Instructions for Side by Side Printing
- Print the notecards
- Fold each page in half along the solid vertical line
- Cut out the notecards by cutting along each horizontal dotted line
- Optional: Glue, tape or staple the ends of each notecard together
Class quizzes
front 1 Light can be thought of a particles called photons. The energy, E( measured in joules), of a photon is proportional to its frequency, according to the formula, E=hv. In this formula, what is "h"? | back 1 Planck's constant |
front 2 What are the components of a typical single-beam spectrophotometer include which of the following? | back 2
|
front 3 Absorbance is proportional to the concentration of light absorbing molecules in a sample and the pathlength of substance through which the light travels. The dependence on concentration and pathlength is expressed by the formula A=εbc which is also known as: | back 3 Beer-Lambert's law |
front 4 List three difference between atomic absorption spectrometry and atomic emission spectrometry: | back 4 atomic absorption:
emission:
|
front 5 What is the definition of spectral interference? | back 5 When analyte signal overlaps signal from other species |
front 6 What is the definition of chemical interference? | back 6 Caused by any substance that decreases the extent to which an analyte breaks apart into gaseous atoms |
front 7 What is the definition of ionization interference? | back 7 A problem often associated with the presence of alkali metals with low ionization potentials |
front 8 What is the definition of atomization? | back 8 The process of breaking an analyte into gaseous atoms |
front 9 The pH of a buffer depends on which of the following? | back 9
|
front 10 The central equation for buffers is the ______ equation, which is simply a rearranged for of the Ka equilibrium expression. | back 10 Henderson-Hasselbalch |
front 11 What is the definition of autoprotolysis? | back 11 A substance that acts as both an acid and a base |
front 12 What is the definition of Kb? | back 12
|
front 13 What is the definition of a weak base? | back 13 substance that reacts with water by abstraction of a proton to H2O |
front 14 What is the definition of a weak acid? | back 14 Substances that reacts with water by donating a proton to H2O |
front 15 What is the definition of mean? | back 15
|
front 16 What is the definition of median? | back 16 the middle number in a series of measurements |
front 17 What is the definition of standard deviation? | back 17
|
front 18 What is the definition of range? | back 18 The difference between the highest and the lowest values in a data. |
front 19 What is the definition of selectivity? | back 19 The ability of a method to distinguish the analyte of interest form other species in the sample |
front 20 What is the definition of sensitivity? | back 20 The ability of a method to respond reliably and measurable to changes in analyte concertation |
front 21 What is the definition of detection limit | back 21 The smallest quantity of analyte that is significantly different from the blank. |
front 22 What is the definition of linear range? | back 22 The analyte concentration range over which response in proportional to concentration. |
front 23 What is the definition of retention time? | back 23 The maximum time of elution of an analyte measured from the time of injection or from the hold-up time |
front 24 What is the definition of retention factor? | back 24 Measurement of how long analytes spend on the column compared to the mobile phase. |
front 25 What is the definition of resolution? | back 25 The ability to separate co-eluting analytes to yield distinct peaks with no overlap. |
front 26 What is the definition of chromatogram? | back 26 The two-dimensional visualization of how analytes elute based on the time (volume) and detector response. |
front 27 The______ equation describes the various factors influencing plate height (H) in terms of eddy diffusion(multiple paths), longitudinal diffusion, mass transfer and the flow rate. | back 27 Van Deemter |
front 28 In 1-2 sentences, explain why preparatory LC methods, such as solid phase extraction, size exclusion, partition, or affinity chromatography are sometimes performed prior to introducing the sample into an automated HPLC system: | back 28 to clean up the sample matrix, enrich target analytes and, if necessary, convert the analytes into species amenable to instrumental analysis through chemical reaction. |
front 29 The components of a typical HPLC system include which of the following? | back 29
|
front 30 In electrophoresis, cations flow towards the cathode while anions flow towards the anode. In capillary electrophoresis, ____________ cause the bulk flow of all solute polarities towards the cathode. | back 30 electroosmosis |
front 31 What is the definition of redox reactions? | back 31 The transfer of electrons from one species to another. |
front 32 What is the definition of potentiometry? | back 32 The use of voltage measurements to extract chemical information. |
front 33 What is the definition of voltammetry? | back 33 A quantitative procedure in which the current is measured while voltage between two electrodes is varied |
front 34 What is the definition of coulometry? | back 34 A quantitative procedure in which electrons participation in a chemical reaction are counted to determine how much analyte reacted. |
front 35 What is the definition of stripping analysis? | back 35 A quantitative procedure in which analyte from a dilute solution is first concentrated into a single drop, or thin film, |
front 36 What is the definition of amperometry? | back 36 A quantitative procedure in which the electric current between a pair of electrodes that are driving an electrolysis reaction is measured. |
front 37 What is the definition of junction potential? | back 37 the voltage difference that develops when two dissimilar electrolyte solution are placed in contact. |
front 38 What is the definition of electrogravimetric analysis ? | back 38 A quantitative procedure in which an an analyte is plated out on an electrode and weighed. |
front 39 What is the definition of Ion selective electrode? | back 39 A conductor that responds preferentially to one ionic species in a solution. |
front 40 What is the definition of polarography? | back 40 A quantitative procedure in which voltammetry is conducted with a dropping-mercury electrode. |
front 41 Select the correct word from the bracketed section. In a redox reaction, a molecule is said to be oxidized when it [loses, gains] electrons and reduced when it [loses, gain] electrons. An oxidizing agent [takes, gives] electrons from another substance; whereas a reducing agent [takes, gives] electrons to another substance. | back 41
|
front 42 [True or false] Electric current is proportional to the rate of a redox reaction | back 42 True |
front 43 [True or false] A reference electrode maintains a (fixed) potential against which the potential of another half-cell may be measured. | back 43 True |
front 44 The net driving force for a redox reaction is expressed by the ______ equation, whose two term include the driving force under standard condition and a term that shows the dependence on concentrations. | back 44 Nerst |
front 45 What is the definition of heterogenous? | back 45 immunoassay technique that requires separation of the analyte-antibodies complex from the remaining sample prior to analysis. |
front 46 What is the definition of homogeneous? | back 46 Immunoassay technique that does not require separation of the analyte-antibody complex prior to analysis |
front 47 What is the definition of Radial immunodiffusion(RID)? | back 47 Single diffusion technique in which reagent antibodies are incorporated into the agar and patient sample is added to a well in the agar. Patient antigens diffuse into the agar and complex with the antibodies to form a measurable precipitate ring. |
front 48 What is the definition of ouchterlony immunodiffusion? | back 48 Double diffusion technique in which reagent antibodies are added to a well in the agar and patient sample is added to another well. Both the antibodies and antigens diffuse into the agar where they can complex and form a precipitate line. |
front 49 What is the definition of rocket immunoelectrophoresis? | back 49 single diffusion technique in which reagent antibodies are incorporated into the agar and patient samples are added to individual wells in the agar. Patient antigens migrate into the agar n an electric field and complex with the antibodies to form measurable precipitate peaks. |
front 50 What is the definition of counter immunoelectrophoresis? | back 50 Double diffusion technique in which reagent antibodies are added to a well in the agar and patient sample is added to another well. An electrical current is applied to speed up the migration of the antibodies and antigens into the agar where they can complex and form a precipitate line. |
front 51 What is the definition of nephelometry? | back 51 Immune complexes form between the patient analyte and the reagent antigen/antibody competes with analyte in the sample for binding sites. |
front 52 What is the definition of turbidimetry? | back 52 immune complexes form between the patient analyte and the reagent antigen/antibody in solution causing precipitation. The amount of light transmitted through the solution is measured by a light detector placed at a 180° angle. |
front 53 What could cause falsely altered patient results using immunoassay techniques? | back 53
|
front 54 In 1-2 sentences, describe how the "hook effect" may alter patient results when using immunoassay techniques? | back 54 It gives falsely low results with certain immunoassays in the presence of excess amount of analyte of interest |
front 55 If a discrepant/discordant result occurs when testing a patient sample by immunoassay, what is the first step in investigating the possibility of the hook effect? | back 55 perform a serial dilution. |
front 56 What is the definition of chromatography? | back 56 a process in which compounds are separated from on another by passing a mixture through a column that retain some compounds longer than others. |
front 57 What is the definition of effluent? | back 57 The fluid entering the chromatographic column |
front 58 What is the definition of eluate? | back 58 The fluid exiting the chromatographic column |
front 59 What is the definition of theoretical plates? | back 59 a concept wherein a column in thought of as having discrete sections in which a solute molecule equilibrates between the mobile and stationary phases. |
front 60 What is the definition of adsorption chromatography? | back 60 Solutes tick to the surface of the solid stationary phase. |
front 61 What is the definition of partition chromatography? | back 61 Solutes equilibrate between the mobile phase and a thin liquid station film |
front 62 What is the definition of molecular exclusion chromatography? | back 62 Solutes penetrate into pores in the station phase allowing the largest solutes to elute first. |
front 63 What is the definition of Ion-exchange chromatography? | back 63 Solute ions are attracted to specific groups attached to the stationary phase. |
front 64 Describe the differences between classical liquid chromatography and high-performance liquid chromatography. | back 64 the solvent in LC travels by the force of gravity. In the application of HPLC, the solvent travels under high pressure obtained by means of a pump to overcome the pressure drop in the packed column, which reduces the time of separation. |
front 65 Elution with a single solvent of a constant solvent mixture is called______ elution; whereas, in ______ elution, the solvent in changed continuously from weak to stronger eluent strength. | back 65
|
front 66 What is the definition of electrophoresis? | back 66 The migration of ions in an electric field |
front 67 What is the definition of capillary electrophoresis? | back 67 A high-resolution separation Technique conducted with solution of icons in a narrow bore tube. |
front 68 What is the definition of electro osmosis? | back 68 The propulsion of fluid inside a fused-silica capillary form the anode toward the cathode caused by the applied electric field |
front 69 What is the definition of capillary gel electrophoresis? | back 69 a separation technique in which macromolecules are separated by sieving as they migrate through a gel inside a capillary tube. |
front 70 What is the definition of electric charge(q)? | back 70 A fundamental property of some subatomic particles (electrons, protons) that is measured in coulombs |
front 71 What is the definition of electric current(I)? | back 71 The movement or flow of charged particles measured in amperes |
front 72 What is the definition of capillary gel electrophoresis electrical work(W)? | back 72 The work done as electrons move between two points that is expressed in joules |
front 73 What is the definition of potential difference (E)? | back 73 The difference in electric potential between two points that is measured in points. |
front 74 What is the correct word in each bracketed section? In electrochemistry, the [anode, cathode] is the electrode where [oxidation, reduction] occurs resulting in a loss of electrons; where as the [ anode, cathode] is the electrode where [oxidation, reduction] occurs resulting in a gain of electrons. | back 74
|
front 75 Zn(s) +H2SO4(aq) →ZnSO4(aq) + H2(g) In this reaction the electron donor is ___ and the electron acceptor is_____
| back 75 d |
front 76 A device used to maintain electrical neutrality in a galvanic cell is termed the;
| back 76 e |
front 77 A(n)_____ electrode is designed to respond preferentially to a particular a species in solution.
| back 77 d |
front 78 samples introduced into a mass spectrometry system are volatilized and then ionized so the charged molecules and fragments can be separated according to their___?
| back 78 e |
front 79 What are the basic components that are standard in all mass spectrometers. | back 79
|
front 80 What is the definition of electron ionization? | back 80 ionization technique in which molecules are bombarded with high-energy electrons resulting in the formation of charge molecular ions and fragments |
front 81 What is the definition of electrospray ionization? | back 81 Ionization technique in which the LC effluent passes through a capillary to which a voltage has been applied. Energy is transferred to the solvent droplets, becoming charged, and evaporation of the solvent leads to coulombic repulsion and election of ions. |
front 82 What is the definition of atmospheric pressure chemical ionization? | back 82 Ionization technique in which the LC effluent is rapidly desolvated to from a gas phase that interacts with a cloud of electrons formed by a corona discharge needle to become ionized |
front 83 What is the definition of Matrix-assisted laser desorption ionization? | back 83 Ionization technique in which the sample is mixed with a matrix solvent and spotted onto a stainless-steel plate. A laser pulse irradiates the sample causing desorption and ionization of the matrix and the sample. |
front 84 Which of the following mass spectrometer technologies can effectively concentrate the ions of interest yielding greater analytical sensitivity?
| back 84 b |
front 85 In a tandem mass spectrometer, or triple quad, which of the quadrupoles function as a collision cell?
| back 85 b |
front 86 The conjugate base of HNO3 would be:
| back 86 b |
front 87 A solution with a [H]=1.0x10-4 M has a pH of _____ and would be considered_____
| back 87 b |
front 88 Which of these is the strongest acid listed?
| back 88 c |
front 89 Which parameters are required for evaluation of a FDA approved method? | back 89
|
front 90 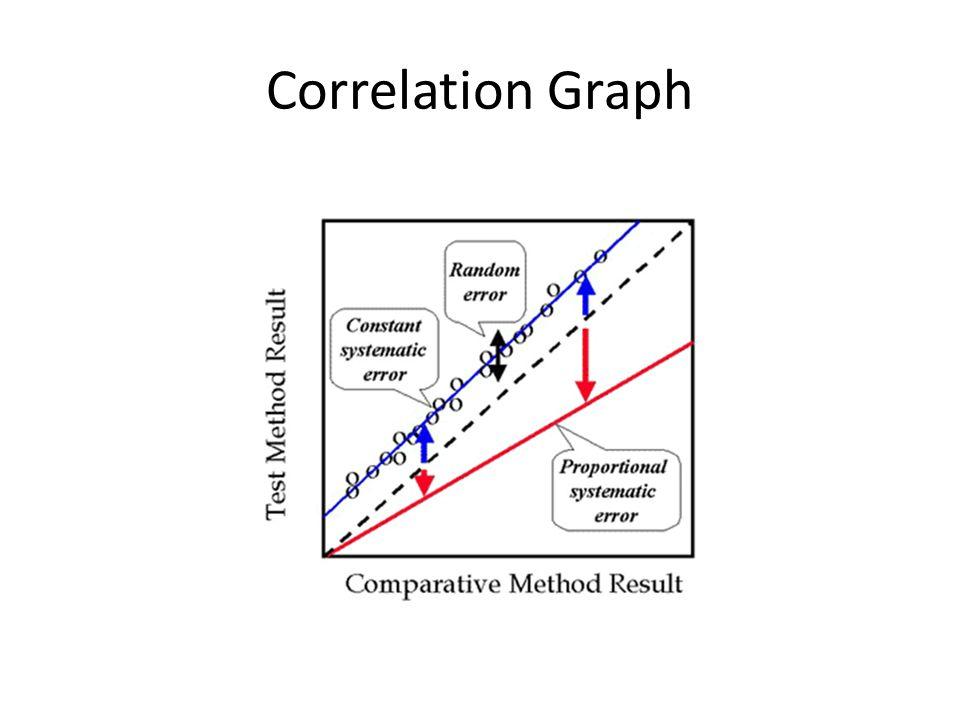 Know the different lines represent the different types of errors. | back 90 Know the different lines represent the different types of errors. |
front 91 Which of the following test parameters could be evaluated by testing 5 samples with known concentration, in duplicate and assessing the linearity between the expected and measured values?
| back 91 c |
front 92 What are the labels used in immunoassays? | back 92
|
front 93 Chose the correct word in the bracketed section In a competitive immunoassay, labeled[antigens, antibodies] in the reagent compete with antigens in the patient sample for a limited number of binding sites. In this type of immunoassay, the generated signal is [ directly, inversely] proportional to the a pro | back 93
|
front 94 What term refers to an immunologic phenomenon that occurs when excess analyte{ such as patient antigen or antibody} overwhelms the test system leading to a false inaccurate result? | back 94 Hook effect |