Instructions for Side by Side Printing
- Print the notecards
- Fold each page in half along the solid vertical line
- Cut out the notecards by cutting along each horizontal dotted line
- Optional: Glue, tape or staple the ends of each notecard together
Unit 1 Laboratory Basics and Pertinent Lab Safety
front 1 How do you prepare a 1:24 dilution? | back 1 - 1 part sample |
front 2 What if you have too much of something? | back 2 DILUTION! |
front 3 You have 5 mL of sample and 5 mL of H2O - how many parts do you have? - What's the ratio? - What is the dilution factor? - What is the concentration of the sample after its diluted? | back 3 - 10 parts |
front 4 You have 5 mL of sample and 10 mL of H2O - How many parts do you have? - Whats the ratio - What is the dilution factor? - What is the concentration of the sample after its been diluted? | back 4 - 15 parts |
front 5 When would we use serial dilution in the lab? | back 5 when we want to access antibody concentration/strength |
front 6 7 mL of a 23 mg/dL sample is dilutes up to a final volume of 83 ml. what is the final concentration? | back 6 1.94 mg/dl |
front 7 1 mL of a 20 mg/dL sample is diluted and the final concentration is 17 mg/dl. what is the final VOLUME. | back 7 1.18 mL |
front 8 12 mL of a 0.5 mg/dl sample is diluted and the final concentration is 0.012 mg/dl. what is the final VOLUME? | back 8 500 mL |
front 9 You need 115 mL of a 4% solution. How many mL of a 10% stock solution do you need? | back 9 46 mL |
front 10 How would you perform serial dilutions to get 1:125? Which tubes would you pre fill and how much? | back 10 125 cubed is 5. |
front 11 Convert to metric: 1 Quart | back 11 0.95 liter |
front 12 Convert to metric: 1 Fl Oz | back 12 30 mL |
front 13 Convert to metric: 1 ounce | back 13 28.350 grams |
front 14 Convert to metric:1 pound | back 14 454 grams |
front 15 Convert to metric: 1 meter | back 15 3.28 feet |
front 16 Convert: Fahrenheit to Celsius | back 16 C=(F-32)/1.8 |
front 17 Convert: Celsius to Fahrenheit | back 17 (C x 1.8) + 32 |
front 18 convert: 35g/dL as mg/l | back 18 350,000 |
front 19 convert Normality to Molarity | back 19  Normality = Molarity * eq/mol |
front 20 You have 370 mL of urine in your bladder. How many liters of fluid are in your bladder? | back 20 0.370 L |
front 21 You have 5.0 mg/dL of calcium in your blood. what is the molar concentration of Ca in your blood? (1 mol Ca= 40 g) | back 21 0.00125 mol/L{M) Ca |
front 22 What is Avogadro's number ? | back 22 6.02 x 1023 |
front 23 What are equivalents? | back 23 charged particles given off from a substance |
front 24 What is the formula for Normality ? | back 24 molarity * Valence |
front 25 What is the normality of HCl | back 25 HCL = 36.5 g/mol |
front 26 What is the normality of H2SO4 | back 26 H2SO4 = 98 g/mol |
front 27 Give the instructions for preparing one liter of a 2N solution of Mg (OH)2 | back 27 Molar mass = 58 valence = 2 Normality = molarity * valence, 1*2 = 2 N |
front 28 What are the different types of percent Solutions? | back 28 (w/v)% = g of solute/100mL of solution |
front 29 Calculated osmolality from slides? | back 29 2 x [Na] + [glucose/18] + [urea BUN/2.8] + [EtOH/4.6] aka optional only used in suspected alcohol overdose. |
front 30 What is the Specific gravity of pure water? | back 30 1.0 g/mL OR 1.0 kg/L and has a SG of 1.0 |
front 31 If a solution has a SG greater than 1it will ? If a solution has a SG less than 1 it will? | back 31 sink in water float in water |
front 32 How would you prepare 100 mL of a 2% w/v solution of MgSO4 * 7H2O | back 32 NOTICE THOSE WATER IONS WILL SCREW UP THE CALCULATION. |
front 33 How do i know when to multiply or divide by the hydration ratio? | back 33 Multiply the ratio of the Hydrated Compound divided by the Anhydrous Compound Hydrated Compound / Anhydrous Compound = Hydration Correction Ratio note: The hydration correction ratio is always greater than 1. |
front 34 calculate how much you would need to weigh out to get 3.4 g of CuSo4
from a bottle containing 1000 g of CuSO4*5H2O | back 34 5.3 g |
front 35 serum glucose is ran 5 times and you got the results of 127, 124, 112, 116, 127. whats the mean median and mode. Does this patient have diabetes? | back 35 mean- 121.2 |
front 36 Find the SD of 3,4,2,3,5 | back 36  1.019 |
front 37 Compare the coefficients of variation when you have two tests. The first test had an SD of 2 and the second one a sd of 1. The mean in both cases is 10. what test has the greater coefficient of variation? | back 37 SD of 2 = (2 / 10) x 100 = 20 |
front 38 True of False we call normal or undiseased patient values "normal range" | back 38 FALSE. we call them reference intervals this is because what is normal for one hospital will be different than what's "normal" at another |
front 39 What is critical range, and what should you do with it? | back 39 is a result over a specific value, over which deleterious effects are
nearly uncertainty |
front 40 phone call for a critical value should include what 5 things? | back 40 1- ask if they are taking care of the pt |
front 41 What is the Westgard rule for 12s ? | back 41 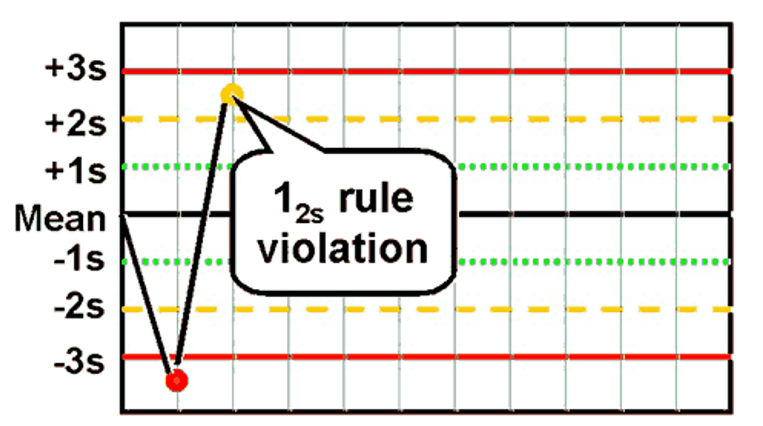 One value >2SD away from the mean (if the limits of the test are 2SD) |
front 42 What is the Westgard rule for 13s ? | back 42 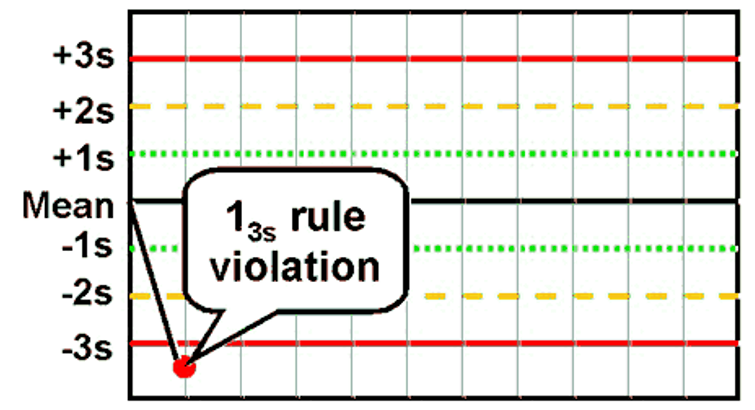 One value >3SD away from the mean |
front 43 What is the Westgard rule for 22s? | back 43 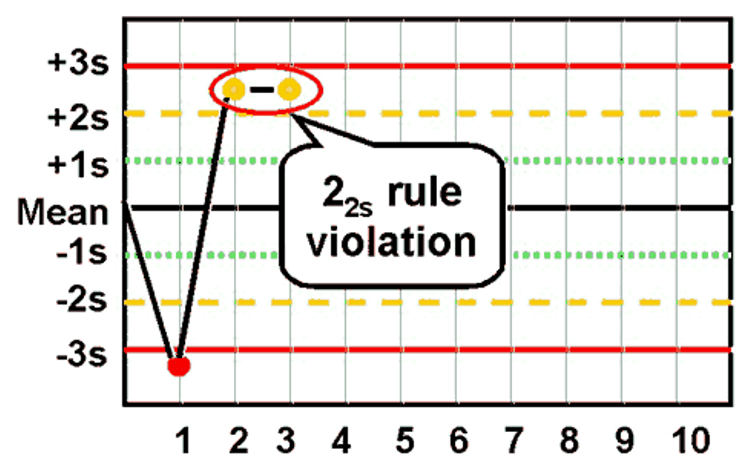 Two consecutive values exceed the same 2SD away from the mean |
front 44 What is the Westgard rule for R4s ? | back 44 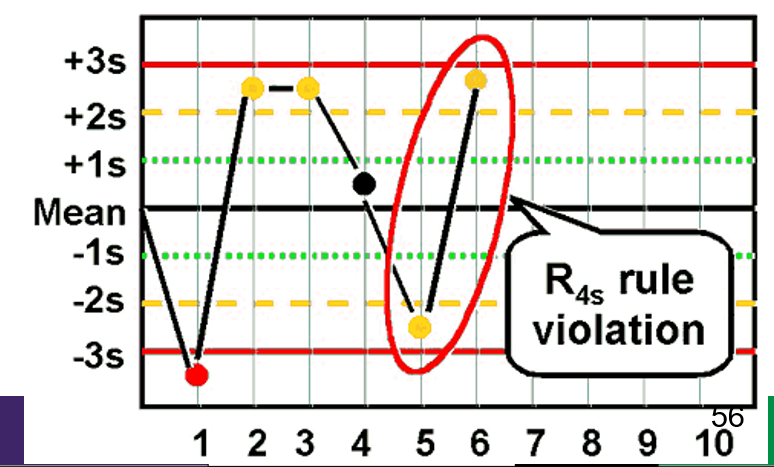 - Two consecutive values that exceed 4SD from one another |
front 45 What is the Westgard rule for 41s ? | back 45 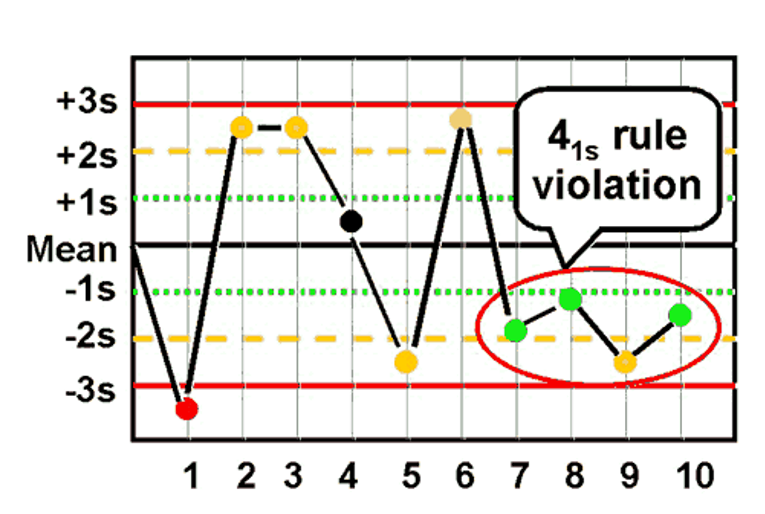 - Four consecutive values exceeding the same 1SD from the mean |
front 46 What is the Westgard rule for 10x? | back 46 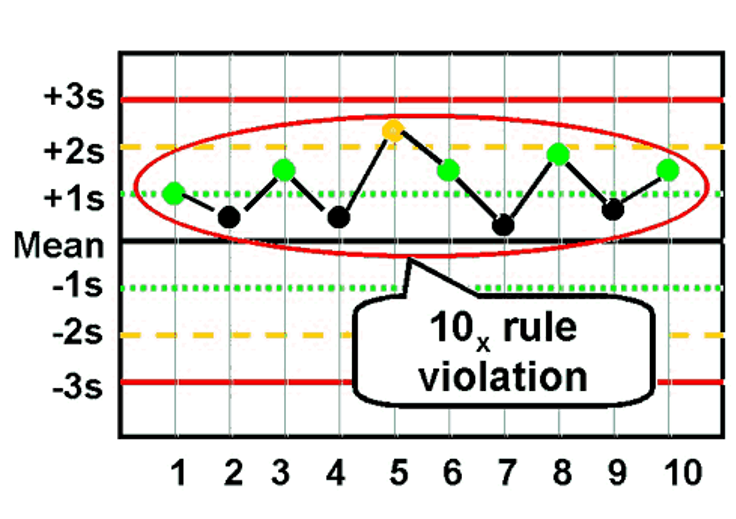 10 consecutive values on one side of the mean |
front 47 Which Westgard rule is the warning rule? ie probation. | back 47 12S |
front 48 What is a Chemical Hygiene Plan? | back 48 list of all chemicals in the lab and instructions of proper disposal. directions on maintaining safety devices. keeps record of employee training with PPE |
front 49 What is a chemical exposure plan? | back 49 list of steps that should be preformed upon exposure to a chemical |
front 50 What is a laboratory infection control plan? | back 50 designed to ensure the protection of lab employees against potential expose of blood born pathogens |
front 51 What is a laboratory exposure control plan? | back 51 designed to ensure the protection of the lab employees against potential exposure to blood borne pathogens. every lab is required to have sops for this plan. |
front 52 Name the 5 most common reasons for specimen error values? | back 52 1- Glucose drawn from an IV line infusing dextrose (Dextrose has a
higher concentration than normal glucose) |
front 53 How long are controls good for? | back 53 only good for one day or a few hours on the day of testing, but they are performed to improve reliability. |
front 54 When performing QUANTITATIVE ASSAYS how many controls are required to be ran with the patient specimens? | back 54 Two controls (high and low) |
front 55 When performing QUALITATIVE ASSAYS, how many controls do we run? | back 55 One (positive or negative) without a range of values |
front 56 What are some reasons the controls could be out? List 7 examples. | back 56 1) Bad/incorrect reagents |
front 57 What does the blue represent on the diamond-shaped hazardous materials placard. | back 57  - Health hazard blue diamond - 0-4 the higher the # the more deadly. 0 = normal material 4 = deadly Note: water= health |
front 58 What does the red represent on the diamond shaped hazardous materials placard? | back 58  - Fire hazard red diamond - 0-4 highest temps stuff will burn to lowest. the lower the temp the more dangerous. 0 = will not burn at all 4 = will burn at room temp (73 F) note:fire= red |
front 59 What does the yellow represent on the diamond shaped hazardous materials placard? | back 59  - Reactive isotopes yellow diamond. 0-4 0 = stable 4 = may detonate @ rest Note: yellow= yellow hazmat for radiation |
front 60 What does the white represent on the diamond shaped hazardous materials placard? | back 60  "grab bag for whatever doesn't fit in the other
categories" |
front 61 How is purification done to get clinical laboratory reagent water (CLRW) ? | back 61 reverse osmosis or distillation. |
front 62 Define reverse osmosis. | back 62 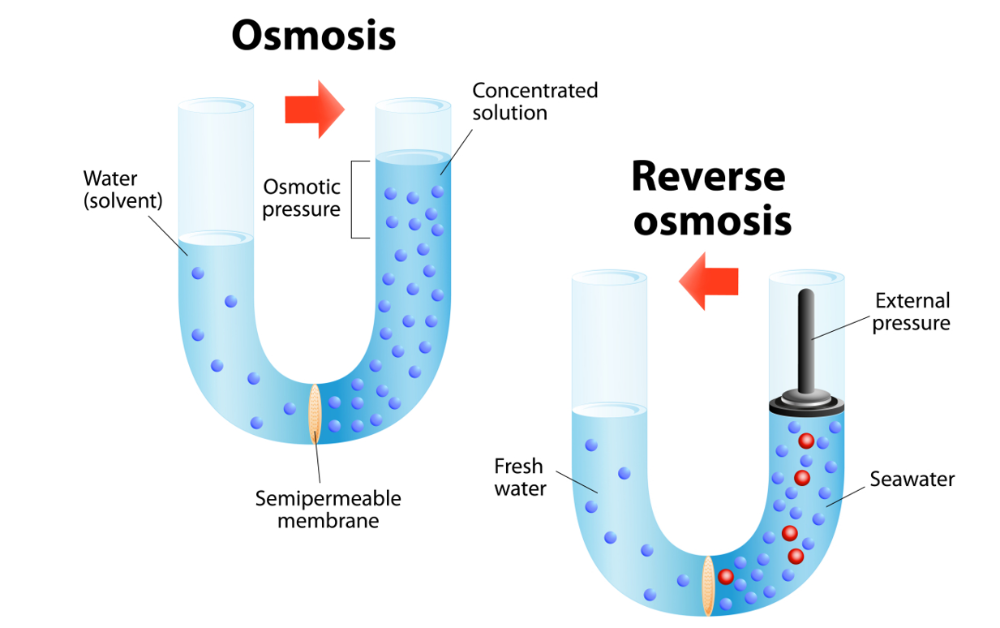 water is forced through a semipermeable membrane that acts as a filter. |
front 63 How does deionization work in osmosis? | back 63 1) reverse osmosis 2) Deionization filter water passes through ion-exchanged filters and reduces mineral content in water. 3) charcoal filters may be added to further reduce organic compounds present. 4) particulate filter" gatekeeper filter then blocks anything bigger than 0.22 Micrometers from passing through. (relatively inefficient at removing glucose or other biomolecules |
front 64 What are the 3 grades of water in clinic? | back 64 type 1 - Most pure critical laboratory applications
ex: HPLC, Gas chromatography, mass spectrometry, cell culture, and
in-vitro fertilization |
front 65 Identify the 5 clinical laboratory reagent water minimum standards. | back 65 1) resistivity - @ 25 C > 10 mega alms (too much means a lot of
ions to conduct electricity |
front 66 What are the types of plastic and what are there uses? | back 66 PolyProPylene - Plastic Pipette tips, flexible or rigid, chemically resistant. PolyeThylene - Test tubes, bottles, stoppers, racks. May (e)bsorb proteins, dyes, stains, and picric acid. PolyCarbonate - Centrifuge tubes, graduated Cylinders, and flasks. Usable temperature range is -100˚C to 160˚C. Not for strong acids, strong bases, or oxidizing agents. PolyStyrene - Rigid, should NOT be autoclaved. Will crack and Splinter when crushed. Not resistant to most hydrocarbons, ketones, and alcohols. |
front 67 What are the different types of glass ware? | back 67 Low actinic glass, used in control material, calibrators, and reagents high thermal resistance, with red color added to provide protection from light and still permit visibility Regular glass, used in old, decrepit houses, and possibly some outdated beakers and flasks Low thermal resistance, no/minimal protection from light, break easily |
front 68 What are the two categories of pipettes? | back 68 1) Transfer - volumetric 2) Measuring - graduated or serologic |
front 69 What are the 3 subclassifications of pipettes? | back 69 To contain (TC) - how much the pipette holds, Class A is the most accurate To deliver (TD) - how much the pipette actually delivers To deliver/blowout (TD/blowout) - requires an extra step which may introduce some inaccuracy |
front 70 What is a serological pipette used for ? | back 70 to transfer amount of sample between 2 lines. |
front 71 What is a volumetric pipetmost used for? | back 71 most accurate in measuring volume. bubble in the center. T/C. |
front 72 What is air displacement micropipettes used for? | back 72 uses a piston to displace air and facilitate aspiration and ejection
of the sample. ex- disposable pipette tips. |
front 73 What is a positive displacement and micropipettes used for ? | back 73 uses a capillary tip made of glass or plastic with a teflon coated plunger. requires washing to flushing between samples. |
front 74 What is the largest size of a particulate that could be in any grade of CLRW? | back 74 0.21 micro meters |
front 75 if a patient has a positive result does that mean they have the disease. | back 75 NO. we have true positives but it can also be a false positive. Be more specific with test types. |
front 76 What an explosive compound used in the laboratory similar to TNT? | back 76 Picric acid - caution when using glass jars with evaporated picrate on lid. associated with Jaffe reagent for creatine testing |
front 77 what is a swinging-bucket rotor? | back 77 allows sample to move in a horizontal position creating a solid pellet at bottom of tube. |
front 78 What is a fixed - angle rotor centrifuge? | back 78 centrifuges at angles between 25-52. during centrifuge particles move down the sides of the tube and form sediment. designed for much faster speeds and forces |
front 79 What is an ultra centrifuge? | back 79 MUCH larger, used to fractionate lipoproteins, drug-binding assays, and prepare tissue. |
front 80 If you have a single pellet at the bottom of the test tube, what centrifuge was used?- | back 80 a swinging bucket rotor |
front 81 What the 4 types of chemical grades? | back 81 1 - analytical grade : the most pure substances available, list all
impurity's |
front 82 What was the purpose of Resource Conservation and Recovery Act? | back 82 If you see an answer that has to do with proper use and conservation of any resource that you use in the labs, that’s going to be the right answer. We don’t need to go into these in depth, because they make up such a miniscule portion of your test and there are so many regulations, that you would put in so much effort for such little benefit. |
front 83 What is the most important factor in infection control and chemical hygiene? | back 83 Washing your hands after you take your gloves off. The use of antibacterial soap is NOT as important as the physical act of washing your hands with water. |
front 84 Juan is transferring two saliva samples, housed in their containers,
and packaged | back 84 He is walking into the laboratory with a POTENTIALLY hazardous substance. If he is only taking it over to the lab, he should put on gloves. If he is performing the test, then he will need a lab coat, possibly a respirator if they are performing Tuberculosis testing. If this sample is to be assessed for a suspicion of Tuberculosis, then it should be transported with special flags. |
front 85 Jane, the new lab tech, accidentally spills an old bottle of acid in
the lab and is | back 85 SDS in the laboratory. Sure, she could roll the dice and try to find a website, but unless her google-fu is strong, she is likely to find some misleading information. |
front 86 Friederich is a German exchange student in Medical Laboratory
Sciences. His | back 86 Jaffe reagent and he is using picric acid |
front 87 1. A 37-year-old male walks into a bar, he starts a bar fight, ya
know, the kind you only see in movies. Chairs are breaking, liquor is
flowing all over the floor, and women and children are running for the
hills. He decides that he needs a cigarette because he forgot to put
on his nicotine patch before he started this brawl. He ends up being
irresponsible and doesn’t dispose of his cigarette butt properly and
was acting like he hadn’t done anything irresponsible up to that
point. The butt was thrown at an opponent behind the bar, which
immediately went up in flames | back 87 The correct answer is B) A, B, dry chemical. This would be considered a class B fire because of all the oozing booze on the floor. Remember, flammable liquids kick the classification up to class B. There are also elements of ordinary burning material, like various linens, wood, the bar, and chairs are probably made of wood. If there is metal in the chairs, then it may or may not combust, but the firefighters are prepared for that if it should happen. |
front 88 What policy in the alphabet soup below provides guidelines for
general laboratory safety and exposure control for
laboratorians? | back 88 The correct answer is B) CLSI. The Clinical Laboratory Safety Institute was previously called the Clinical Laboratory Improvement Act (CLIA), so you may hear that term as well. If you do, they mean the same thing. |
front 89 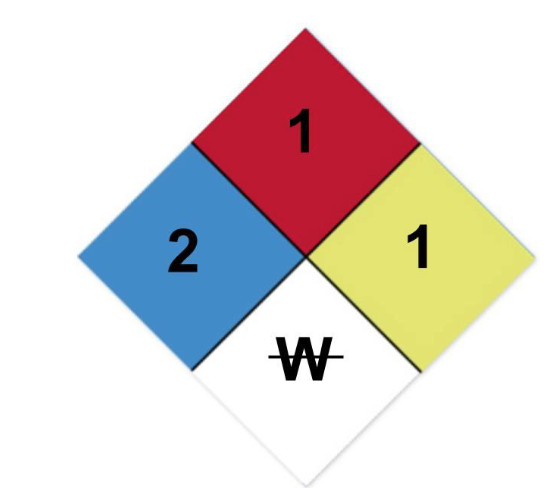 Correctly interpret the hazardous materials placard with the given
labels: | back 89 correct answer is C) Hazardous is the 2 in the blue square, Flash
point >200 ̊F is |
front 90 Testing for serum albumin, the first value is 2SD above the mean, and
the next | back 90 The run should be rejected because of the R4s rule. |
front 91 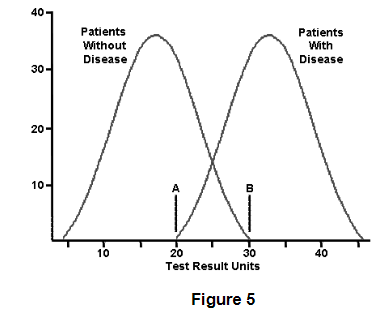 What patients with a plasma HCO3- of 23 mg/dL have acetaminophen toxicity according to Figure 5? | back 91 10 patients do have acetaminophen toxicity when the disease is at 23 mg/dL. 20 patients do NOT have acetaminophen toxicity at 23 mg/dL. Roughly 2/3 do NOT have the disease at the same level that 1/3 of patients with acetaminophen toxicity do have the condition. You can never be too careful when interpreting test results because some values can exist in this normal/disease curve overlap. |
front 92 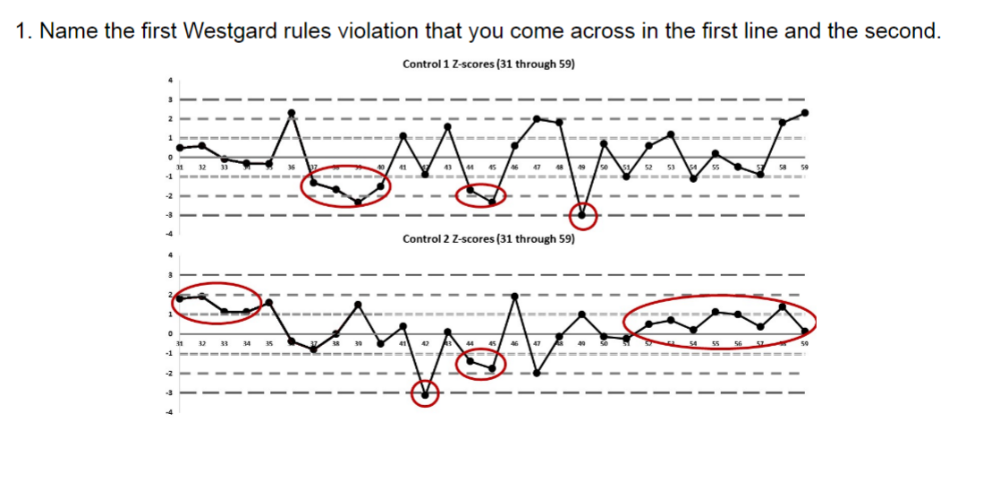 Name the first Westgard rules violation that you come across in the first line and the second. A. 13s | back 92 The correct answer is C) 41S, because you see there are 4 values in a row above the 1SD line. This might suggest a shift or a drift. The proper procedure would be to reject the QC run and rerun the samples. Then if they are still out, consider grabbing different bottles of standards |
front 93 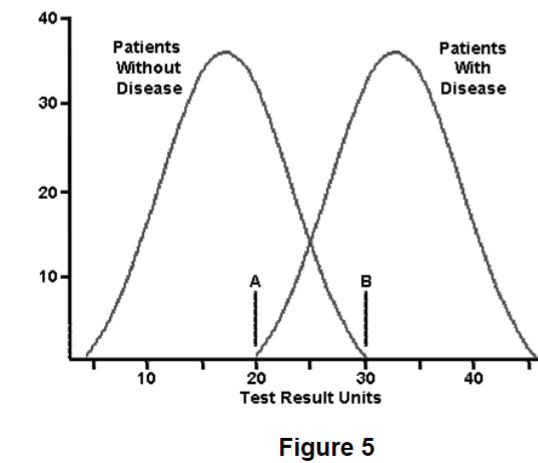 A 52-year-old male patient with diabetes has an glucose level of 125 mg/dL. He has been working on his diet and exercise over the past few weeks and takes aspirin, metoprolol, and lisinopril religiously. His prior blood sugar was 140 mg/dL one month ago. A blood sugar of 126 mg/dL is diagnostic of diabetes, where do you think he falls on the disease reference curve seen in Figure 5 right now? A. Far left of the normal reference curve | back 93 Answer: Even though 126 is technically diagnostic of diabetes, this patient has been steadfast in performing lifestyle modification to reduce his risks for diabetic complications. This is the likely reason why the fasting plasma glucose came down to 126 mg/dL. That value is technically considered negative for diabetes, but in the clinical context the physician needs to watch this patient closely over the next few weeks. He is still hyperglycemic, just not as much as he was last month. He might be diagnosed with insulin insensitivity. |
front 94 You begin running QC on a chemistry analyzer, but as you look at the
Levy-Jennings chart, you notice something awful, you will be forced to
start your assay over again. Breaking which of the Westgard Rules
would NOT force you to do this? | back 94 The correct answer is A) 12s. This is the initial test before moving on to any other tests. If there is not a single value >2SD, then you can accept the run. 68.2% of the time, this will be the case. For the other 31.8% of the time, when the value exceeds 1SD, then we go down the laundry list of other Westgard Rules we need to follow. Basically, if you have a standard that is >2SD, then it flags us to a potential problem. That is okay if it happens once, but we need to keep our ears to the ground because if anything was fishy, then if something is wrong or is getting worse, then it will likely break one of the other Westgard Rules on the next run. If you are back within 2SD, that means that the value was just an outlier. 4.2% of the time, the value will be between 2-3SD from the mean value ... and that is completely fine. We just have to make sure that weird values like that don’t continue |
front 95 A patient comes into the emergency room with a broken arm after
falling off the | back 95 The sample is likely lipemic because he just ingested an entire
burger and a |
front 96 A tube of blood comes from the ED for stat chemistry testing. You see
that it is | back 96 Granted, crazy things happen in the ED, so it’s not easy to get 5 mL of blood on each patient. The issue is that depending on which vacutainer tube was used, it could have an anticoagulant in it that will interfere with the testing at such high concentrations, because now the proper anticoagulant :blood ratio has been thrown off. In all likelihood, I think this sample could still be ran with a note on the result informing the doctor what happened with the sample. They’re used to this sort of thing and know how to interpret it |
front 97 1 mL of a 50 mg/dL sample is diluted up to a final volume of 15 mL. What is the final concentration? | back 97 3.33 |
front 98 What is the normality of 1 M HCl? | back 98 1 N HCL because there is 1 equivalent in 1 mol of HCl |
front 99 A 37-year-old male feels extremely hot in his Arizona apartment, but
looks at the thermometer and realizes that it’s a chilly 42.4 ̊. He
immediately seeks medical treatment because he is sweating and it’s so
cold. What is most likely to have happened and how hot is it
really? | back 99 The correct answer is A) He didn’t look closely at the units. 42.4 ̊C is 100 ̊F, which is pretty hot. |
front 100 What is the molarity of 3.7 mg of HCL in 0.7 L of water? GMW of HCL = 36.46g/mol | back 100 The correct answer is 0.00014 M. |
front 101 What is the final concentration if 1 L of a 3.5 mg/dL NaCl solution is diluted in 2 dL of water? | back 101 The correct answer is 2.9 mg/dL |