials if nucleophile is a weak base (aka a good leaving group) |
front 8  | back 8 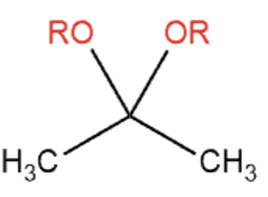 Alcohols attack carbonyl groups to form acetals. Common catalysts are TsOH and sulfuric acid |
front 9  | back 9 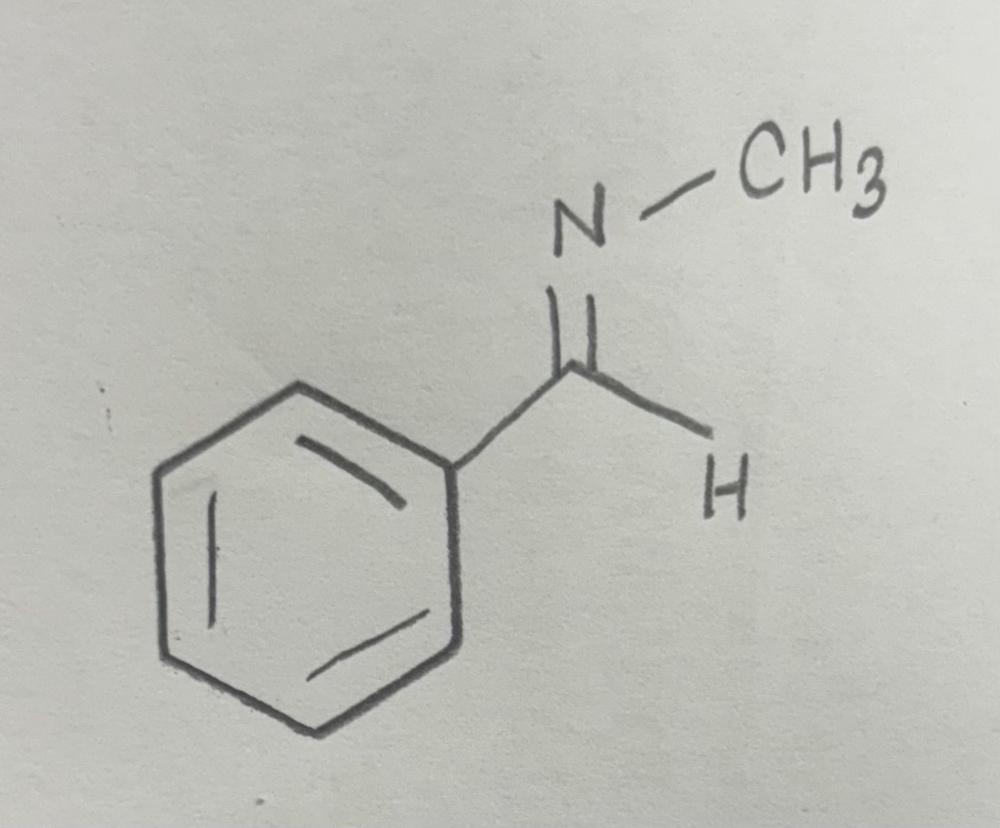 Imine formation using primary amine |
front 10  | back 10 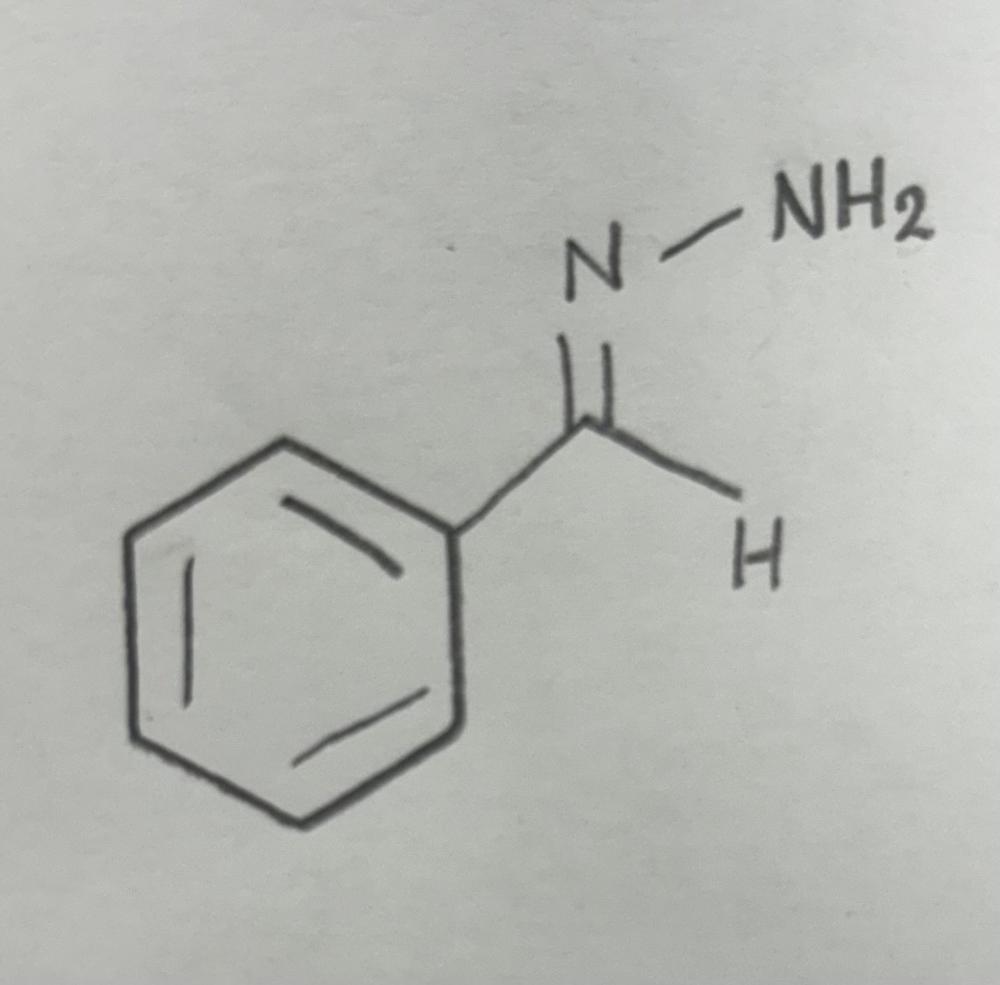 Hydrazone formation using primary amine |
front 11 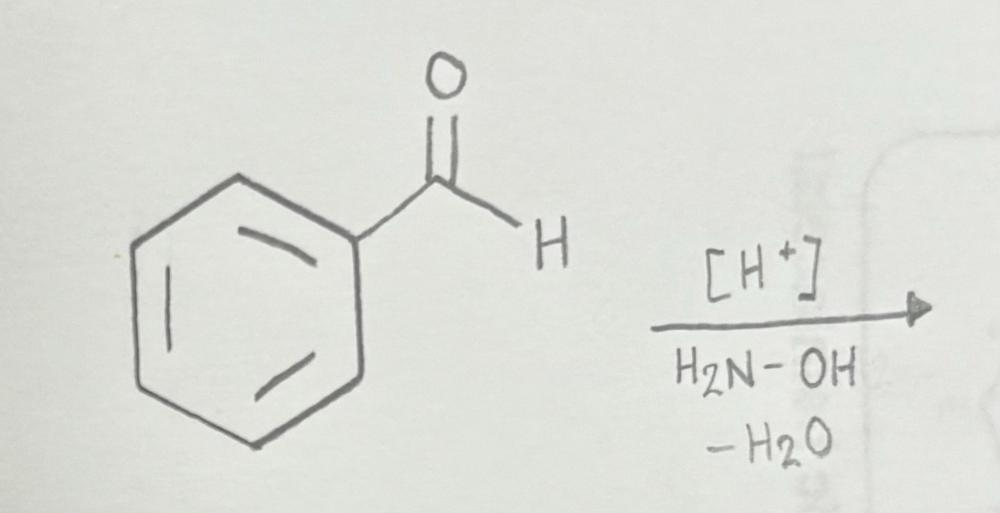 | back 11 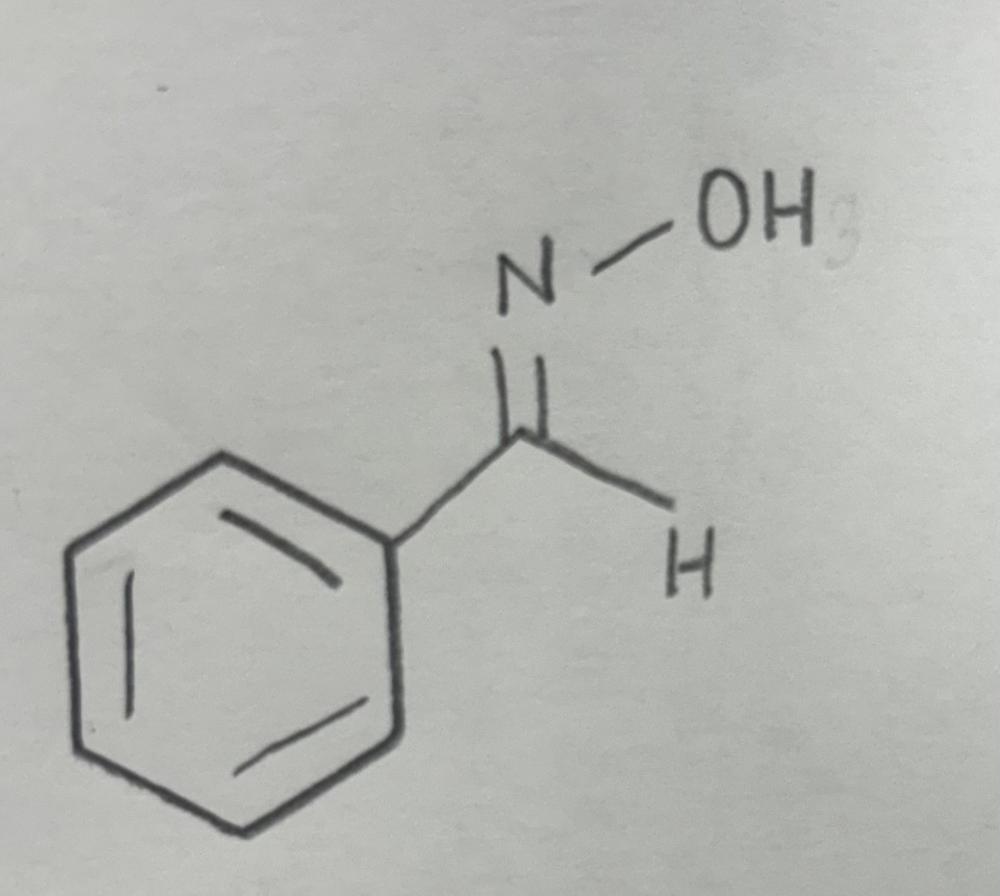 Oxime formation using primary amine |
front 12  | back 12 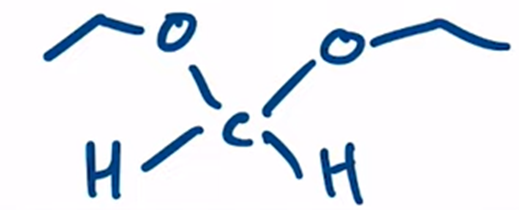 Acetal formation from formaldehyde products are favored with aldehydes |
front 13 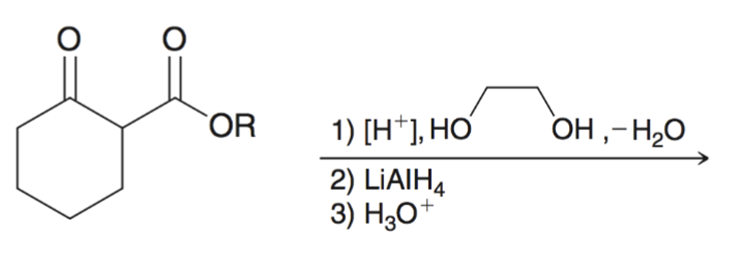 | back 13 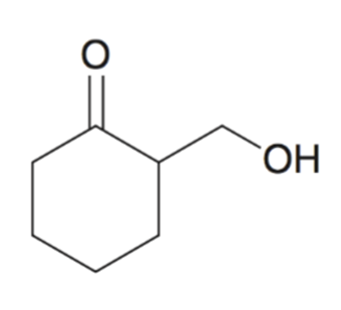 Using cyclic acetal as a protecting group |
front 14 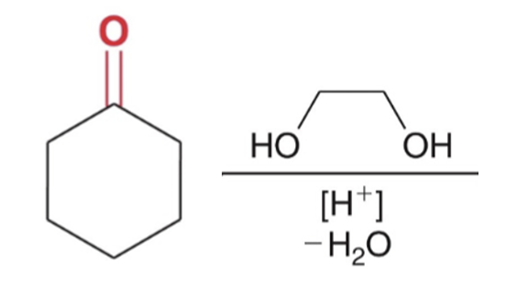 | back 14 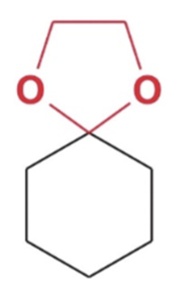 Cyclic acetal formation, Minus water favor products |
front 15  | back 15 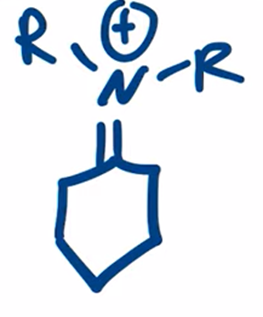 Secondary amine forms enamine |
front 16  Name That Functional Group! | back 16 Enamine |
front 17 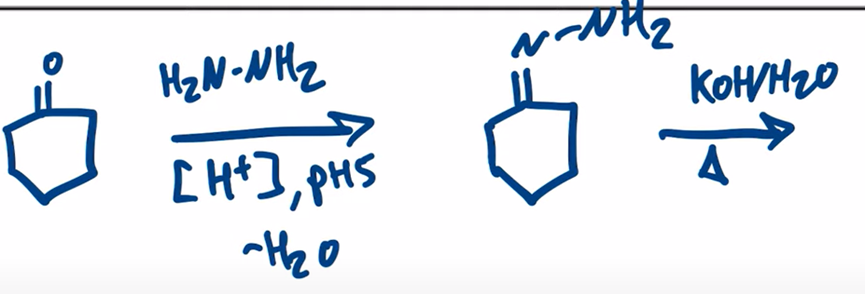 | back 17 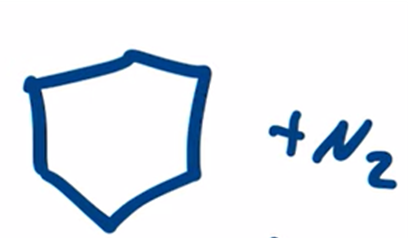 Wolff-Kishner Reduction obtain alkane from aldehyde or ketone by forming a hydrazone |
front 18 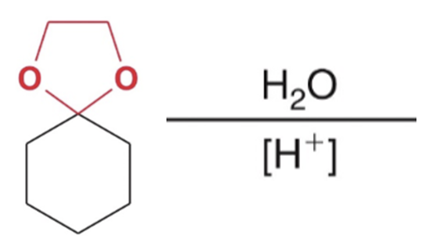 | back 18 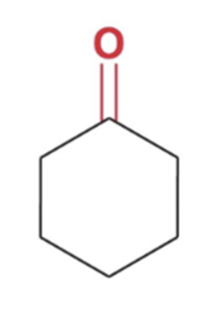 Reverse cyclic acetal formation, adding water favor reactants |
front 19 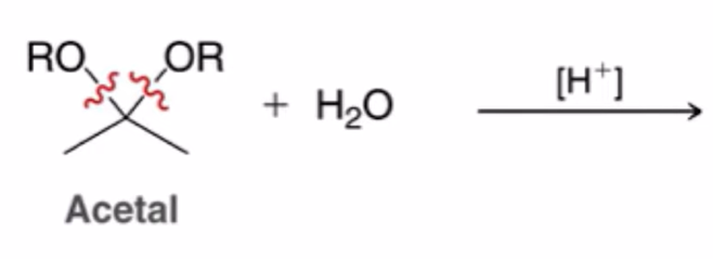 | back 19 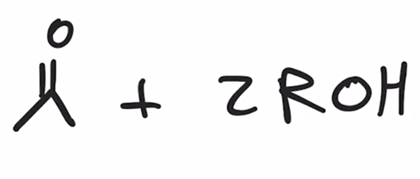 hydrolysis of acetals to yield ketone or aldehyde |
front 20 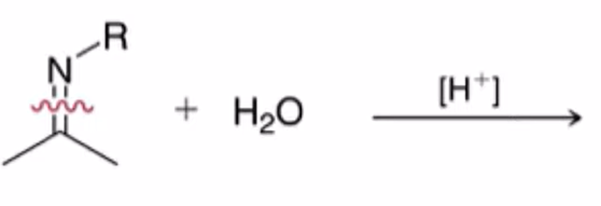 | back 20 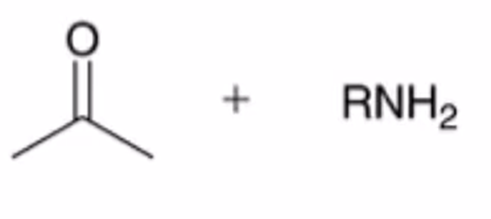 hydrolysis of imine to yield ketone or aldehyde ALSO WORKS WITH HYDRAZONES AND OXIMES |
front 21 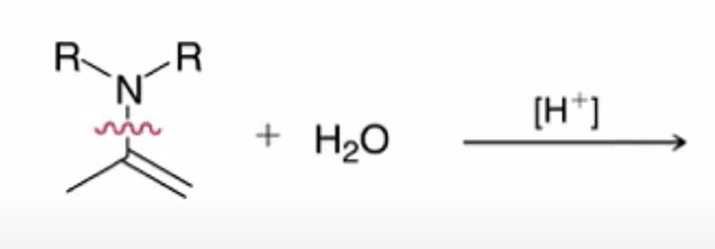 | back 21 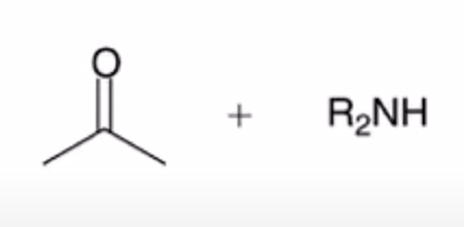 hydrolysis of enamine to yield ketone or aldehyde |
front 22  | back 22  under acidic conditions, an aldehyde or ketone will react with 2 equivalents of thiol to produce a thioacetal |
front 23  Name That Functional Group! | back 23 Thioacetal |
front 24 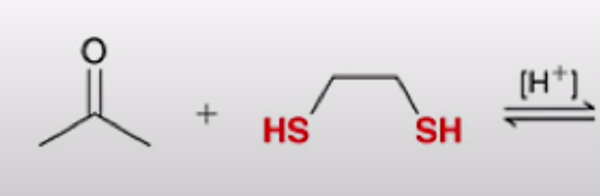 | back 24 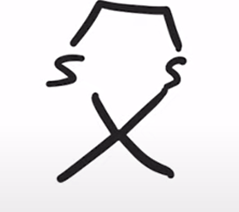 Formation of a cyclic thioacetal |
front 25 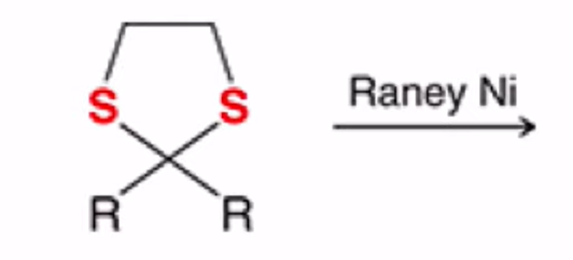 | back 25 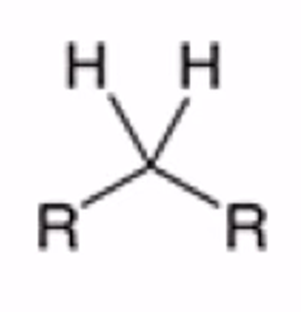 Cyclic thioacetal can be desulfurized using Raney nickel, yielding the alkane |
front 26 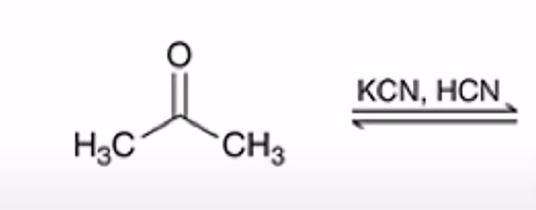 | back 26  Formation of a cyanohydrin |
front 27  Name That Functional Group! | back 27 Cyanohydrin |
front 28 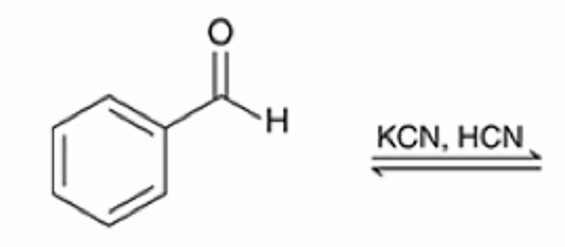 | back 28 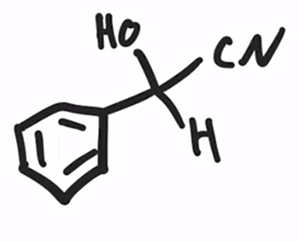 Formation of a cyanohydrin |
front 29 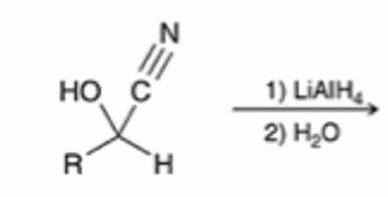 | back 29 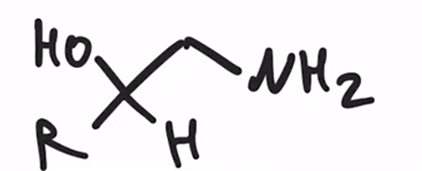 LAH REDUCTION: Cyanohydrin to primary amine |
front 30 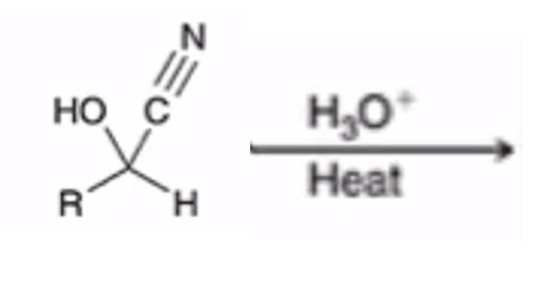 | back 30 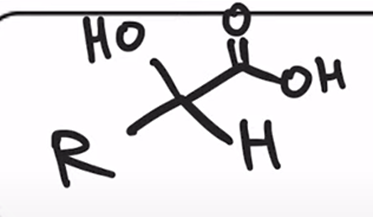 Acidification of cyanohydrin yields the carboxylic acid (oxidation) |
front 31 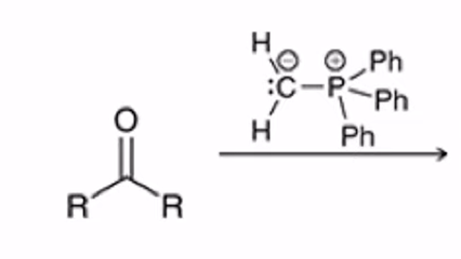 | back 31 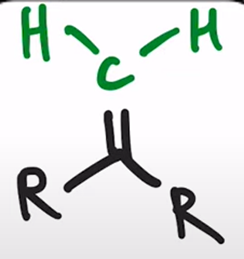 Wittig Reaction Benzene ring or electron donating group makes (E)-alkene will be preferred |
front 32 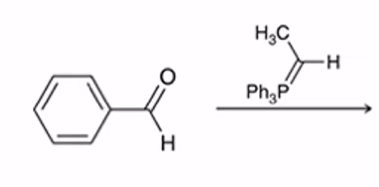 | back 32 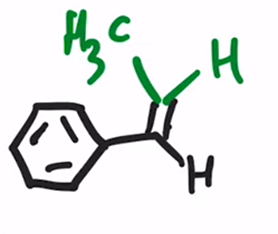 Wittig Reaction Benzene ring or electron donating group makes (E)-alkene will be preferred |
front 33 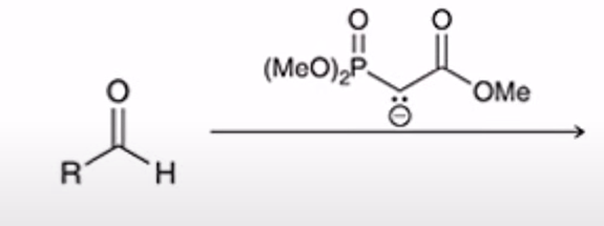 | back 33 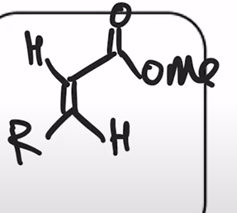 Horner-Wadsworth-Emmons Reaction (HWE reaction) phosphonate easter carbanion reagent (HWE reagent) reacts with aldehyde or ketone to yield the (E)-alkene as the major product |
front 34 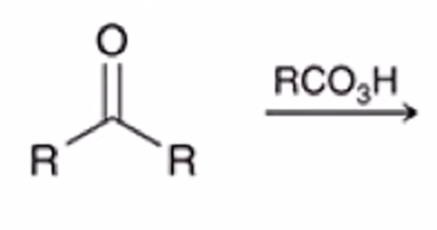 | back 34 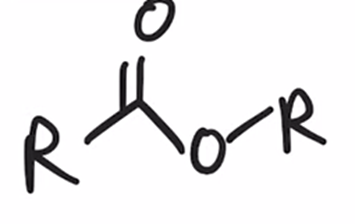 Baeyer-Villiger Oxidation Ketone converted to ester when treated with peroxy acid |
front 35 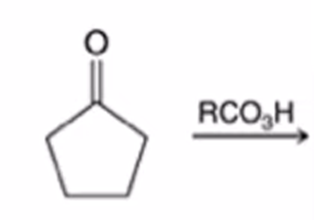 | back 35 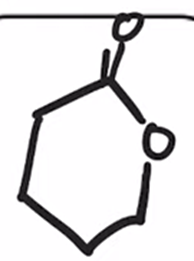 Baeyer-Villiger Oxidation Cyclic ketone to a lactone (cyclic ester) |
front 36  Name That Functional Group! | back 36 Lactone (cyclic ester) |
front 37  Name That Functional Group! | back 37 Acid Anhydride |
front 38  | back 38 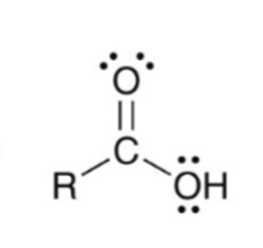 Forming a carboxylic acid via Grignard reagent and CO2 |
front 39  | back 39 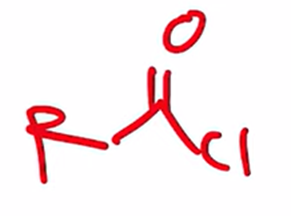 Preparation of acid chloride |
front 40 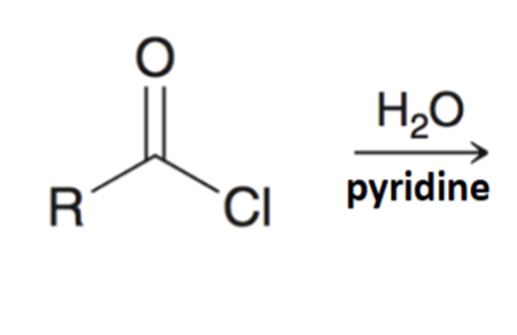 | back 40 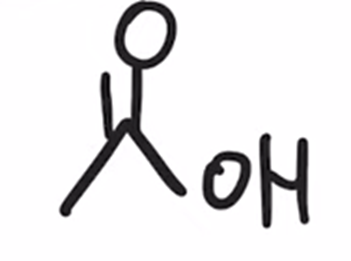 Reverse preparation of acid chloride via hydrolysis |
front 41 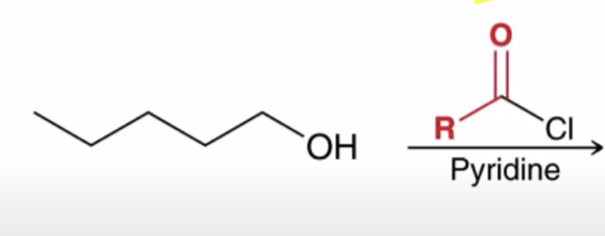 | back 41 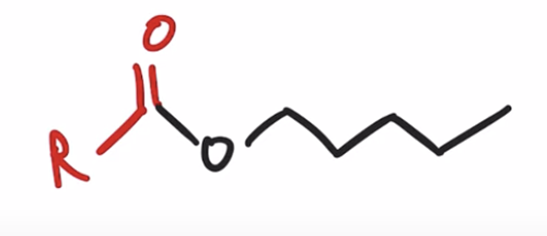 Acid chlorides react with an alcohol to make an ester |
front 42 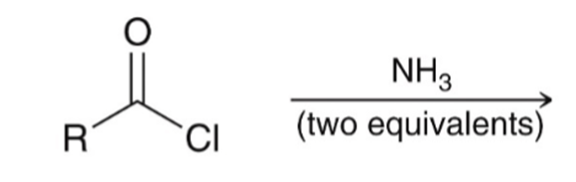 | back 42 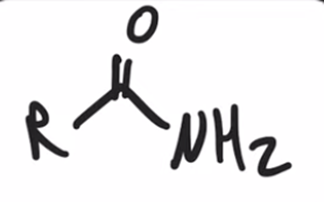 "Aminolysis" Acid chloride reacts with an amine to convert to an amide |
front 43 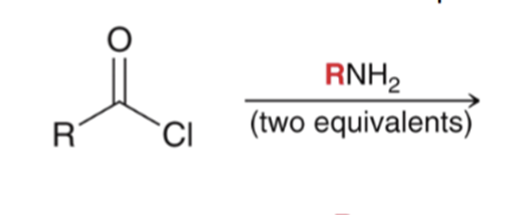 | back 43 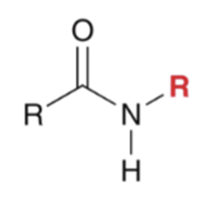 "Aminolysis" Acid chloride reacts with an amine to convert to an amide. Second equivalent of amine mops up HCl |
front 44 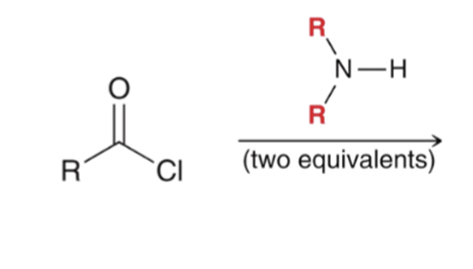 | back 44 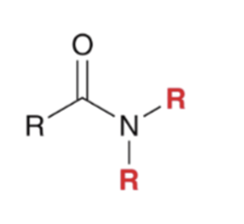 "Aminolysis" Acid chloride reacts with an amine to convert to an amide . Second equivalent of amine mops up HCl |
front 45 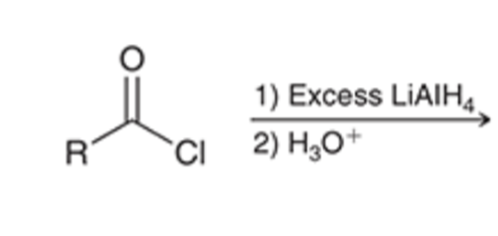 | back 45 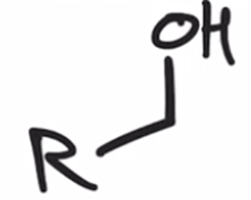 Acid chloride reduced to alcohol using LAH |
front 46 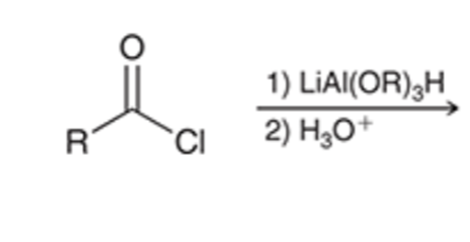 | back 46 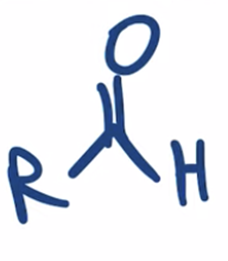 Stop the reduction of an acid chloride to an alcohol at the ALDEHYDE stage by using a bulkier hydride reagent |
front 47 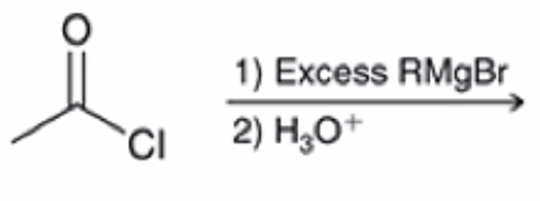 | back 47 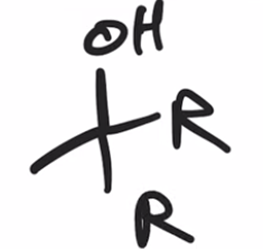 Acid chloride reacts with Grignard reagent and ADDS R GROUP TWICE IN PLACE OF Cl |
front 48 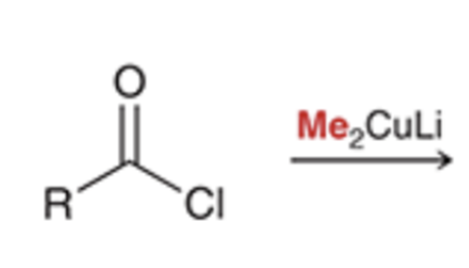 | back 48 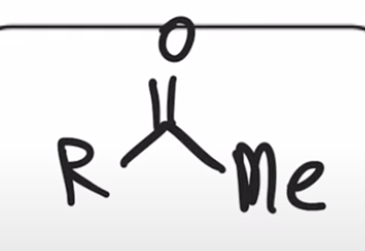 Gilman reagent replaces chloride with alkyl group |
front 49 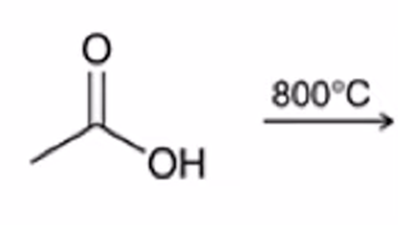 | back 49 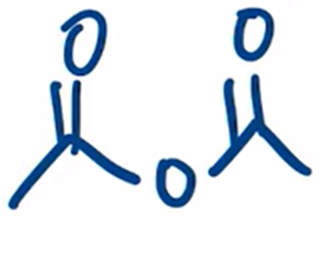 excess heating used to convert carboxylic acid to an anhydride |
front 50 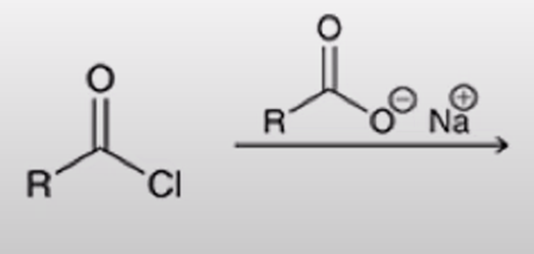 | back 50 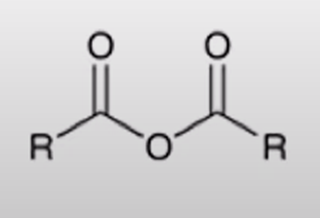 acid chloride + carboxylate salt turn into anhydride |
front 51 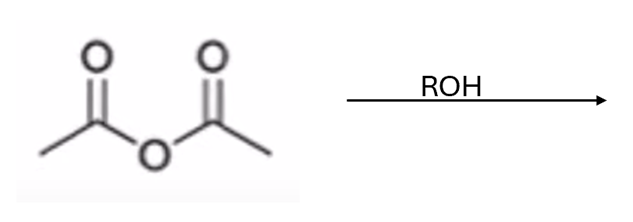 | back 51 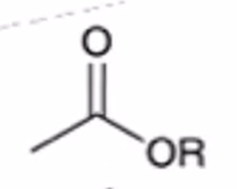 anhydride to ester |
front 52 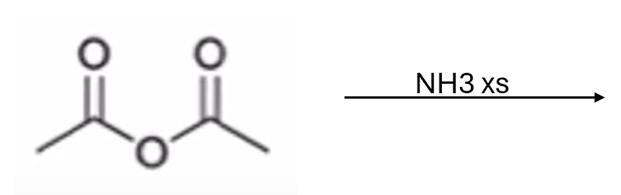 | back 52 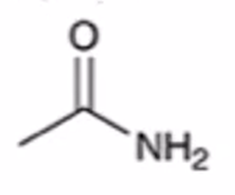 anhydride to amide |
front 53 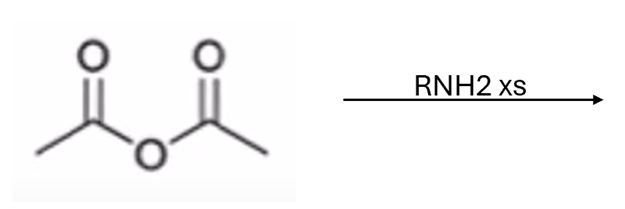 | back 53 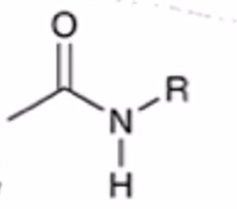 anhydride to amide |
front 54 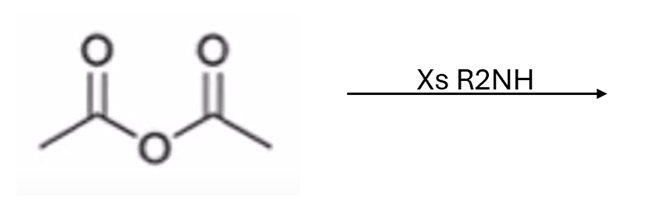 | back 54 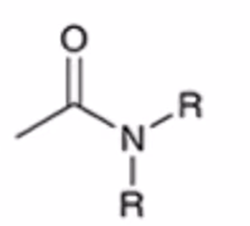 anhydride to amide |
front 55 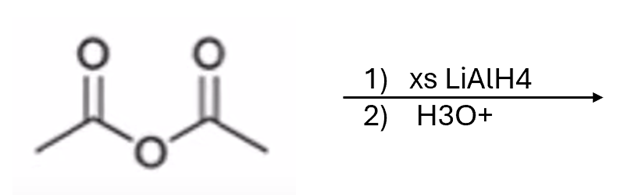 | back 55 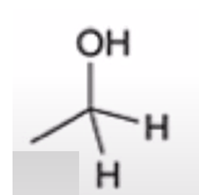 anhydride to alcohol |
front 56 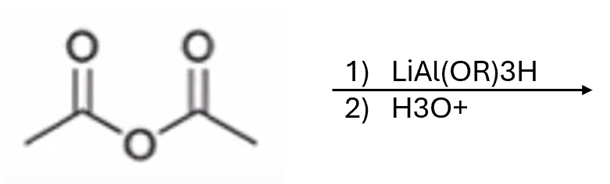 | back 56 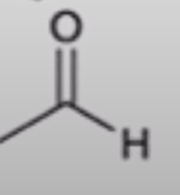 anhydride to aldehyde |
front 57 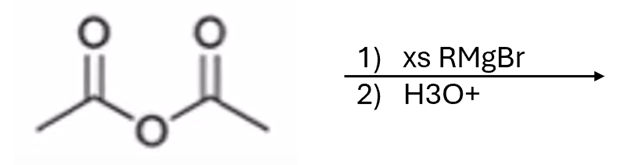 | back 57 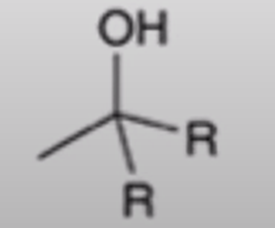 Grignard adds R group twice and creates an alcohol |
front 58 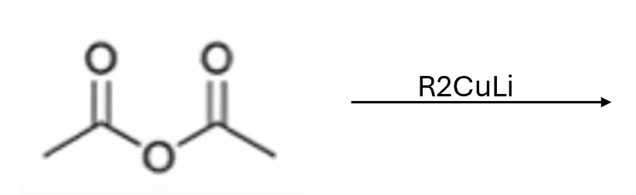 | back 58 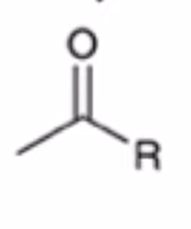 Gilmen reagent converts anhydride to ketone |
front 59 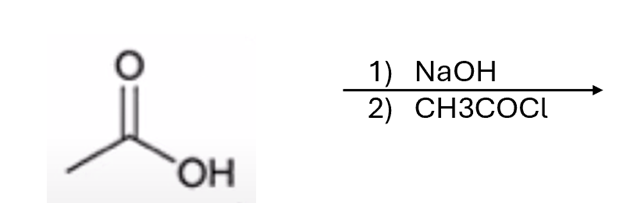 | back 59 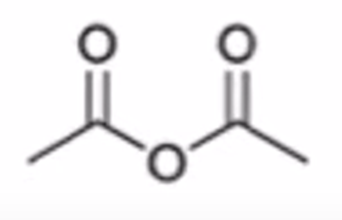 carboxylic acid to anhydride |
front 60 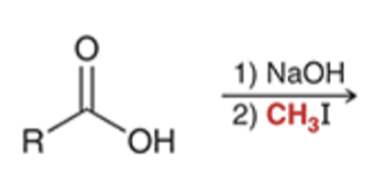 | back 60 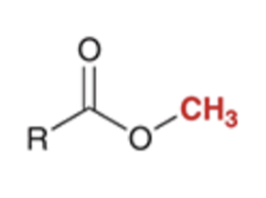 preparation of esters via SN2 reaction |
front 61 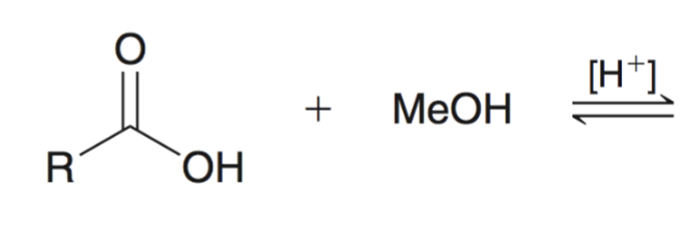 | back 61  Fischer Esterification |
front 62 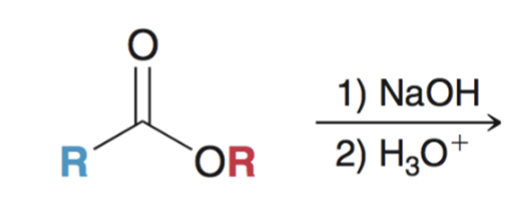 | back 62 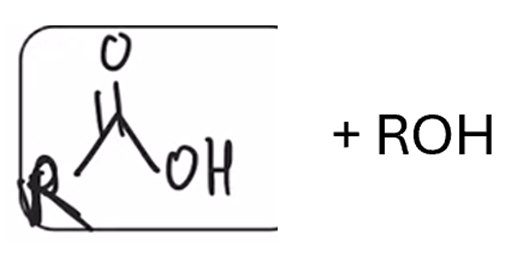 Saponification (hydrolysis using basic conditions) converts ester to carboxylic acid |
front 63 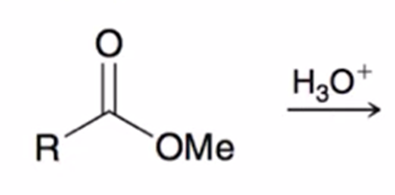 | back 63 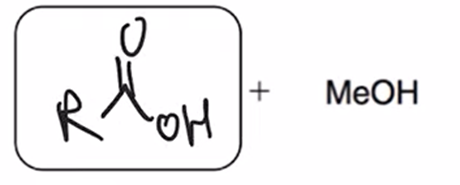 acid hydrolysis (reverse of Fischer esterification) coverts ester to carboxylic acid |
front 64 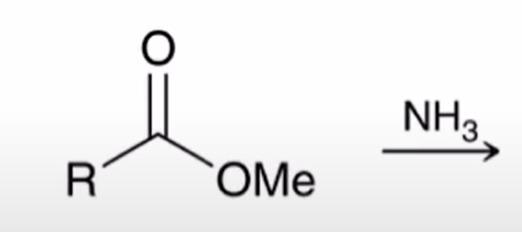 | back 64  Aminolysis of esters converts ester to amine it is slow and has little synthetic utility |
front 65 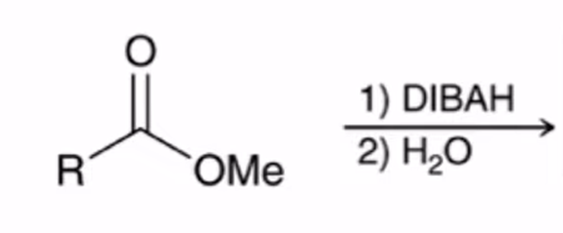 | back 65 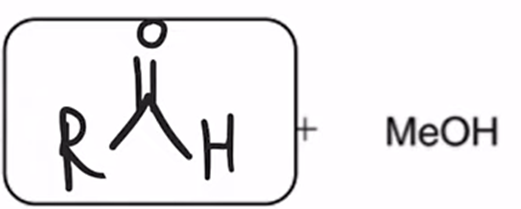 DIBAH Reduction of ester converts ester to aldehyde |
front 66 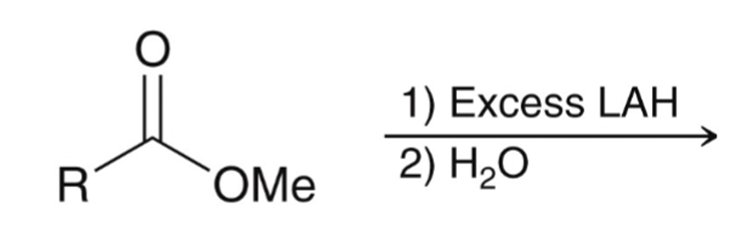 | back 66 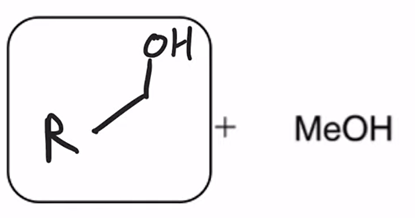 LAH reduction of ester converts ester to alcohol |
front 67 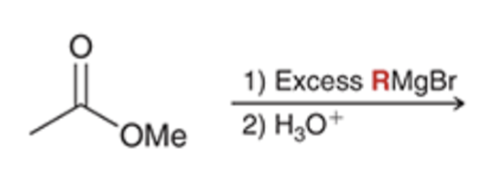 | back 67 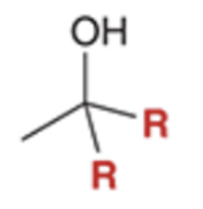 Grignard reacts and converts carbonyl to alcohol and adds R group twice |
front 68 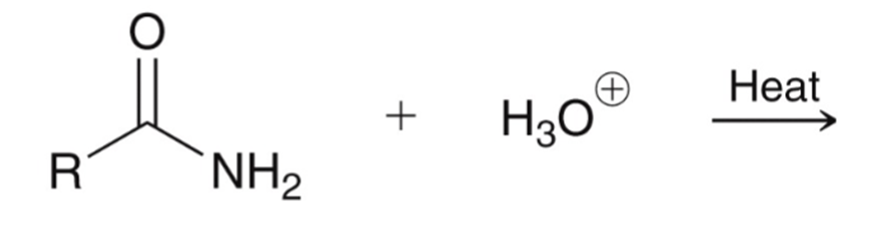 | back 68 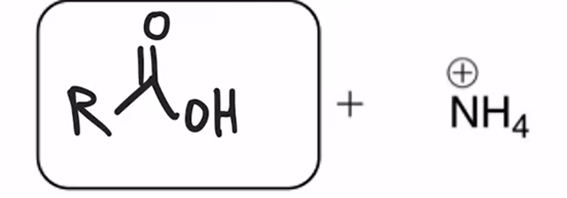 Amide hydrolyzed to a carboxylic acid |
front 69 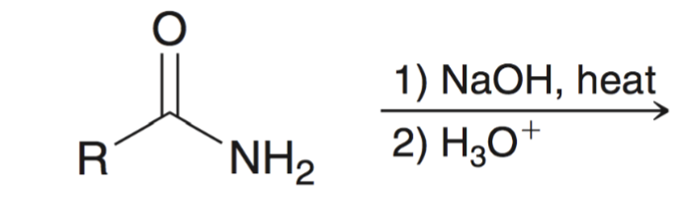 | back 69 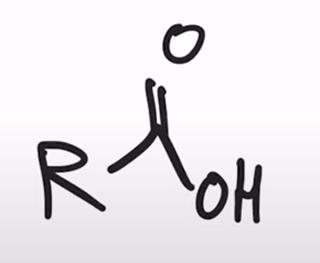 base hydrolysis of amides converts amide to carboxylic acid |
front 70 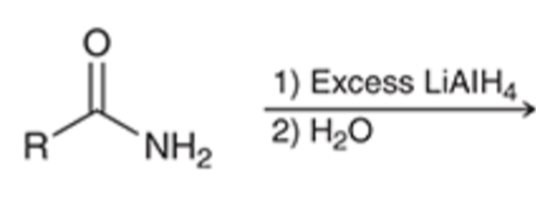 | back 70  LAH reduction removes carbonyl group |
front 71 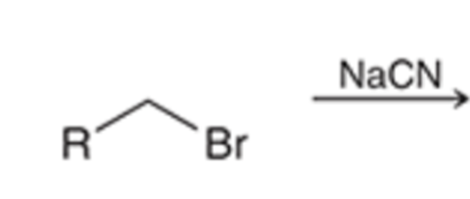 | back 71  Preparation of nitrile using SN2 (does not work with tertiary alkyl halides |
front 72 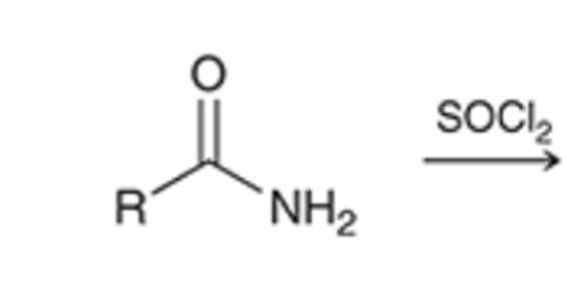 | back 72 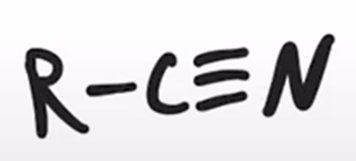 thionyl chloride converts amide to a nitrile |
front 73 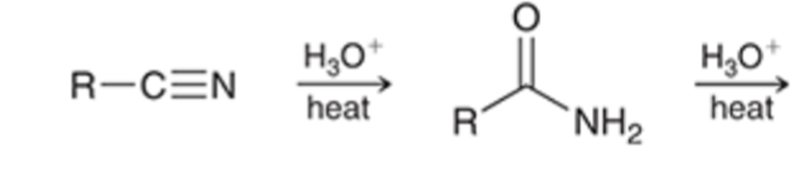 | back 73 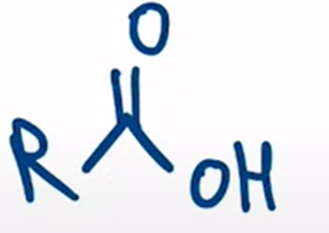 nitriles can be hydrolyzed to the carboxylic acid going through an amide intermediate |
front 74 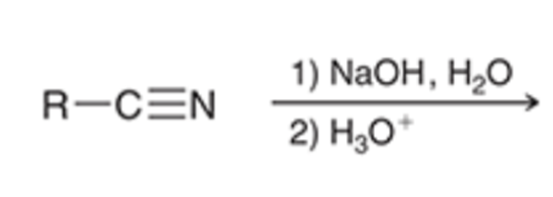 | back 74  nitriles can be hydrolyzed to the carboxylic acid in basic conditions |
front 75 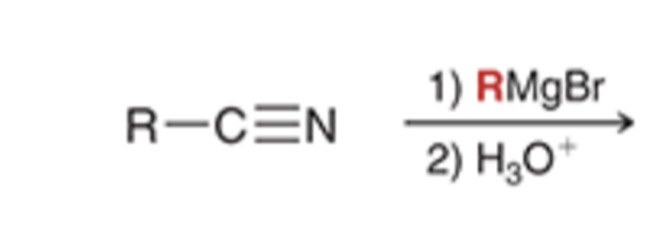 | back 75 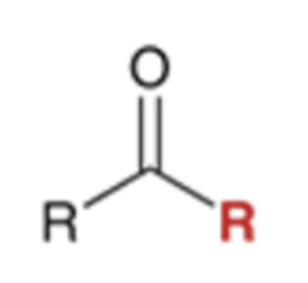 Grignard reagent converts nitriles to a ketone |
front 76 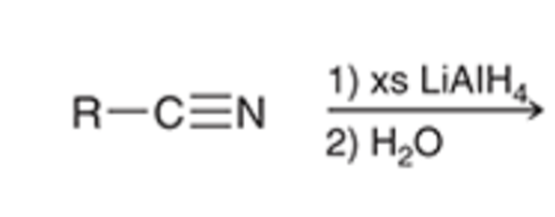 | back 76  LAH reductions converts nitriles to a primary amine |