Instructions for Side by Side Printing
- Print the notecards
- Fold each page in half along the solid vertical line
- Cut out the notecards by cutting along each horizontal dotted line
- Optional: Glue, tape or staple the ends of each notecard together
Unit 2: Instrumentation
front 1 Light behaves as a | back 1 particle and wave |
front 2 Define: Period | back 2 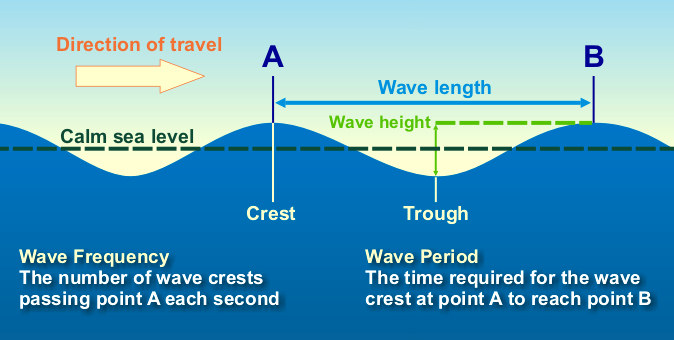 Time between passage of successive crests |
front 3 Define: Amplitude | back 3 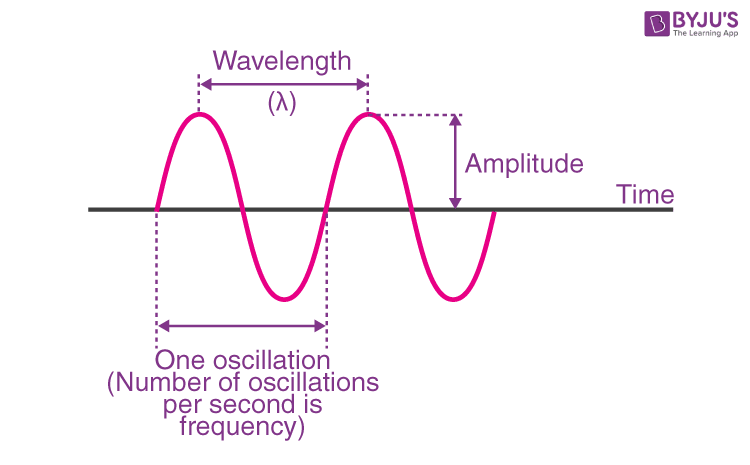 Height of peak or trough from the midline |
front 4 Define: wavelength | back 4  distance from crest to crest (or trough to trough) |
front 5 what formula should you know for Beers law equation/ | back 5 C1/A1=C2/A2 |
front 6 Beers law equation is used when you are dealing with | back 6 Absorbances |
front 7 The instrument that has a photomultiplier tube is the | back 7 Spectrophotometer |
front 8 In a fluorometer the light waves from the light source hits what first? | back 8 The diffracting gradient |
front 9 A nephelometer measures what? | back 9 Light scatter by particles in a solution |
front 10 A collimator is a device that creates what? | back 10 Parallel beams of light |
front 11 A collimator is found in what instrument? | back 11 A nephelometer |
front 12 In the mass spec, ionization refers to what process? | back 12 ions getting separated by a quadrupole |
front 13 List in order the stages within the mass spec | back 13 ionization, acceleration, deflection, detection |
front 14 During which stage in the mass spec are the ions sorted by their mass:charge ratio? | back 14 Deflection |
front 15 Gas chromatography separates compounds based on their | back 15 differential affinities for the carrier gas and the column |
front 16 What are the components of flurometer | back 16 1.Light Source 2.Diffraction Grating 3.Light Slit 4.Sample Cuvette 5.Detector (or filter) at an angle 6.Readout |
front 17 Densitometers are generally used to separate and detect | back 17 proteins in a sample/specimen |
front 18 List in order from left to right the different peaks seen on a serum protein electrophoresis | back 18 Albumin, Alpha1, Alpha2, Beta, Gamma |
front 19 Color with longest wavelength and lowest energy | back 19 Red |
front 20 Color with the shortest wavelength and the highest energy | back 20 Violet |
front 21 X rays have ___ wavelengths than IR waves | back 21 Shorter |
front 22 Define: Reflected light | back 22 light that bounces off a surface |
front 23 Define: Transmitted light | back 23 light that passes through the object |
front 24 Define: Absorbed light | back 24 light that enters but does not leave an object |
front 25 Define: Diffuse reflection | back 25 Reflection of light in all directions |
front 26 Before traveling through the sample which is being assayed, what must happen to the incident light for accurate measurements? | back 26 The incident light travelling from the light source is white light and must be split into different wavelengths for more accurate measurements. This is done with a monochromator by diffraction through a prism or diffraction grating of some sort. The adjustable aperture can move up and down to allow for the use to determine which wavelength of light will hit the sample. It is very precise and you may need to change this for each assay, so make sure that you’re using the proper wavelength for assessment. |
front 27 What is the mechanism which deflects the ions after they are accelerated in mass spectrometry? | back 27 A magnet is used to deflect the already-accelerated ions in the tube. If the ions are too big and heavy, then they will keep moving forward and hit the side of the tube with all of their momentum. If ions are too light, then the magnetic field will pull them significantly and they will hit the other side of the tube. Now, that’s how we look at ionic size. Let’s take a look at ionic charge. If the ion is highly charged, then the pull in the electric field will overcome the force of momentum, and it will hit the side of the tube. Likewise, if the ion is not highly ionized, then it will also hit the side of the tube. Only the ions with the proper mass/charge ratio will hit the detector! |
front 28 Which component of HPLC and gas chromatography allows for separation of the solutes? | back 28 The column |
front 29 Calculate the absorbance when the specimen blank reads 0.997 and the sample has a transmittance of 0.217. | back 29 Answer: Transmitted light / Incident light = %T 0.217 / 0.997 x 100 = 21.8 2 - log %T = A 2 - log 21.8 = 0.662 |
front 30 What principle do both pH and pCO2 electrodes use to detect H+? | back 30 Potentiometry |
front 31 What will impurities do to any liquid solution? | back 31 Increase the boiling point and Decrease the freezing point. Freezing point depression is really the only colligative property that we use clinically. |
front 32 Does the wide band on the right side of lane 1 correlate with the largest DNA fragment? | back 32 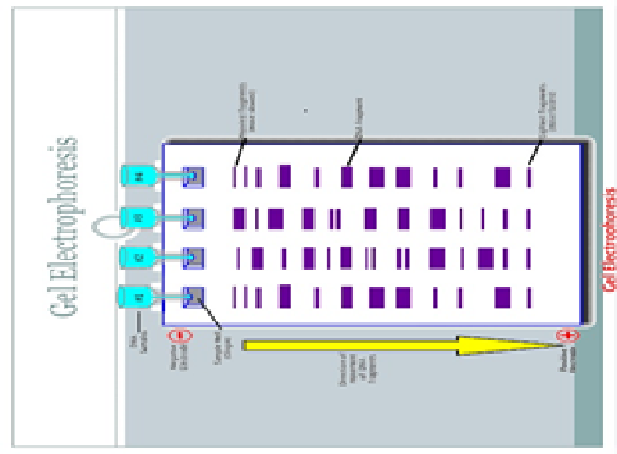 No. Don’t be fooled by the optical density (thickness) of the band. This just signifies the amount of the substance present. Even though the band size is larger to the right, the size of the individual molecules in that band are actually smaller. |
front 33 Define: Specular reflection | back 33 mirror-like reflection of light in a single direction |
front 34 what is the Absorbance equation? | back 34 A=2-log %T |
front 35 What is the equation for %T? | back 35 %T=T X 100 |
front 36 Absorbance is ____ while % transmittance is ____ | back 36 linear; logarithmic |
front 37 What does Potentiometry measure? | back 37 Measurement of the current produced by an oxidation-reduction reaction |
front 38 What do Potentiometry electrodes measure | back 38 PCO2, pH, ion-selective, reference |
front 39 Wire used in potentiometry is made of what? | back 39 Ag/AgCl |
front 40 What does Amperometry measure? | back 40 current produced by an oxidation-reduction reaction |
front 41 What does Amperometry electrodes detect? | back 41 PO2 and heavy metals Note: Clark PO 2 electrode and electrodes measuring heavy metals (e.g. Lead) |
front 42 What equation should you know for Ohms law? | back 42 V=IR |
front 43 What does Coulometry measure? | back 43 Measures the quantity of electricity by quantifying the amount of a substance that can be reduced or oxidized. The entire sample is consumed . Reported in Coulombs |
front 44 What does Voltammetry measure? | back 44 In voltammetry, there is minimal analyte consumption, as opposed to coulometry, which essentially converts all of the analyte to the new state. Voltammetry = NO analyte consumption?? |
front 45 What is Conductometry? | back 45 Measures the current-carrying potential of the solution Conductance is the reciprocal of Resistance G = 1/R Expressed in ohms ( |
front 46 What is Resistivity? | back 46 Required for CLRW To meet Clinical Laboratory Standards Institute (CLSI) requirements, resistivity must be >10M⇼cm |
front 47 What are the colligative properties of a solution? | back 47 Freezing point depression Boiling point elevation Vapor pressure depression Osmotic pressure note: Freezing point depression and Osmotic pressure are the most commonly used in the medical laboratory |
front 48 Flow cytometry is used predominantly for | back 48 hematology to evaluate Leukemias |
front 49 Flow cytometry measures what of every single cell? | back 49 size, cytoplasmic complexity, DNA, RNA, membrane-bound and intracellular proteins |
front 50 Define: Retention time | back 50 Time from injection to detection |
front 51 What does retention factor refer to? | back 51 the differential affinity of the substance to the cellulose acetate / mobile phase, used in thin layer chromatography |
front 52 what is the Retention factor equation | back 52 Rf = Solute distance / solvent distance |
front 53 What Factors affect chromatographic resolution? | back 53
|
front 54 Osmotic pressure is measured across the membrane comparing the | back 54 two chambers note: measure the height to get an accurate reading. |
front 55 What does the Nernst equation measure | back 55 the reduction potential in a cell |
front 56 In electrophoresis separation is based on? | back 56 the size and charge of the molecules (usually proteins) |
front 57 Lipophilic molecules are typically what charge? | back 57 No charge |
front 58 What do you need to know about the Nernst equation? | back 58 Need to know the result is directedly related to concentration of the reduced species over the oxidized species. Cred/Cox more reduced species the higher the electric potential will be. |
front 59 What does Valinomycin do ? | back 59 it is a selectively permeable membrane that only allows K+ to cross |
front 60 We are using a machine that has a light bulb, diffraction grating, and a detector at a 90 ̊ angle from the light path. There are no mirrors. What is this method called and how does the light hit the detector? A. Chromatography, forward scatter | back 60 Answer: |
front 61 What was the Transmittance reading on a sample with a %T of 75 and a
blank of 0.784? | back 61 The correct answer is A) 0.588. |
front 62 A pH electrode has what two special features? | back 62 The correct answer is C) Semipermeable glass membrane and Ag/AgCl internal This semipermeable glass membrane is only permeable to H+, not H2 or other funky types of hydrogen. 2H+ are able to combine with 2e- to create H2, but in the presence of a copper bar, copper has a much higher affinity for electrons than H2 does, so H2 quickly becomes 2H+ again. H+ then goes on and creates a potential difference in a hydrogen-gas electrode. |
front 63 what causes a sample to go into the prozone? | back 63 too many antibodies |
front 64 `what causes a sample to go into the postzone? | back 64 too many antigens |
front 65 What are three ways to cause interference to the immunoassay? (Hint,
there are | back 65 The most commonly-tested ones are when an antibody binds to junk,
interfering |
front 66 What method functions to separate based on differential affinity of
the solid | back 66 Adsorption |
front 67 What is the Rf of an analyte that traveled halfway down the lane? | back 67 Rf = 0.5. Just think of the Rf as a proportion of the gel that the
solute |
front 68 What has the business world given to the name of the process which
reduces | back 68 Lean Six Sigma |
front 69 A patient with a rip-roaring infectious mononucleosis infection has a negative monospot immunoassay. How is this possible? A. The sample is in prozone | back 69 he correct answer is B) the sample is in postzone. This means that the sample has too many antigens in the reaction. This is a “rip-roaring infection”, so the antigenic burden will likely be high and may kick an immunoassay into postzone, producing a false result. |
front 70 You are performing a testosterone assay via HPLC and your substance
of interest is eluting off the column and hitting the detector after
15 minutes. Since the assay only last 15 minutes, | back 70 The correct answers are A) It has a high affinity for the column, and
D) It has a low |
front 71 The clinical chemistry laboratory is audited and it is found that it
takes on average 295.3 minutes to perform an electrolyte test and
report the results. The pathologists realize that the samples are
sitting on the bench for batch testing for an extended period of time.
This was an old practice, but they have since bought a new analyzer
that does not require batch testing for electrolytes. This practice is
corrected and the average time has now dropped to 63.1
minutes. | back 71 Answer: |