Instructions for Side by Side Printing
- Print the notecards
- Fold each page in half along the solid vertical line
- Cut out the notecards by cutting along each horizontal dotted line
- Optional: Glue, tape or staple the ends of each notecard together
Lab Exam 2
front 1 three chemical structures for unsaturated hydrocarbons | back 1 alkenes alkynes |
front 2 What color changes did you observe when you brominated ethene? What type of reaction was that? | back 2 The solution experienced a color change to dark red |
front 3 Write a chemical reaction for the conversion of benzene into bromobenzene by the reaction with bromine in the presence of iron (III) chloride. What type of reaction was that? | back 3 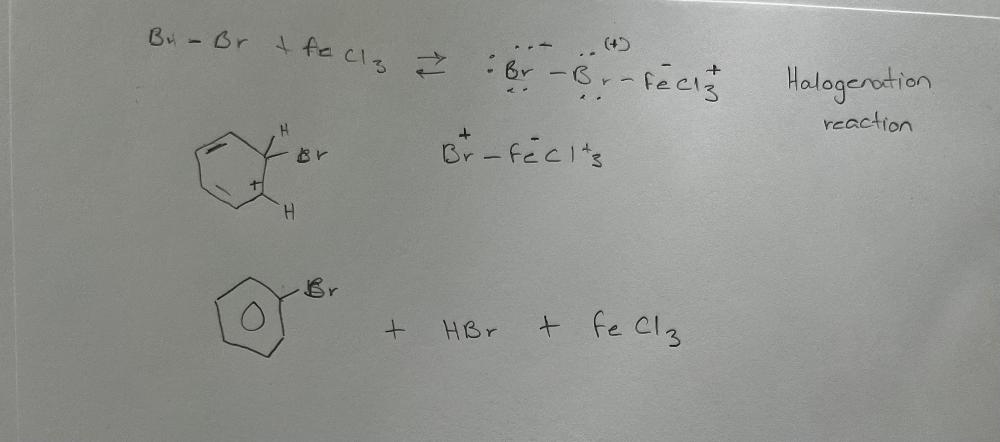 |
front 4 What happens when you react cyclohexene with bromine and cold potassium permanganate? | back 4 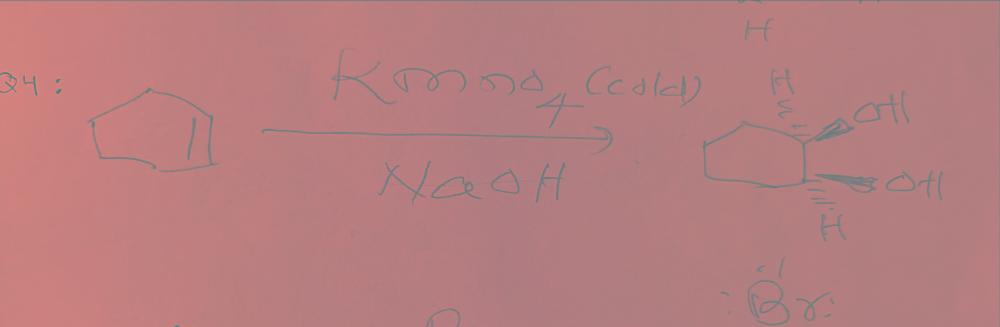 |
front 5 Write a chemical equation for the reaction of acetylene with 2 mols of bromine in the presence of a solvent carbon tetrachloride? | back 5 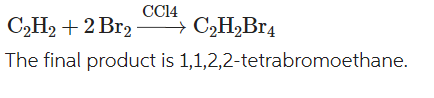 |
front 6 What is the main safety concern about hydrocarbons? | back 6 Hydrocarbons are highly flammable liquids and vapor. It is also fatal if swallowed. |
front 7 Why should bromine be handled with care? | back 7 Bromine is corrosive and the dark vapor of bromine can irritate eyes. |
front 8 Where should all reactions for this lab (reactions of hydrocarbons) take place. | back 8 It should take place in the fume hood. |
front 9 product of reaction of given hydrocarbon and bromine | back 9  |
front 10 What visual observation would you expect if you ran the reaction of the above hydrocarbon with bromine? | back 10 When bromine is added to 2-butene, a light brown color would occur. After, the reaction is completed the brown color would completely fade (meaning the bromine was completely consumed by the alkene). |
front 11 what precautions should one use when working with H2SO4 and H3PO4 | back 11 Gloves, closed toed shoes, lab coat, goggles because it can burn skin. |
front 12 Calculate the theoretical yield | back 12 no data |
front 13 when 2- butanol undergoes E1 dehydration, three alkenes are obtained. Draw the structures for these alkenes. Which is the most abundant. | back 13 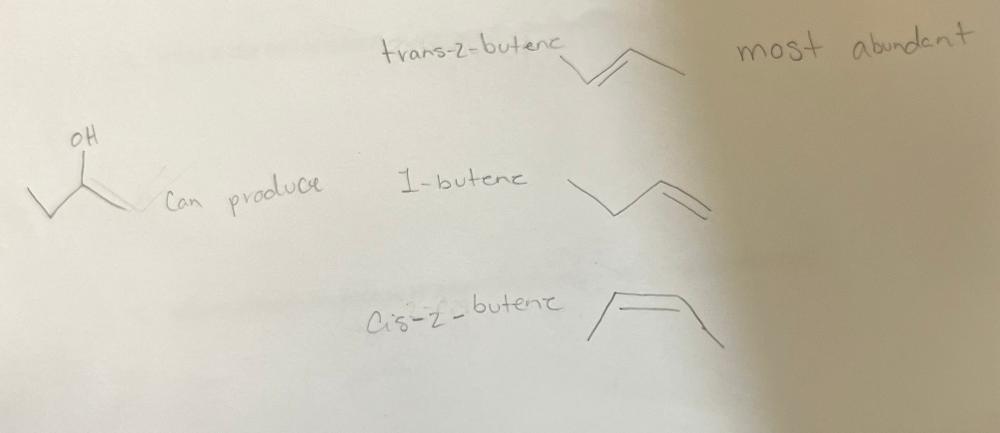 Most abundant is trans 2-butene |
front 14 Write a chemical equation to convert cyclohexanol into cyclohexene in the presence of an acid catalyst. | back 14 C6H11+H3PO4----^C6H10+H2O |
front 15 between ethanol and tert-butanol which alcohol dehydrates at much faster rate? | back 15 Tert-butanol will dehydrate faster because it is a tertiary alcohol. |
front 16 What is the role of a catalyst during dehydration? | back 16 The role of a catalyst during dehydration is to drive the reaction in one direction only and speeds up the reaction. |
front 17 Why acid catalysts such as HCl and HBr are not used during dehydration? | back 17 Because their conjugate bases are good nucleophiles that can cause a substitution reaction to occur. |
front 18 What precautions should one use when working with potassium hydroxide? | back 18 Plastics and some rubber might be affected by the potassium hydroxide. |
front 19 why is it important that the number of moles of 2 naphthoxide ion be the same as the number of moles of iodoethane used for the synthesis of nerolin? | back 19 We use the same number of moles for both because in the synthesis of nerolin the molar ration of naphthoxide to iodoethane is 1:1. This ensures a that no extra reactants are found after the reaction. |
front 20 What effect would not properly drying the filtered nerolin have on your results? | back 20 This would affect your experimental and percentage yield. |
front 21 Identify the nucleophile and leaving group for the reaction | back 21 2-naphthoxide acts as the nucleophile and Iodoethane as the leaving group. |
front 22 Write a chemical equation for the formation of nerolin from 2-hydroxynaphthalene | back 22 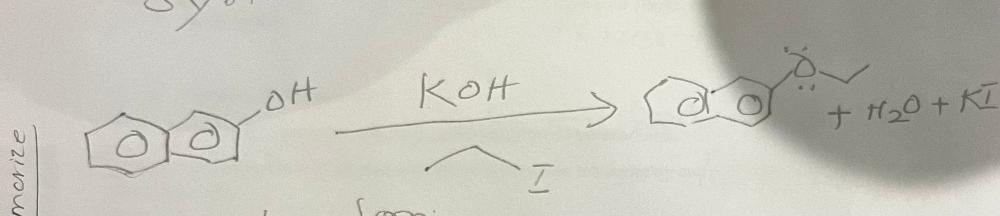 |
front 23 write a rational mechanism for the formation of nerolin | back 23 paper |
front 24 What is nerolin used for | back 24 it is used for perfumes, as a stabilizer in powders. |
front 25 calculate percent yield: | back 25 actual/theoretical*100 |
front 26 How do you know nerolin is pure: | back 26 melting point and solubility in alcohol. Does not dissolve in water. |
front 27 What does sn1 mean | back 27 Substitution, nucleophilic and unimolecular |
front 28 how to identify an SN1 reaction | back 28 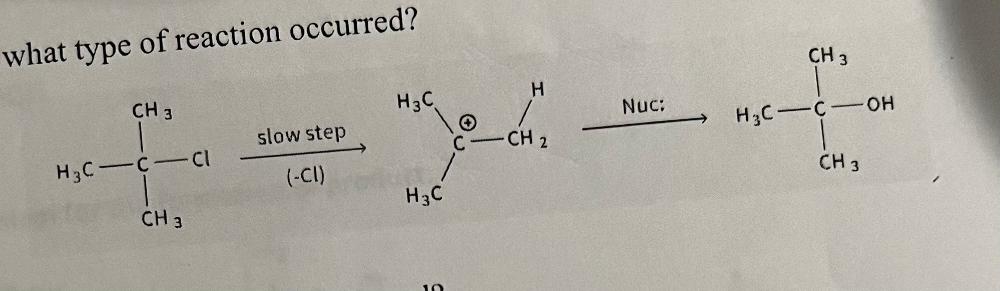 |
front 29 how to identify an E1 reaction | back 29 will have a leaving group followed by a carbocation intermediate and then the base will create an alkene |
front 30 which of the alcohols named below will react fastest with HCl to form the corresponding alkyl chloride via an Sn1 pathway? | back 30 t-butyl alcohol . This is because it is able to form a tertiary carbocation when the leaving group is gone unlike other structures. |
front 31 The rate of a first order chemical reaction is proportional to the concentration of electrophile | back 31 yes because the concentration and the rate of the first reaction are directly proportional. |
front 32 Write a chemical equation for the conversion of tbutyl chloride with water | back 32 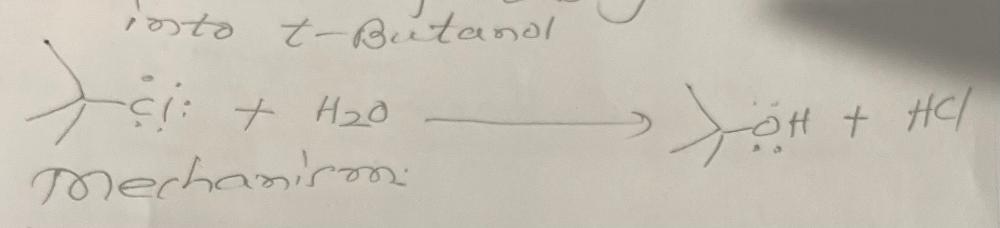 |
front 33 Write a rational mechanism for the formation of product | back 33 experiment 8 SN reaction |
front 34 What happens when ethyl chloride is treated with NaOH | back 34 C2H2Cl+NaOH---^C2H5OH+NaCl |