Instructions for Side by Side Printing
- Print the notecards
- Fold each page in half along the solid vertical line
- Cut out the notecards by cutting along each horizontal dotted line
- Optional: Glue, tape or staple the ends of each notecard together
Electrode potentials
front 1 What is the definition of Oxidation? | back 1 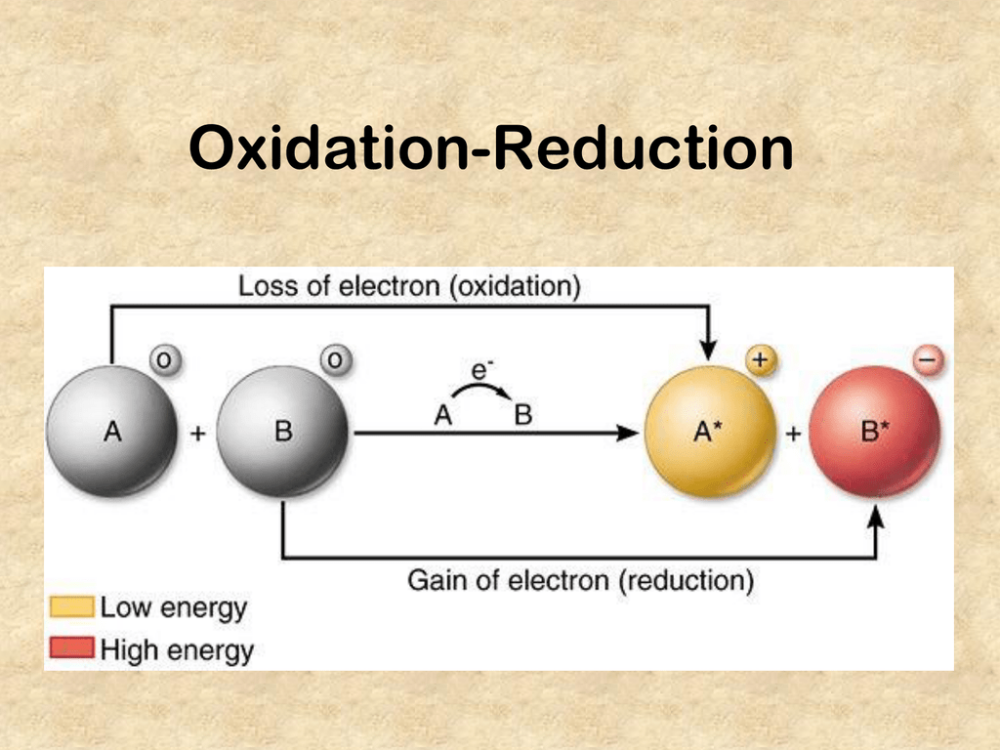 loss of electrons |
front 2 What is the definition of Reduction? | back 2  gain of electrons |
front 3 What is the definition of a redox reaction? | back 3 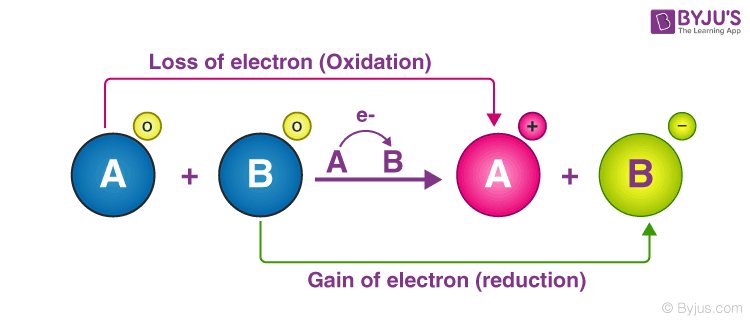 electrons are transferred from one species to another. A molecule is said to be oxidized when it loses electrons. It is reduced when it gains electrons. |
front 4 What is the definition of oxidizing agent? | back 4 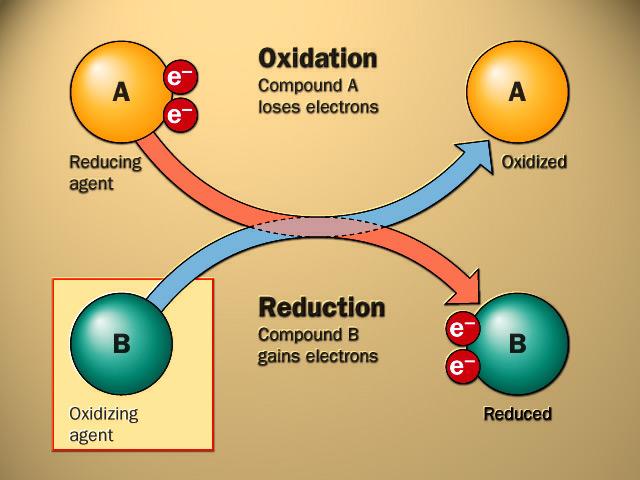 also called an oxidant, takes electrons from another substance and becomes reduced. |
front 5 What is the definition of reducing agent? | back 5  also called a reductant, gives electrons to another substance and is oxidized in the process. In the reaction |
front 6 What is the definition of coulombs? | back 6 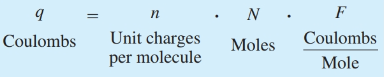 Electric charge (q) |
front 7 What is the definition of Faraday constant? | back 7 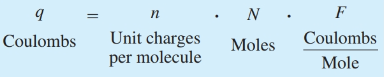 F=96 485.3321 s A / mol |
front 8 What is the definition of Electric current? | back 8 (I) is the quantity of charge flowing each second past a point in an electric circuit. |
front 9 What is the definition of ampere? | back 9 
|
front 10 What is the definition of an electrode? | back 10 a device to conduct electrons into or out of the chemicals involved in the redox reaction. |
front 11 What is the definition of an electroactive species? | back 11 a molecule that can donate or accept electrons at an electrode. |
front 12 What is the definition of Electrolysis? | back 12 is a chemical reaction in which we apply a voltage to drive a redox reaction that is not spontaneous (not energetically favorable) and would not otherwise occur. |
front 13 What is the definition of electric potential? | back 13 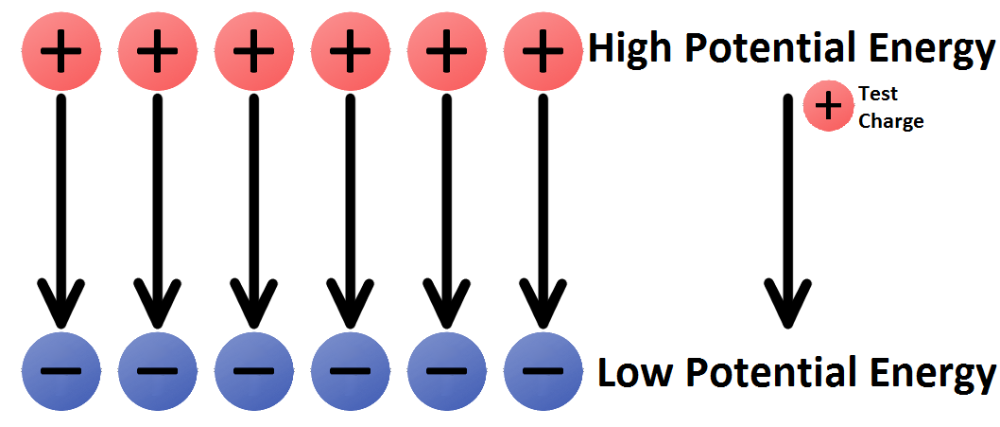
|
front 14 What is the definition of a spontaneous reaction? | back 14 it is energetically favorable for reactants to be converted into products. Energy released from the chemicals could be available as electrical energy. |
front 15 What is the definition of a galvanic cell? | back 15 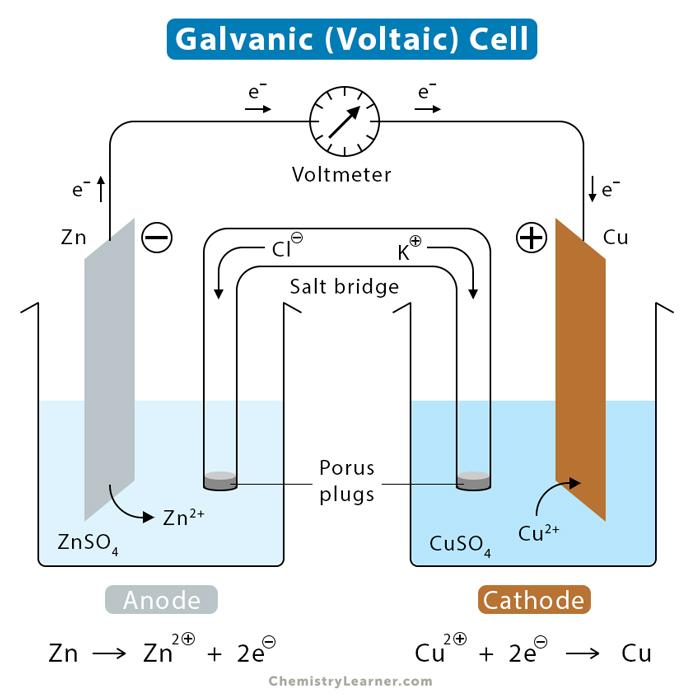
|
front 16 What is the definition of a salt bridge? | back 16 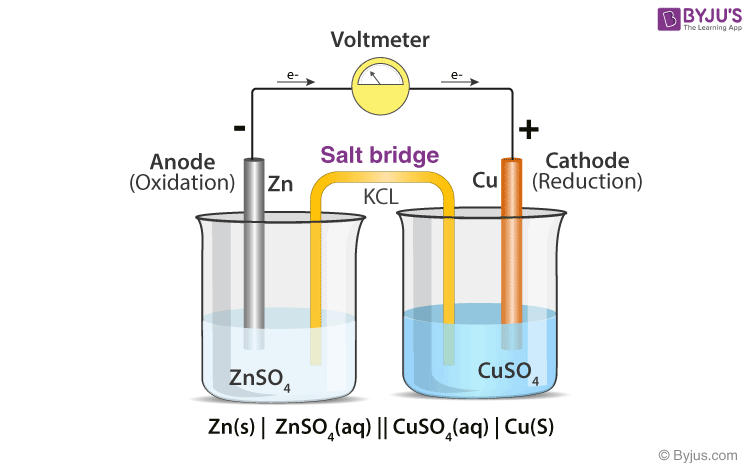 is a junction that connects the anodic and cathodic compartments in a cell or electrolytic solution. |
front 17 What is the definition of a cathode? | back 17  electrode at which reduction occurs |
front 18 What is the definition of an anode? | back 18  the electrode at which oxidation occurs |
front 19 What is the definition of standard reduction potential (E°)? | back 19
|
front 20 What is the definition of standard hydrogen electrode (S.H.E.)? | back 20 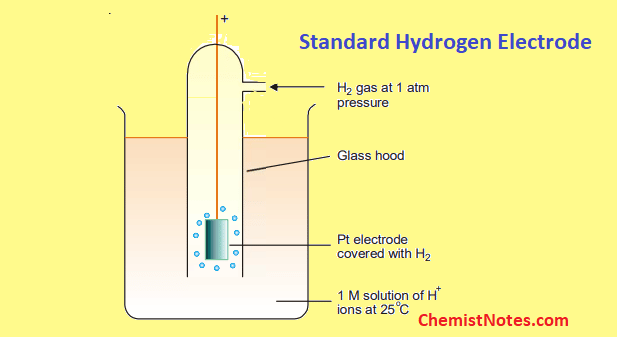
|
front 21 What is the definition of formal potential? | back 21 The potential for a cell containing a specified concentration of reagent other than 1 M |
front 22 What is the definition of an Indicator electrode? | back 22 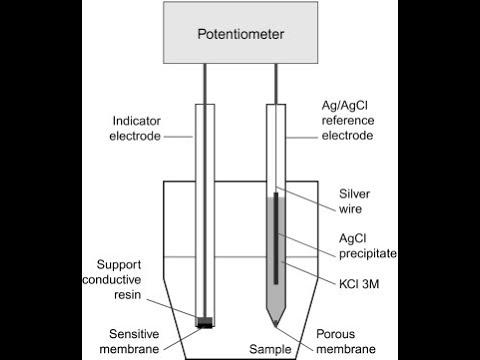
|
front 23 What is the definition of a Reference electrode? | back 23 
|
front 24 What is the definition of a silver-silver chloride electrode? | back 24 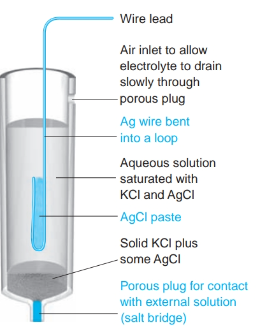
|
front 25 What is the definition of a saturated calomel electrode S.C.E. | back 25 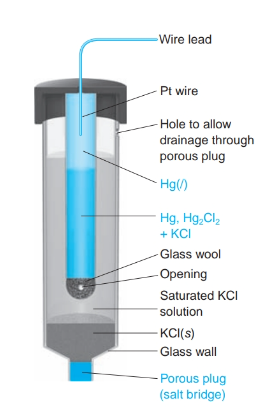 a reference electrode based on the reaction between elemental mercury and mercury (I) chloride. *when saturated Cl- does not change with evaporating liquid. |
front 26 What is the definition of a volt? | back 26  the SI unit of electromotive force, the difference of potential that would drive one ampere of current against one ohm resistance. |
front 27 When is the double junction electrode used? | back 27 if you do not want the analyte and electrode to have direct contact. |
front 28 What are some common indicator electrodes? | back 28
|
front 29 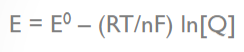 What does the variable "E" stand for in the Nernst equation? | back 29 Electromotive force of a cell |
front 30 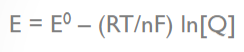 What does the variable "E0" stand for in the Nernst equation? | back 30  standard reduction potential |
front 31 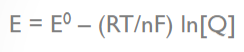 What does the variable "R" stand for in the Nernst equation? | back 31 molar gas constant |
front 32 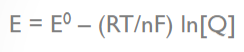 What does the variable "T" stand for in the Nernst equation? | back 32 temperature in degrees Kelvin |
front 33 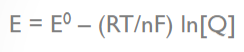 What does the variable "n" stand for in the Nernst equation? | back 33 number of moles of electrons transferred |
front 34 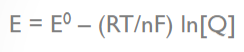 What does the variable "F" stand for in the Nernst equation? | back 34 Faraday’s constant (96,500 C/mol) |
front 35 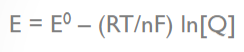 What does the variable "Q" stand for in the Nernst equation? | back 35 the reaction quotient (a function of the activities or concentrations of the chemical species involved) |
front 36 What is the definition of electrolyte? | back 36 a substance that dissociates into ions in solution and acquires the capacity to conduct electrons |
front 37 What is the definition of electrode potential? | back 37 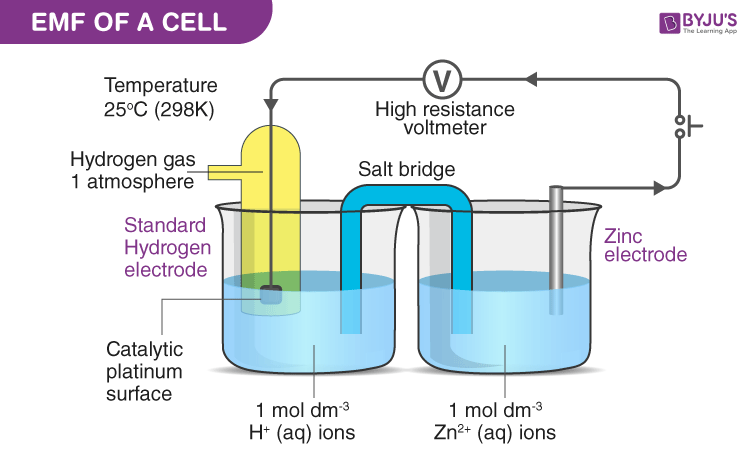
|
front 38 What isthe standard state of an element? | back 38
|
front 39 What is the Nernst equation for? | back 39 o Calculate the voltage of an electrochemical cell |
front 40 What is the definition of ELECTROLYTIC CELL? | back 40 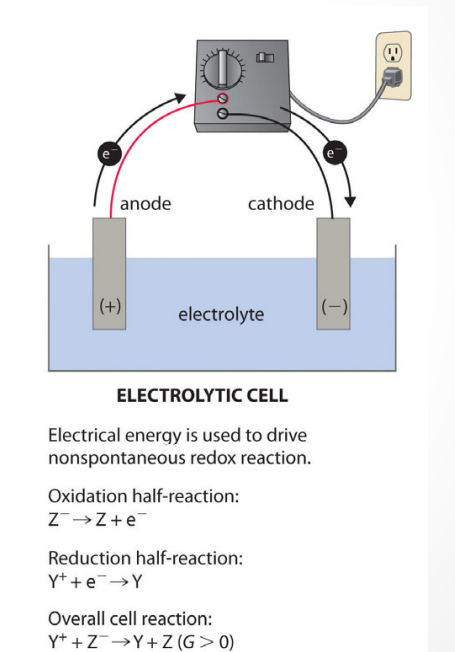 an electrochemical cell in which chemical reactions occur through the application of an external potential difference |