Instructions for Side by Side Printing
- Print the notecards
- Fold each page in half along the solid vertical line
- Cut out the notecards by cutting along each horizontal dotted line
- Optional: Glue, tape or staple the ends of each notecard together
Instrumental methods in electrochemistry
front 1 What is Electrolysis? | back 1 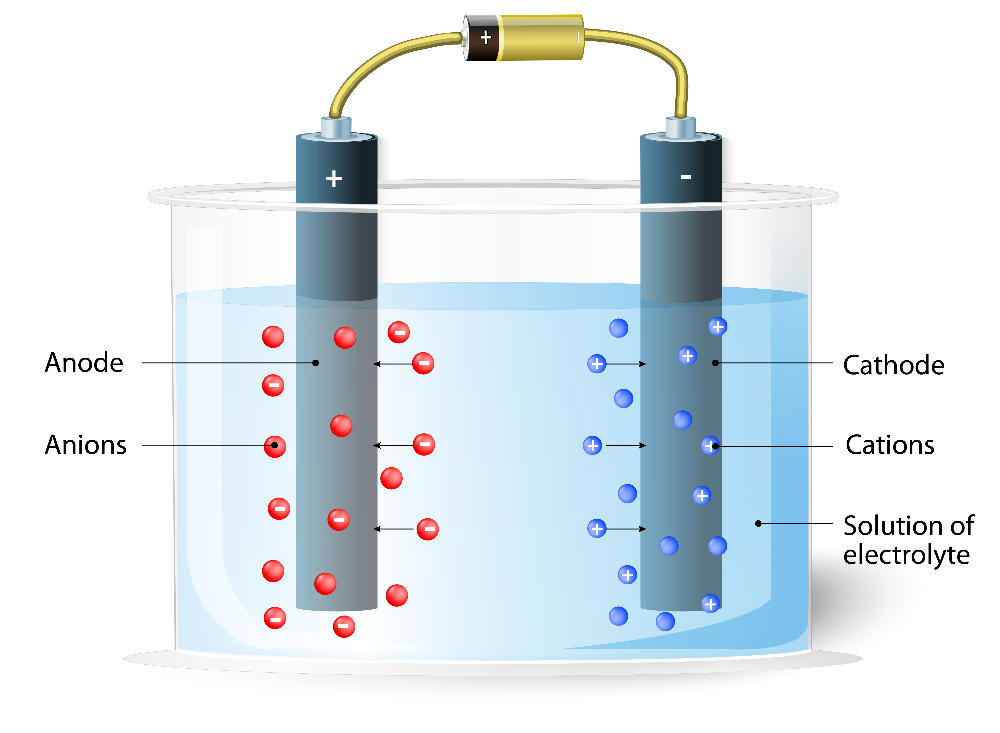 a chemical reaction in which we apply a voltage to drive a redox reaction that would not otherwise occur |
front 2 What is an electroactive species? | back 2 it is a one that can be oxidized or reduced at an electrode. |
front 3 What is electrogravimetric analysis? | back 3 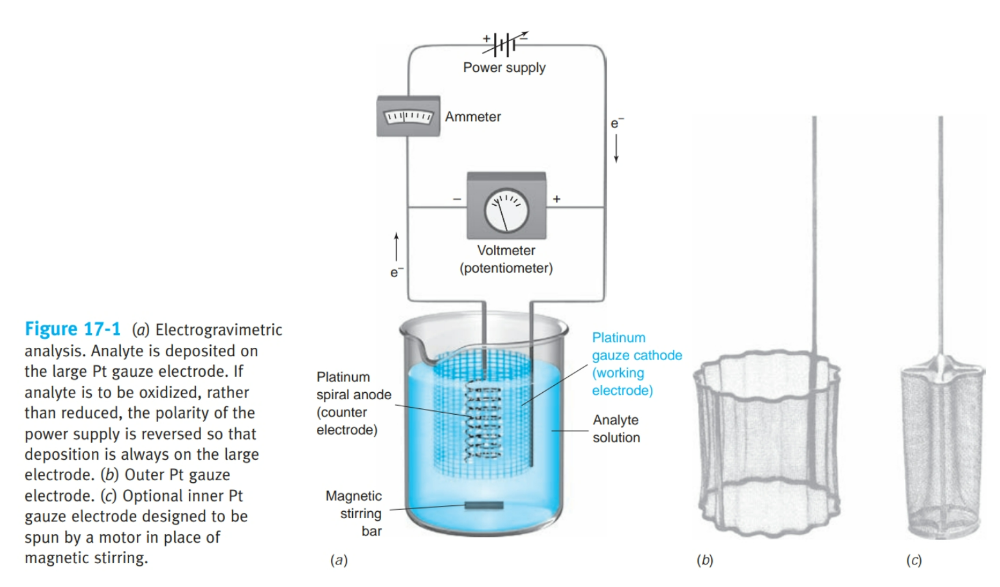 analyte is plated out on an electrode and weighed. |
front 4 How do you find out when electrolysis is complete? | back 4 1. Disappearance of color 2. Deposition on freshly exposed electrode surface 3. Qualitative test for analyte in solution |
front 5 What is coulometry? | back 5
|
front 6 What is amperometry? | back 6
|
front 7 What is a Clark electrode? | back 7 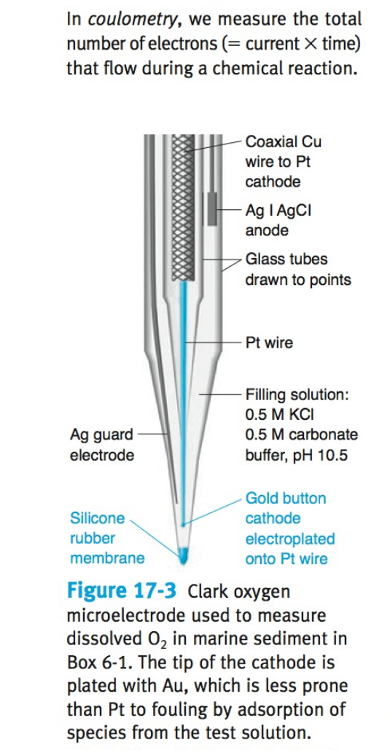 an electrode that measures ambient oxygen partial pressure in a liquid using a catalytic platinum surface according to the net reaction |
front 8 What is a biosensor? | back 8 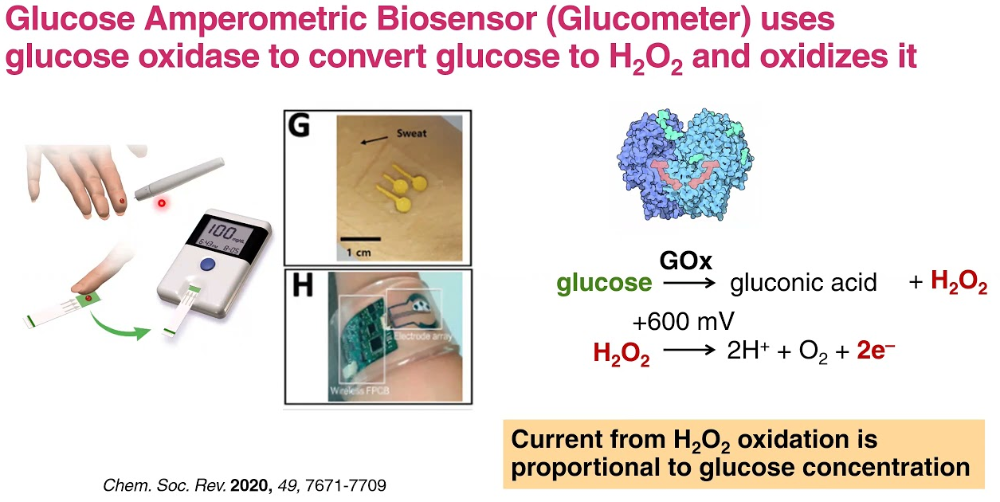 a device that uses a biological component such as an enzyme or anti- body for highly selective response to one analyte. |
front 9 What is a mediator? | back 9 transports electrons between analyte and the working electrode. The mediator undergoes no net reaction itself. |
front 10 What is a Reference electrode? | back 10 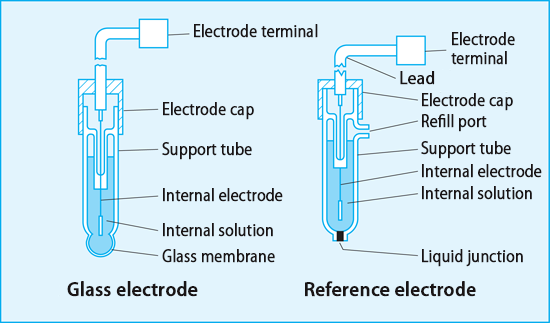
|
front 11 What is a Potentiostat? | back 11 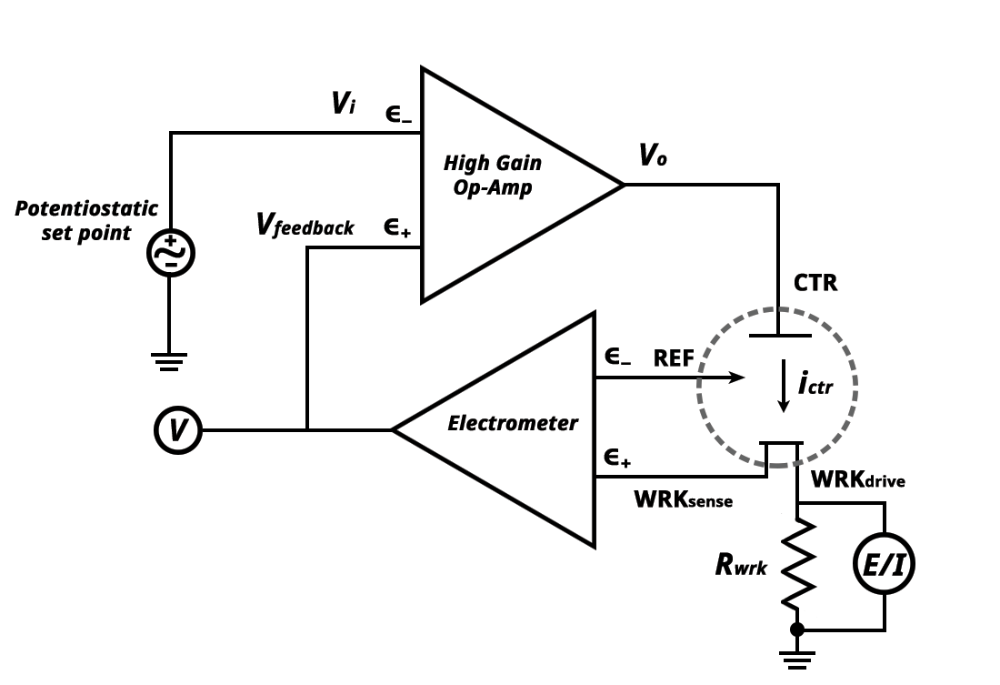
|
front 12 What is a Working electrode? | back 12 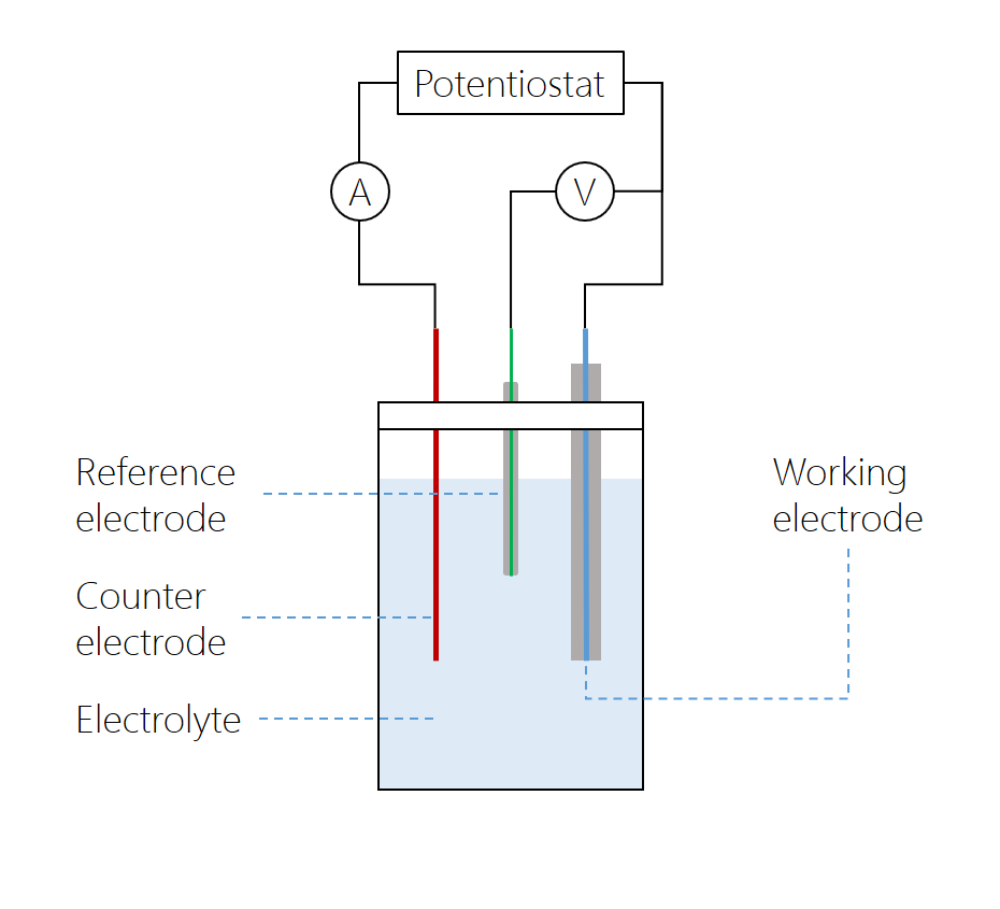
|
front 13 What is a Auxiliary electrode? | back 13 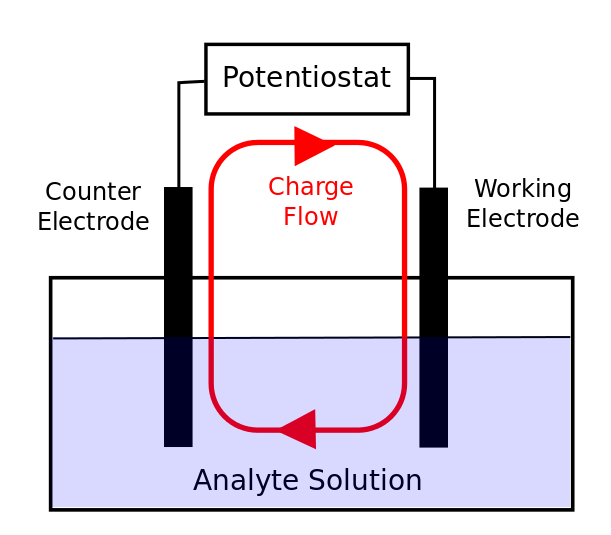
|
front 14 What is voltammetry? | back 14 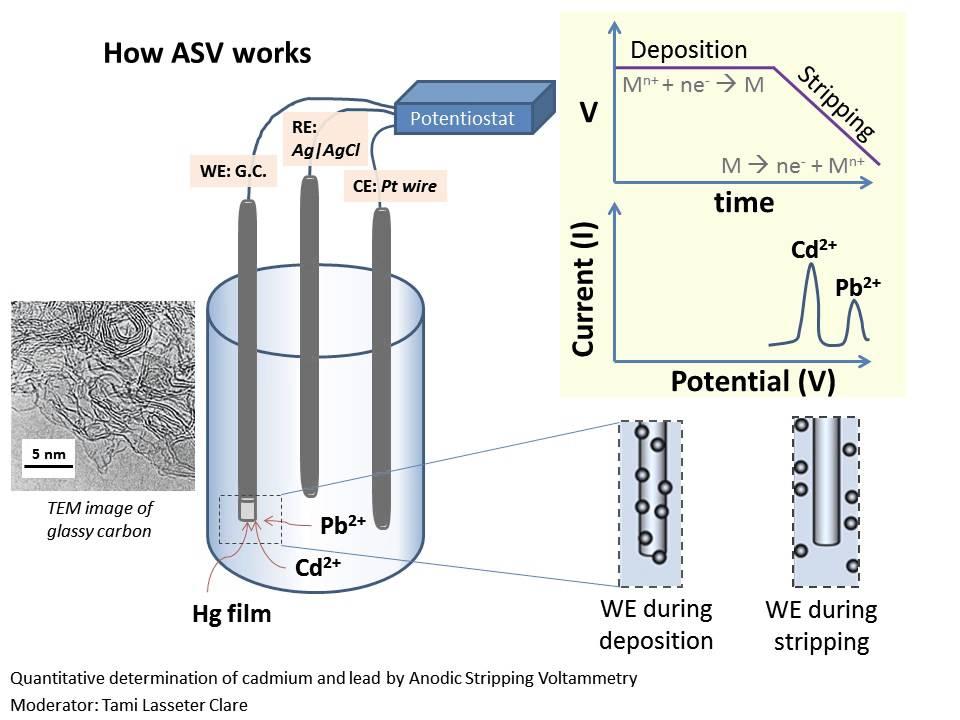
|
front 15 What is Polarography? | back 15 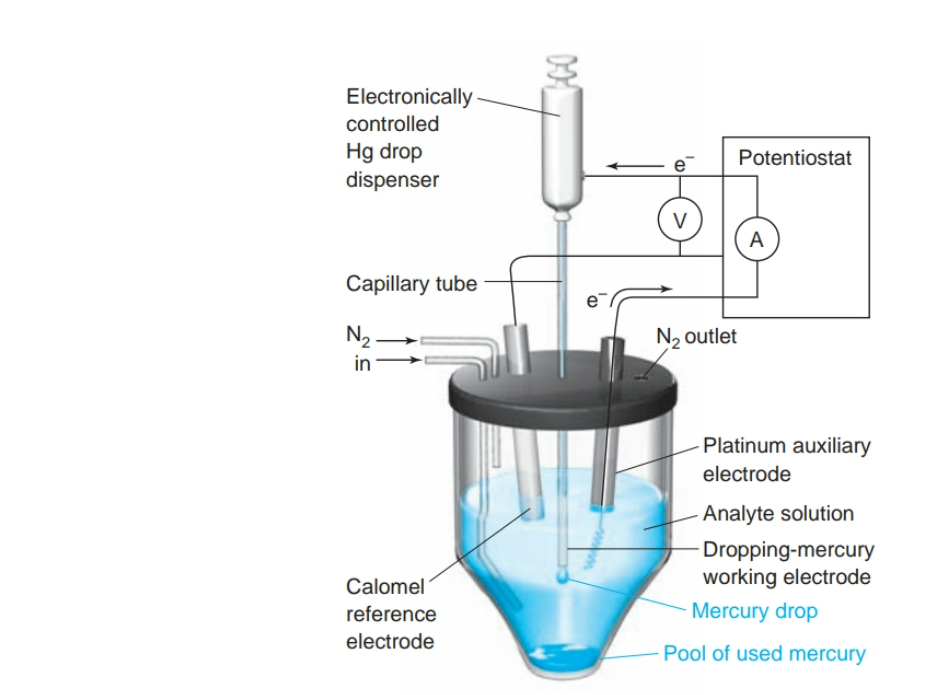
|
front 16 What is a polarogram? | back 16 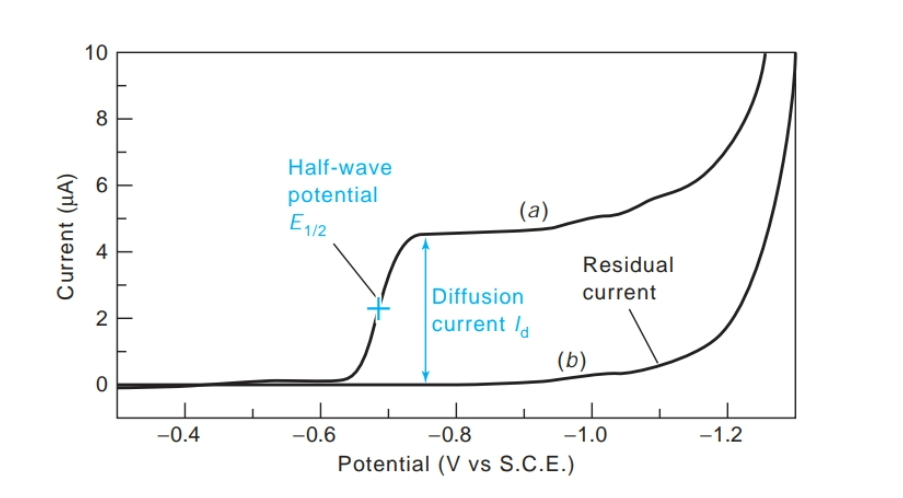 is a graph of current versus voltage |
front 17 What is the half-wave potential (E1/2)? | back 17  The potential at which half the maximum current is reached |
front 18 What is the diffusion current? | back 18 
|
front 19 What is the residual current? | back 19  the absence of analyte due mainly to reduction of impurities in the solution and on the surface of the electrodes |
front 20 What is faradaic current? | back 20 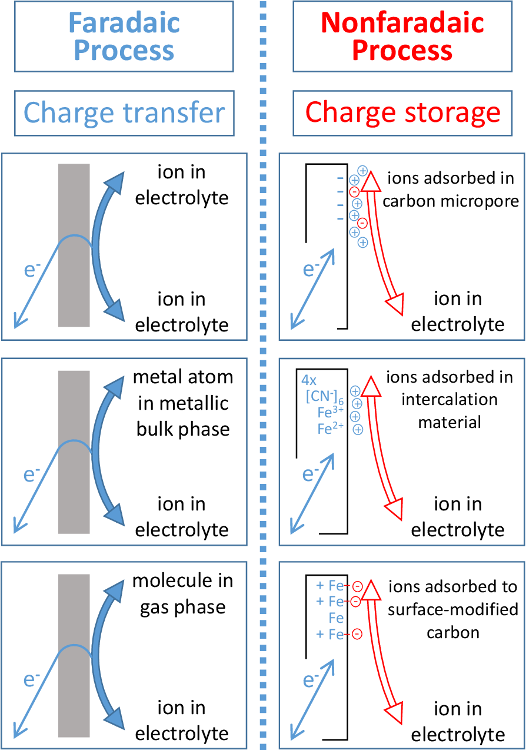
|
front 21 What is Charging current (capacitor current)? | back 21
|
front 22 What are Advantages of square wave voltammetry? | back 22 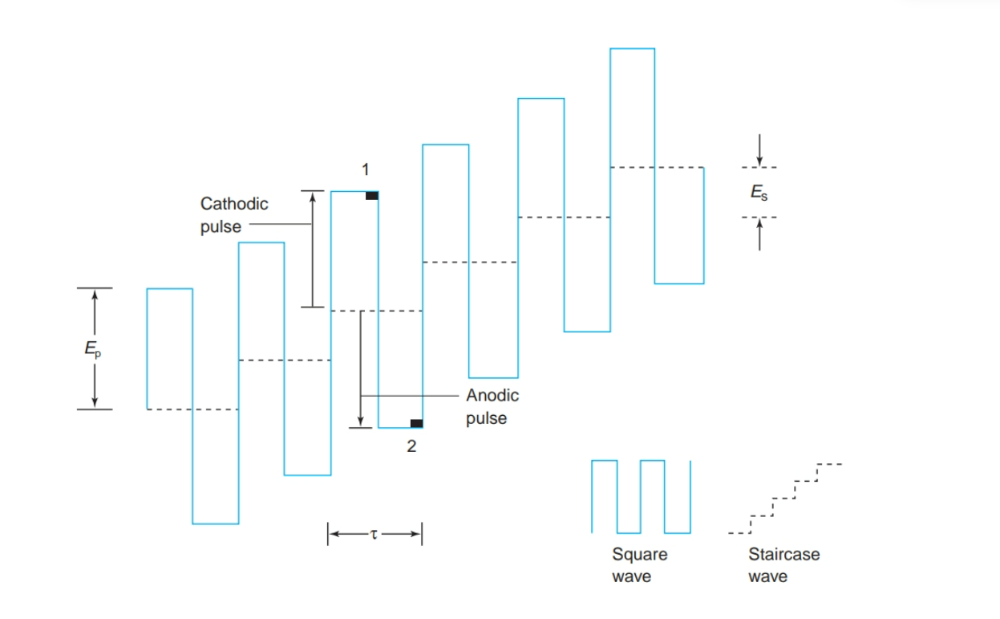 • Increased signal • Derivative (peak) shape provides better resolution of neighboring signals •Faster measurements |
front 23 What are the steps of stripping analysis? | back 23 1. Concentrate analyte into a drop of Hg by reduction. 2. Reoxidize analyte by making the potential more positive. 3. Measure polarographic signal during oxidation. |