Instructions for Side by Side Printing
- Print the notecards
- Fold each page in half along the solid vertical line
- Cut out the notecards by cutting along each horizontal dotted line
- Optional: Glue, tape or staple the ends of each notecard together
Electrode Measurements
front 1 What is the definition of potentiometry? | back 1 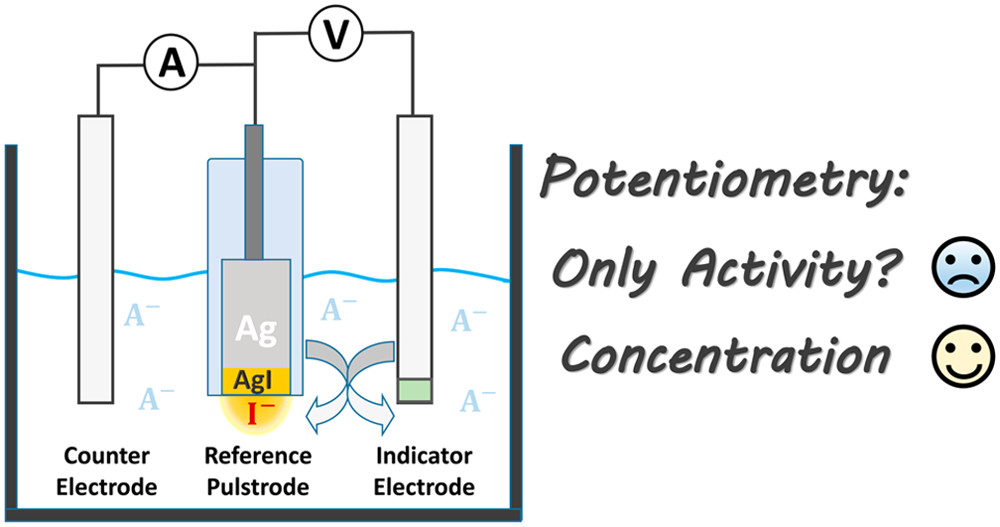 measurement of the electrical potential difference between two
electrodes in an |
front 2 What is the definition of oscillating reactions? | back 2 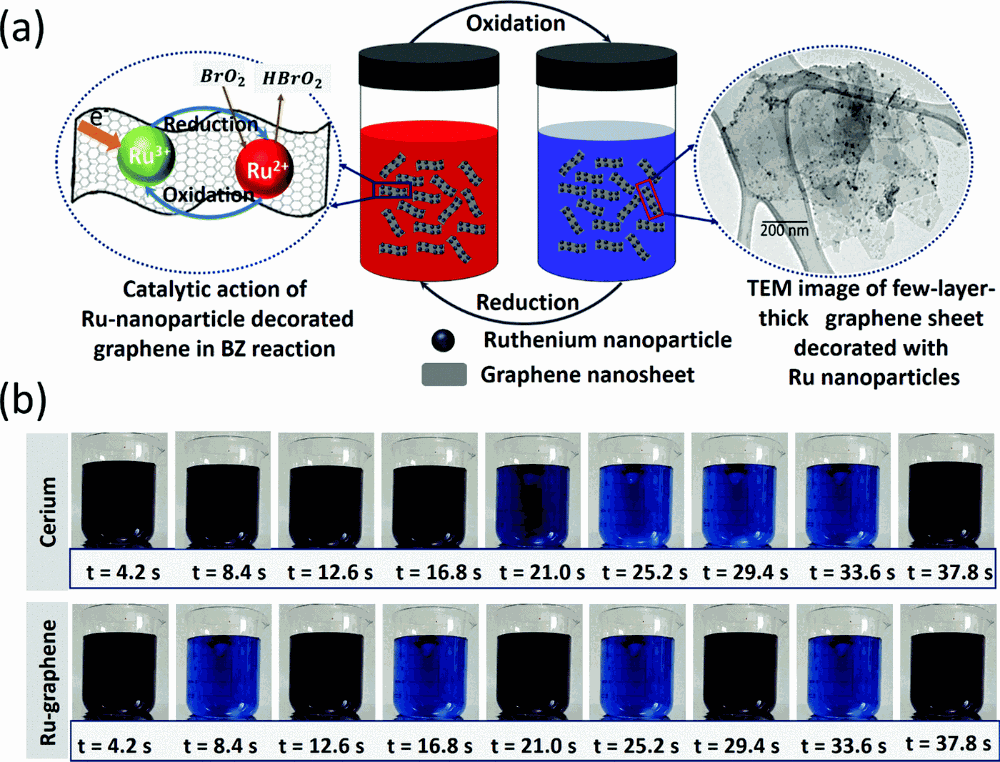 in which chemical concentrations oscillate between high and low values. |
front 3 What is the definition of junction potential? | back 3 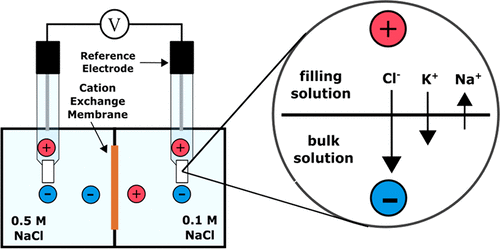 When two dissimilar electrolyte solutions are placed in contact, a voltage difference develops at the interface. |
front 4 What is the difference between direct and relative potentiometric measurements? | back 4 Direct
Liquid-liquid junction at 4% among 14 measurements by direct potentiometry
Relative
|
front 5 What is a junction potential? | back 5
|
front 6 What are double-junction reference electrodes used for? | back 6 typically used for ISE [Ion selective electrode] analysis where the leached electrolyte must not interfere with the ISE being used. They are also particularly useful for pH analysis of products that must not have KCl or a strong electrolyte contaminating the sample |
front 7 What is an ISE and what are some advantages? | back 7 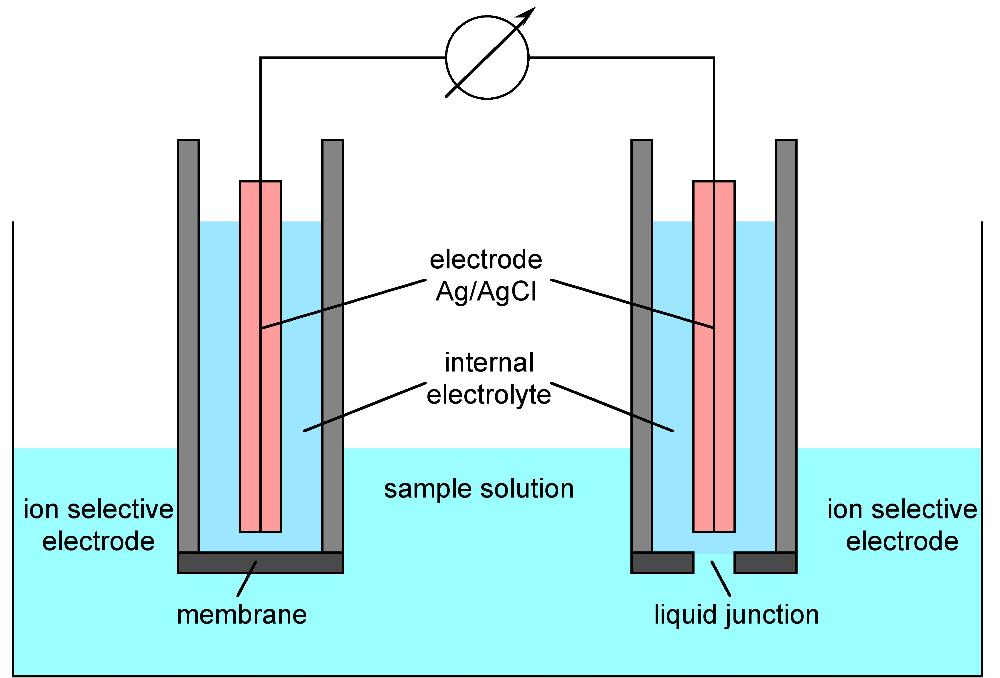 is an analytical technique used to determine the activity of ions in aqueous solution by measuring the electrical potential.
|
front 8 What is the function of an ISE (Ion-selective electrode)? | back 8 selectively binds one ion—no redox chemistry |
front 9 What is the function of a Metal electrode? | back 9 It is a surface on which redox reaction takes place. |
front 10 How does a glass pH electrode work? | back 10
|
front 11 What are different errors of pH measurement? | back 11
|
front 12 What is a combination electrode? | back 12 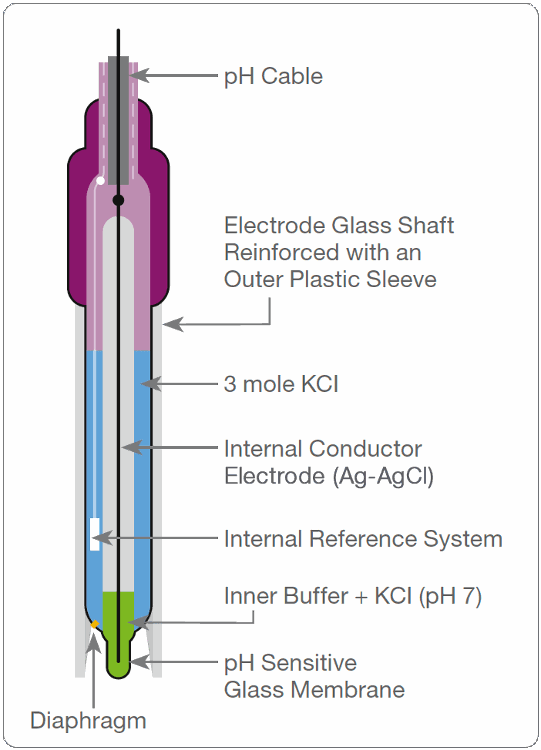 incorporating both the glass and reference electrodes in one body |
front 13 What should be done to pH electrode before use? | back 13 They must be calibrated before use. It should be calibrated every 2 h in sustained use. Ideally, calibration standards should bracket the pH of the unknown. |
front 14 What should you not do to a glass electrode? | back 14 Don’t leave it out of water (or in a nonaqueous solvent) longer than necessary. |
front 15 What are some advantages and disadvantages of ISE (Ion-selective electrode)? | back 15 Advantages:
Disadvantages:
|
front 16 What is a solid-state ion-selective electrode? | back 16 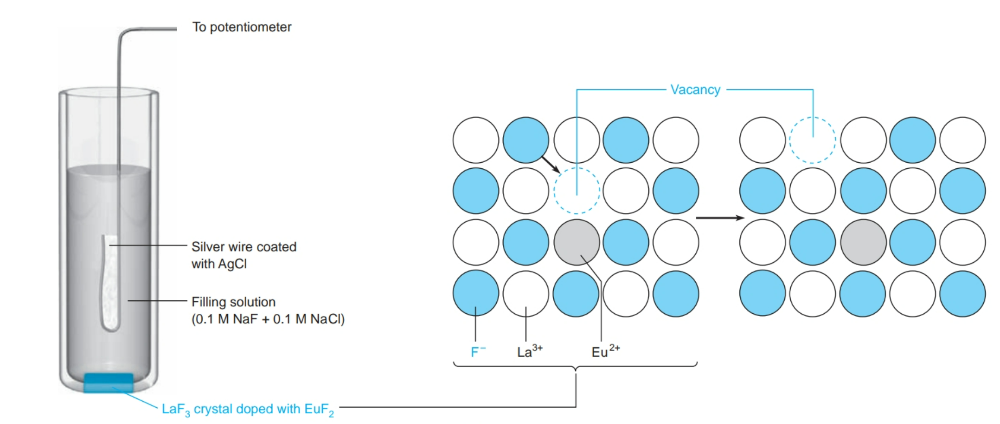
|
front 17 What is a liquid-based ion-selective electrode? | back 17 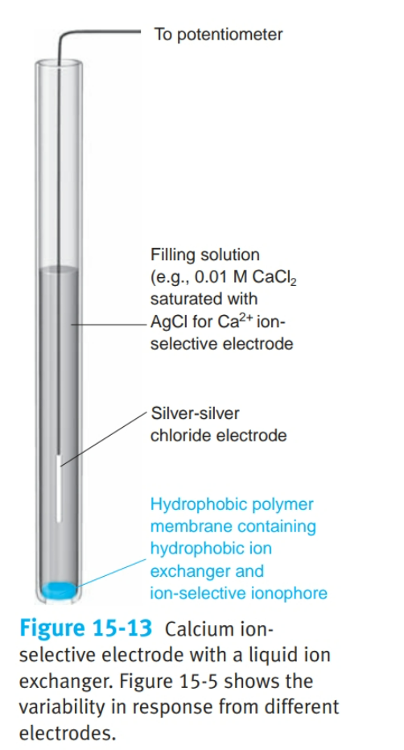
|
front 18 What is a compound electrode? | back 18 a conventional electrode surrounded by a membrane that isolates (or generates) the analyte to which the electrode responds.
|
front 19 What are the different types of membrane electrodes? | back 19 There are four main types of ion-selective membrane used in ion-selective electrodes (ISEs): glass, solid state, liquid based, and compound electrode. |
front 20 What is the quantity, method, and analyte for potentiometry? | back 20 
|
front 21 What is the quantity, method, and analyte for coulometry? | back 21 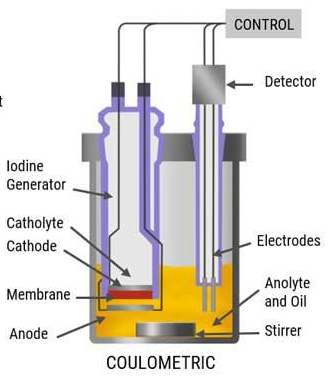
|
front 22 What is the quantity, method, and analyte for amperometry? | back 22 
|
front 23 What is the quantity, method, and analyte for voltammetry? | back 23 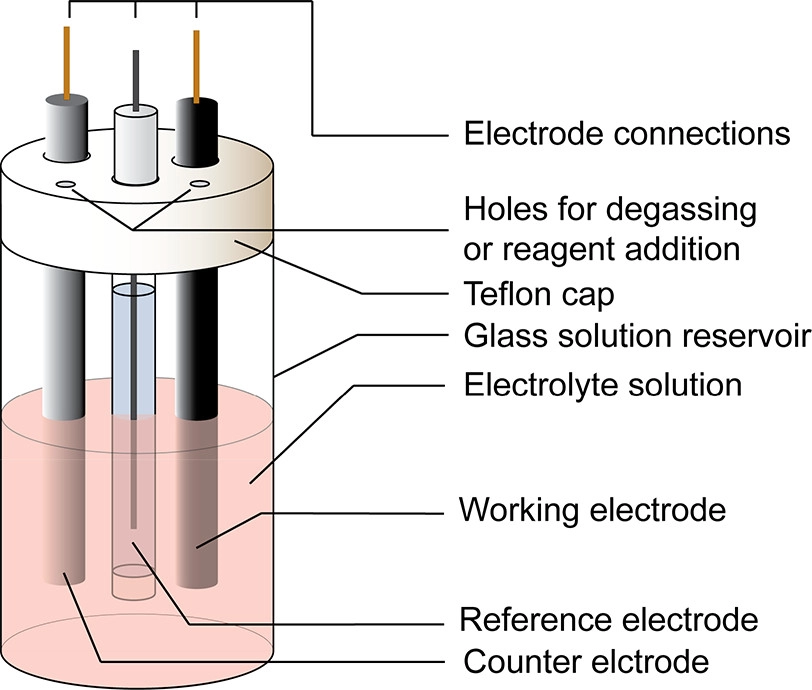
|