Instructions for Side by Side Printing
- Print the notecards
- Fold each page in half along the solid vertical line
- Cut out the notecards by cutting along each horizontal dotted line
- Optional: Glue, tape or staple the ends of each notecard together
CH 18 Fundamentals of Spectrophotometry
front 1 Spectroscopy | back 1 the theoretical approach to the science of studying the interaction between matter and radiated energy |
front 2 Spectrometry | back 2 the practical application of spectroscopy |
front 3 Spectrometry uses instruments called | back 3 spectrometers |
front 4 Spectrophotometry | back 4 the method used to measure how much achemical substance absorbs light as a beam of light passes through asample solution |
front 5 Spectrophotometers | back 5 the instruments used to quantitatively measure the reflection or transmission properties of a material as a function of wavelength (a spectrum) |
front 6 Spectrum | back 6 the reflection or transmission properties of a material as a function of wavelength |
front 7 Light behaves as: (2) | back 7 a WAVE and a PARTICLE |
front 8 Light as a WAVE | back 8 - has a wavelength and frequency - exhibits the wave phenomena of interference, diffraction, and reflection. - Wave properties govern light behavior such as interference and diffraction |
front 9 Wave properties govern light behavior such as | back 9 interference and diffraction |
front 10 Light as a PARTICLE (a photon) | back 10 - carries a discrete energy that can be absorbed or emitted by a molecule. - The interaction of light with chemicals is described using the particle nature of light—the photon and its energy |
front 11 Light waves consist of | back 11 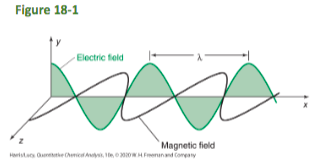 perpendicular, oscillating electric and magnetic fields |
front 12 Wavelength, λ | back 12 the distance between wave crests |
front 13 Wavelength units | back 13 m, µm, nm |
front 14 Wavenumber (v) | back 14 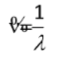 a measure of spatial frequency (v = 1/λ) |
front 15 Frequency, ν | back 15
|
front 16 Speed of light, c, formula | back 16
|
front 17 Light has a duality of | back 17 Wave-Particle |
front 18 What is the smallest amount of light that can be generated by a light source? | back 18 A photon is the smallest amount of light that can be generated by a
light source. |
front 19 The energy of a photon can be calculated from its | back 19 frequency |
front 20 Photon energy formula | back 20 Ephoton = hν where h = Planck’s constant = 6.626 × 10-34 J∙s |
front 21 Constants | back 21 c = 2.998 × 108 m/s (the speed of light in a
vacuum) |
front 22 Equations | back 22 v = 1/λ (conversion equation between wavelength and wavenumbers) λν = c (equation for speed of light in a vacuum) Ephoton = hν (equation for energy of a photon of light) E = hν = hc/λ = hcv |
front 23 Our eyes only see a fraction of the light in the universe. T/F? | back 23 True |
front 24 Spectrophotometry | back 24 Any technique that uses light to measure chemical concentrations |
front 25 Absorption Spectrophotometry | back 25 Any technique that uses the absorption of light to measure chemical |