Instructions for Side by Side Printing
- Print the notecards
- Fold each page in half along the solid vertical line
- Cut out the notecards by cutting along each horizontal dotted line
- Optional: Glue, tape or staple the ends of each notecard together
Electrolytes, Trace Elements, and Vitamins.1
front 1 What are the steps involved in Vitamin D synthesis? | back 1 7-cholecalciferol is present in the skin, upon exposure to ultraviolet light, it is transformed into the inactive form of Vitamin D. This inactive form, must first go to the liver and be 25-hydroxylated, and become 25 hydroxy Vitamin D. This then goes to the kidneys to be 1-hydroxylated to becomev1,25-dihydroxy Vitamin D (The active form of Vitamin D which will have the actions on the gut, bone, and kidneys as described above. |
front 2 What are the effects at the Kidneys for vitamin D, parathyroid hormone, calcitonin | back 2
Vitamin D: ↑ Ca reabsorption (↓ Ca excretion) Parathyroid Hormone: ↑ Ca reabsorption (↓ Ca excretion)
↓ Ca reabsorption (↑ Ca excretion) |
front 3 What are the effects on the GI Tract for vitamin D, parathyroid hormone, calcitonin | back 3 Vitamin D: ↑ Ca absorption Parathyroid Hormone:↑ Ca absorption Calcitonin: - |
front 4 What are the effects on the Bones for vitamin D, parathyroid hormone, calcitonin | back 4 Vitamin D: ↑ bone mineralization(Ca deposition into bone) Parathyroid: Hormone:↑ bone resorption(harvesting Ca and PO4) Calcitonin: Inhibited osteoclastic activity (Inhibited bone resorption) |
front 5 What is the primary difference between Vitamin D and PTH in bone Calcium metabolism? | back 5 Vitamin D stores Ca in the bones and PTH makes free Ca2+ out of the osteoid matrix |
front 6 What are the two ways that we can decrease free Calcium? | back 6 Excrete Ca in the kidneys or increase Phosphate |
front 7 How does the following substance cause hypercalcemia: Hydrochlorothiazide (HCTZ) | back 7 Reduction in renal Calcium excretion |
front 8 How does the following substance cause hypercalcemia: Lithium | back 8 Increases the body’s “set point” for Calcium |
front 9 How does the following substance cause hypercalcemia: Vitamin A | back 9 excess Activation of osteoclasts (bone resorption) |
front 10 How does the following substance cause hypercalcemia: Vitamin D | back 10 excess Increased absorption of Ca in the GI tract and decreased Ca excretion in the kidney |
front 11 How does the following substance cause hypercalcemia: Oral calcium excess (milk alkali syndrome) | back 11 Hypercalcemia, alkalosis, peptic ulcer disease, and renal impairment |
front 12 What may tell you that a person with a cough and nodule in his lungs on a chest X-ray, may have Squamous Cell Lung Carcinoma? | back 12 Elevated PTHrP levels or hypercalcemia |
front 13 A 37-year-old male who smokes 1.5 packs per day comes to the
physician’s office to get checked out. He has had a productive cough
for the past few months, but it hasn’t bothered him much. The most
troublesome fact for him is that he is a construction worker and he
has been feeling so tired lately. He has not been able to function.
The physician orders a Chest X-Ray in the office and there is a nodule
in the upper left lobe of the lung. He wants to order some tests to
check the serum Calcium. What tests would you recommend to assess for
paraneoplastic hypercalcemia (Cancer causing the
hypercalcemia)? | back 13 The correct answer is B) PTHrP. Remember this is a protein that
functions a whole lot like PTH, but it’s not. It’s production is also
unregulated by the body. This will lead to an unregulated increase in
Calcium levels. PTHrP is commonly associated with squamous |
front 14 2. A 57-year-old female presents to the clinic with the recent
history of kidney stones, despite being adequately hydrated. She just
wanted to make sure that everything was okay. She asks about the
possibility of her getting too much Vitamin D because of the frequent
boat trips that she has been taking and sunbathing. The physician
astutely responds with the statement that he’s not too worried that
the kidney stones are caused by excess Vitamin D. Why is that? | back 14 The correct answer is C) Vitamin D increases the reabsorption of Ca in the kidneys. All of these answers correctly describe actions of Vitamin D, but they’re NOT the primary reason why the physician is not worried about Vitamin D producing kidney stones. Think, the reason is because Vitamin D increases the reabsorption of Ca in the kidneys will cause less Ca to be in the tubular fluid, therefore the more likely reason for her would be either dehydration (which would increase the relative concentration of Ca in the kidneys), or a problem related to PTH. |
front 15 A 53-year-old male presents to his primary care physician with
longstanding hypertension (138/102). He is taking blood pressure
medication including an ACE inhibitor, HCTZ, Metoprolol, and a statin
for his elevated lipid levels. The physician notices that he is a
little more depressed than usual. He admits to constipation over the
past week, more frequent kidney stones, frequent vomiting, and that
has all made him unhappy. Which medication is likely to elevate his
plasma Calcium? | back 15 Answer: |
front 16 What are diagnostic criteria for: Rickets | back 16 Age: prepubescent children Abnormal Mineralization: Yes Decreased bone mass: No Bone deformation: Yes Duration: Permanent Primary pathology: Vitamin D deficiency |
front 17 What are diagnostic criteria for: Osteomalacia | back 17 Age: Adults Abnormal Mineralization: Yes Decreased bone mass: No Bone deformation: No Duration: Temporary, until corrective action is taken. Primary pathology: vitamin D deficiency |
front 18 What are diagnostic criteria for: Osteoporosis | back 18 Age: Elderly Abnormal Mineralization: No Decreased bone mass: Yes Bone deformation: No Duration: Temporary, until corrective action is taken. Primary pathology: Calcium deficiency, Sunlight deficiency Weight-bearing, activity deficiency |
front 19 What is the difference between osmolarity and osmolality? | back 19 Osmolarity of volume based (osmoles/L) and osmolality is weight based (osmoles/kg) Because most of human physiology is water-based and 1 L of water is roughly 1kg, these numbers are fairly similar when we’re talking about HUMAN PHYSIOLOGY. Outside of human physiology with other substances, they can vary drastically |
front 20 What K levels you expect in someone with chronic hyponatremia? | back 20 Low levels (<3.5 mmol/L) because the kidneys are trying desperately to hold on to any Na that it comes in contact with, at the expense of K. |
front 21 What pathology will cause all electrolytes to be elevated? | back 21 Dehydration |
front 22 Bone matrix formation has what effect on children’s serum Ca and Mg levels? | back 22 Serum Ca and Mg are both slightly higher in children |
front 23 What is the most abundant extracellular anion? | back 23 Chloride |
front 24 A patient with diabetes has an elevated anion gap. What does this tell you about the current state of his disease? | back 24 Our patient is in diabetic ketoacidosis, undergoing anaerobic glycolysis, and producing lactic acid |
front 25 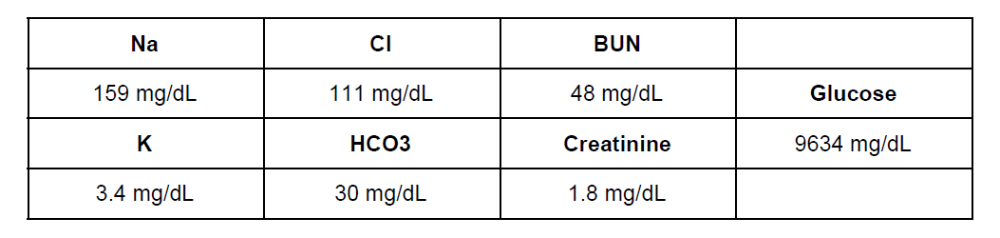 What is the underlying cause for all of her electrolyte abnormalities
is: | back 25 The correct answer is A) Ketoacidosis. She is likely a Type 1 diabetic, her anion gap is 159 - (111+30) = 18, which is mildly elevated so we must have caught it early. She has dehydration because she urinated so many times on the hike and the glucose is causing an osmotic diuresis. She has a low potassium level because her kidneys are attempting to retain all the sodium that they can to maintain her blood pressure, but that is in exchange for the potassium. Her potassium situation is actually more significant that this because of the potassium shift in acidosis. The extracellular hydrogen ions are exchanged for intracellular potassium because the cells want to help the body to handle the extra acid. Upon correction of the acidosis, her potassium may drop below 2 mg/dL. She has prerenal failure, which is consistent with dehydration. BUN/Cr ratio is ~27, which also is not too bad yet, so we’ve caught it before lots of bad things can happen. |
front 26 A 56-year-old male presents to the clinic with vague abdominal pain and hypertension (153/105). The physician asks him first about his lifestyle which reveals that he eats out 4 times a week, does not exercise, and has had high blood pressure even when he did those things. He begins to make food at home more often, limit his sodium intake, and exercise. 3 months later, he returns to the doctor and his values don’t look any better, despite his lifestyle changes. The astute physician orders a serum aldosterone just on a hunch that he might have an adrenal tumor. The aldosterone comes back elevated. What laboratory values would you expect to see in Primary Hyperaldosteronism (Conn Syndrome)? A. Na - 109 mg/dL K - 7.6 mg/dL Cr - 1.8 mg/dL | back 26 The correct answer is D) Na - 164 mg/dL K - 3.1 mg/dL Cr - 1.1 mg/dL Remember, aldosterone is the hormone that retains sodium in exchange for potassium. If you have a lot of aldosterone in your blood, then you’re going to have a very high sodium and a low potassium. The Creatinine value is not significantly elevated because of this pathology. |
front 27 How will you recognize mercury poisoning on the registry exam? | back 27 The patient will have to have some sort of exposure, and will almost always have neurologic side effects |
front 28 How will you differentiate the symptoms of Lead poisoning from Mercury poisoning on the Registry exam? | back 28 Answer: It’s going to be very hard to do that, as both heavy metals cause similar constellations of symptoms. The key to understanding the cause will be in the question stem, if they mention either Mercury or Lead, or I want you to think of Lead poisoning if they mention plasma aminolevulinic acid, whole blood zinc protoporphyrin, or free erythrocyte protoporphyrin. |
front 29 What is the most predominant side effect of Lead poisoning? | back 29 Neurologic effects like behavioral changes, headaches, clumsiness |
front 30 What are the expected total Iron, TIBC, Percent saturation, transferrin, and ferritin values for hemochromatosis? | back 30 Serum Iron ↑ TIBC ↓ % Sat ↑ Transferrin ↓ Ferritin |
front 31 What are the two tests used to assess Copper? | back 31 Total Copper and Ceruloplasmin |
front 32 What is the most likely method of analysis for Arsenic in the medical laboratory? | back 32 mass spectrometry |
front 33 A premature male newborn was seen to have anemia on his initial labs.
The plan is for him to | back 33 The correct answer is B) Copper. This deficiency is only commonly seen in premature infants and adults with malabsorptive pathologies. The presentation would include anemia, hepatosplenomegaly, and osteoporosis. Knowledge of this will help you understand the pathologies presented to you on the Registry exam, so you can get the right answer. |
front 34 A patient of European ancestry comes to the physician complaining of
heart palpitations. A. Transferrin ↑ Ferritin ↓ | back 34 Transferrin ↓ Ferritin ↑ |
front 35 A 37-year-old Female from Uzbekistan is beginning to complain of
numbness and tingling in her hands and feet. She is also experiencing
hair loss and mild anemia. Her husband makes his own bullets to
acquire dinner. She has been diagnosed with skin cancer at the young
age of 35. What trace element is the likely cause for these
symptoms? | back 35 Arsenic causes a whole slew of symptoms, but primarily gastrointestinal symptoms at low doses. Also hematopoietic, neurologic, renal, and hepatic deficiencies. Remember that chronic exposure to this compound can predispose one to skin, liver, lung, bladder, and kidney cancers |
front 36 What do we use to assess protein nutrition in patients? | back 36 Albumin, Transthyretin, Transferrin, CRP, or the Nitrogen Balance equation |
front 37 What is the physiologic action for vitamin: A | back 37 Stored in the liver, important in vision, immune function, and growth; deficiency can lead to blindness |
front 38 What is the physiologic action for vitamin: D | back 38 Synthesized in sequential steps in the skin, liver, and kidney, important in calcium homeostasis, associated with sun exposure; deficiency leads to osteomalacia |
front 39 What is the physiologic action for vitamin: E | back 39 Prevents oxidative stress that cause aging and diseases like hemolytic anemia |
front 40 What is the physiologic action for vitamin: K | back 40 Synthesized by intestinal bacteria and absorbed from leafy green vegetables; deficiency leads to prolonged bleeding |
front 41 What vitamins are found to be deficient if the patient has fat malabsorption in the gut? | back 41 Answer: If a patient has fat malabsorption, then every one of these vitamins can be decreased. It usually takes a while for the symptoms to show up because we have a few months worth of supply stored in our fatty tissues. |
front 42 A 37-year-old male is taking oral antibiotics for pneumonia that just won’t go away. | back 42 Answer: If a patient is taking long-term antibiotics, these can decrease the number of good intestinal bacteria which synthesize about half of your body’s Vitamin K which will cause you to be deficient |
front 43 Vitamin K is important in what physiologic process? | back 43 Coagulation, in German, it’s spelled Koagulation Function is to promote blood coagulation: for the coaguation factors 7,9, and 2 Trick 7, 92, blood coagulates for you |
front 44 What is the effect and deficiency for vitamin: Thiamine (B1) | back 44
Effect: Coenzyme in decarboxylation
of Deficiency: Alcoholism, Causes beriberi |
front 45 What is the effect and deficiency for vitamin: Riboflavin (B2) | back 45 Effect: Various oxidation-reduction reactions
Deficiency: Nutritional
deficiencies, |
front 46 What is the effect and deficiency for vitamin: Pyridoxine (B6) | back 46 Effect: Numerous functions
Deficiency: Uremia, malabsorption, |
front 47 What is the effect and deficiency for vitamin: Niacin | back 47
Effect: Formation of reducing substances Deficiency: Alcoholism, Causes Pellagra |
front 48 What is the effect and deficiency for vitamin: Folate | back 48
Effect: Coenzymes in various one-carbon
Deficiency: Adolescence, lactation,
or |
front 49 What is the effect and deficiency for vitamin: Cobalamin (B12) | back 49 Effect: Coenzyme necessary for hematopoiesis
Deficiency: Pernicious anemia, strict
vegan |
front 50 What is the effect and deficiency for vitamin: Ascorbic Acid (C) | back 50
Effect: Stabilization of collagen, cross-linking of tyrosine to Deficiency: Deficiency seen in low fruit and vegetable intake, known as Scurvy Excess Vitamin C is rare because it is excreted in the urine |
front 51 What is the most common causes of a deficiency in water-soluble vitamins? | back 51 Either low intake (malnutrition) or alcoholism |
front 52 What mineral is likely deficient in the case of refractive hypocalcemia that will not correct? | back 52 Magnesium |
front 53 A 47-year-old male who immigrated from South America is experiencing fatigue and a diminishing sense of touch in his fingers. He has also been complaining recently of a diminished sense of balance. The physician immediately recognizes the this person may have immigrated with Diphyllobothrium latum in his intestines. What vitamin would be deficient in this type of infection? A. A | back 53 Vitamin B12. Diphyllobothrium latum is a voracious tapeworm with an appetite for Vitamin B12. This will cause an infected individual to become deficient rather quickly. Symptoms of Vitamin B12 deficiency are neuropathies (i.e. loss of sense of balance, and sense of touch), and megaloblastic anemia. The physician would likely order a vitamin B12 level, CBC, and Folate level. The symptomatic difference between Folate and Vitamin B12 deficiencies is that Folate only produces megaloblastic anemia without neurologic effects. |
front 54 2. At 6 am, a 69-year-old female is found on a park bench one night in the middle of winter with just a T-shirt on. She is slurring her words, but has reportedly been out since 2 am. A look back in police records show that she has been picked up for similar episodes about 35 times in the past 5 years. What water-soluble vitamins do you suspect her to be deficient in? A. B1 | back 54 all of the above. Sorry I didn’t add that option on purpose, because I wanted you to think about the probabilities of each of these vitamins individually. B1 and B2 are the most likely vitamins to be deficient in this scenario of chronic alcoholism. Vitamin C is also dependent upon diet, and unless she has no access to any citrus or vegetables, it would be quite rare for people who are not stuck on a boat at sea to come down with Scurvy (Vitamin C deficiency). Vitamin B12 is a horse of a different color. Yes, it is water soluble, but we have stores in the liver that can last us for years at a time. So, one has to be deficient in B12 for many years before symptoms begin to arise |
front 55 A 58-year-old female is taking over-the-counter supplements to improve her vision and prevent skin aging to look youthful. She has been taking 700 IU/day. What vitamin is she likely taking? A. A | back 55 The correct answer is A) Vitamin A. Vitamin A primarily enhances vision, but is also is important in immune function, and growth. It has been found to decrease peroxidation of lipids, which will enhance a youthful appearance. Caution should be taken because too much of a good thing isn’t always good. Hypervitaminosis A can occur and cause, ironically, blurry vision, sun sensitivity, dry rough skin, hair loss, and confusion. Not exactly the intended effects of the supplement. This is one of the many reasons why patients should always discuss their supplement usage with their physicians |
front 56 What are the fat soluble vitamins? | back 56 Vitamin A Vitamin D Vitamin E Vitamin K Memorize ADEK, or KADE, or DEAK or something similar |
front 57 What vitamins does the liver store? | back 57 offers a place for storage for vitamins A, B12, D, E, K, and the minerals Iron, and Copper |
front 58 Which vitamin does Erythrocytes store? | back 58 Thiamine |
front 59 What do you need to know about vitamin A? | back 59
|
front 60 What is the pathology of Vitamin D (cholecalciferol) in the human body. | back 60 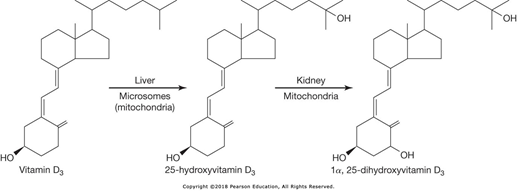
|
front 61 What do you need to know about vitamin D? | back 61
|
front 62 What do you need to know about vitamin K? | back 62
|
front 63 What do you need to know about vitamin C? | back 63
|
front 64 What do you need to know about Thiamine (B1)? | back 64
Measured by HPLC |
front 65 What do you need to know about Cyanocobalamin (B12) ? | back 65
|
front 66 What do you need to know about Folic Acid (B9) AKA folate? | back 66
|
front 67 What are reference values for some nutritional deficiencies? | back 67 Serum Albumin < 3.0 g/dL suggests possible malnutrition (Normal 3.5-5.0 g/dL) Blood Urea Nitrogen <6.0 mg/dL suggests inadequate protein intake HPLC for specific identification of vitamins |
front 68 Which organs maintain calcium homeostasis? | back 68 1.Small intestine 2.Kidneys/Liver 3.Skeleton |
front 69 What is the role of PTH in Ca homeostasis? | back 69 primary job is to increase circulating Ca ++
|
front 70 What is the role of Vitamin D in Ca homeostasis? | back 70 - primary job is to increase total body Ca ++ |
front 71 What is the role of Calcitonin in Ca homeostasis? | back 71 - primary job is to decrease circulating Ca++ |
front 72 What is the difference between the activities of Calcitonin, Parathyroid Hormone, and Vitamin D on bones? | back 72 Calciton”IN” and Vitamin D put calcium INto the bones for storage, ParathyRoID Hormone gets RID of the Calcium in the bones |
front 73 What are six causes of Hypocalcemia? | back 73 1.PTH Deficiency 2.PTH Resistance 3.Vitamin D Deficiency 4.Deficiencies in Bone Mineralization 5.Renal 6.Metastatic |
front 74 What are the mechanisms of Hypercalcemia Malignancy? | back 74 1.Tumors release PTH-related Peptide 2.Osteolytic metastases |
front 75 What happens to the total calcium and PTH with the following disorder: Secondary Hyperparathyroidism | back 75 Total calcium: Increased PTH: Increased |
front 76 What happens to the total calcium and PTH with the following disorder: Hypercalcemia of Malignancy | back 76 Total calcium: Increased PTH: Decreased |
front 77 What happens to the total calcium and PTH with the following disorder: Hypoparathyroidism | back 77 Total calcium: Decreased PTH: Decreased |
front 78 What is the definition of Electrolyte? | back 78 - small ionizable constituents in the body |
front 79 What are the intracellular ions? | back 79 potassium, phosphate, calcium, magnesium |
front 80 What are the Extracellular ions? | back 80 sodium, chloride, bicarbonate |
front 81 What is the Anion gap? | back 81 the normal gap between measured cations and measured anions, the difference represents the unmeasured anions |
front 82 What are the trace elements you need to know? | back 82 the ones that we will worry about are iron, copper, and zinc |
front 83 What should you know about Compartmentalization of electrolytes? | back 83 Intracellular fluid has a substantially higher level of potassium, proteins, organic phosphates and acids. Notes type of pressure: Oncotic pressure Osmotic pressure Hydrostatic pressure |
front 84 How does ANP/BNP effect Homeostatic regulation? | back 84 Natriuretic effects released from the cardiac ventricles |
front 85 How does Renin-Angiotensin- effect Homeostatic regulation? | back 85 Aldosterone System (RAAS):is a critical regulator of blood volume, electrolyte balance, and systemic vascular resistance released from the kidney in response to low O2 saturation in renal blood |
front 86 How does Angiotensin effect Homeostatic regulation? | back 86 released as angiotensinogen from liver, becomes ATI, then is converted to ATII in lungs, potent vasoconstrictor |
front 87 How does Aldosterone effect Homeostatic regulation? | back 87 - secreted by adrenal glands, increase Na+ reabsorption in exchange for K+ and decrease H2O excretion |
front 88 What is the formula for osmolarity? | back 88 (2*Na) + (BUN/2.8) + (Glucose/18) = Note: Normal Range: 275-295 mOsm/L |
front 89 What is the normal osmolar Gap? | back 89 5-10 mOsm/kg note: Anything above 5-10 indicates that there are osmotically active substances which differ from the ones we include in the calculation (e.g. Na, BUN, Glucose) |
front 90 What values should you be aware of for Syndrome of Inappropriate ADH Secretion (SIADH)? | back 90 Expect very low plasma osmolality (e.g. <275 mOsm/kg) Expect very high urine osmolality (>100 mOsm/kg) |
front 91 What are some common ISE? | back 91 1.Sodium 2.Potassium “valinomycin”, 3.Chloride |
front 92 What are the values that should be known for natremia? | back 92
note: Can cause hypovolemia, altered mental status, orthostatic hypotension,
|
front 93 What are the values that should be known for kalemia? | back 93 < 3.5 mEq/L, can be caused by K+ shift in treated acidosis, polyuria, diarrhea, blood dilution, mild dehydration, laboratory error, or sample collection error. This can cause low a K sample > 6.1 mEq/L, can be caused by hypoaldosteronism, extreme dehydration, hemolysis, tourniquet during collection, laboratory error, or sample collection error Critical Values - <3 mEq/L and >8 mEq/L |
front 94 What are the values that should be known for chloremia? | back 94 >108 mEq/L - Intake of Cl- exceeds the output and primarily caused by dehydration and other causes of Na+ retention. REMEMBER the chloride shifts out of cells in alkalosis < 97 mEq/L - Output of Cl- exceeds intake caused by the same list as hyponatremia with an important exception, metabolic alkalosis. This is due to GI loss of Cl- but not Na+. REMEMBER the chloride shifts into cells in acidosis |
front 95 What is Wilson disease? | back 95 Ceruloplasmin levels are very low , but there is a lot of copper deposition in the tissues. This relationship is similar to Iron and Transferrin The deficiency is in a Copper transporter in the liver which removes the Copper from circulation into the bile, so these patients can’t get rid of their Copper! Treatment is intravenous British Anti-Lewisite (BAL), D-penicillamine, or both because they will chelate the copper |
front 96 What are the two fundamental tests used to assess Copper? | back 96 Serum Copper and Serum Ceruloplasmin |
front 97 Formula for calculating anion gap? | back 97 Anion Gap = Na - [Cl + HCO 3 ] Reference range = 7-16 mEq/L |
front 98 What does the acronym MUDPILES stand for? | back 98 Common causes of metabolic acidosis with an elevated anion gap
|
front 99 What is freezing point depression used for? | back 99 for sweat chloride testing for Cystic Fibrosis in neonates we now used a PCR to find the gene. |
front 100 17-year-old Male presents to the physician with abdominal cramping 2 hours after ingesting 12 glazed donuts. What is the full medical break down? | back 100 Test: Serum Na Sample: Serum or Plasma Method: Ion-Selective Electrode Result: 121 mg/dL Disease/Physiology: Lipemia |
front 101 43-year-old Female with kidney stones, constipation, and multiple
fractures. Ultrasound of the thyroid reveals a completely
normal | back 101 Test: Serum Calcium Sample: Serum or Plasma Method: Ion-Selective Electrode Result: 11.3 mg/dL Disease/Physiology: Primary, Hyperparathyroidism |
front 102 68-year-old Male Vietnam veteran is feeling weak and tired all the time, CBC revealed macrocytic anemia. He mentions that he doesn't eat very much for breakfast, lunch, or dinner, but does drink liquor throughout the day What is the full medical break down? | back 102 Test: Thiamine Sample: Whole Blood Method: Rapid HPLC Result: 1.5 μg/dL Disease/Physiology: Chronic Alcoholism |
front 103 29-year-old healthy asymptomatic Male presents to the clinic for his annual physical. What is the full medical break down? | back 103 Test: Serum Potassium and Calcium Sample: Serum or Plasma Method: Ion-Selective Electrode Result: K+ 8.4 mg/dL Disease/Physiology: Sample Collected In Potassium EDTA |
front 104 20-year-old Female with longstanding diarrhea over the past 4 months and presents to her family medicine physician complaining of consistent bleeding from her lips. What is the full medical break down? | back 104 Test: PT/INR A prothrombin time (PT) test measures how long it takes for a clot to form in a blood sample. An INR (international normalized ratio) is a type of calculation based on PT test results Sample: Citrated Plasma (Blue Top) Method: PT/INR Result: 32 sec Disease/Physiology: Vitamin K Dependent Coagulation Factor Deficiency |
front 105 63-year-old Female gets her DEXA scan results back to measure her bone mineral density, they come back quite low. She spends most of her time in front of her computer working and eats microwaveable meals quickly so she can bet back to work. What is the full medical break down? | back 105 Test: Calcium Sample: Serum or Plasma Method: Ion-Selective Electrode Result: 7.8 mg/dL Disease/Physiology: Osteopenia Secondary To Vitamin D Deficiency |
front 106 How do we evaluate Iron status? | back 106 1. Serum Iron - specifically 3+ Iron bound to transferrin, NOT free
floating 2+ Iron |