Instructions for Side by Side Printing
- Print the notecards
- Fold each page in half along the solid vertical line
- Cut out the notecards by cutting along each horizontal dotted line
- Optional: Glue, tape or staple the ends of each notecard together
CHAPTER 8A Problem Solving
front 1 [8A.2] Describe the features of the solution of the particle in a one-dimensional box that appear in the solutions of the particle in two- and three-dimensional boxes. What concept applies to the latter but not to a one-dimensional box. | back 1  |
front 2 [8A.1(b)] Determine the linear momentum and kinetic energy of a free proton described by the wavefunction e^−ikx with k=5nm−1. | back 2  |
front 3 [8A.3(b)] Calculate the energy separations in joules, kilojoules per mole, electronvolts, and reciprocal centimetres between the levels (i) n = 3 and n = 1, (ii) n = 7 and n = 6 of an electron in a box of length 1.50 nm. | back 3  |
front 4 [8.4(b)] Calculate the probability that a particle will be found between 0.65L and 0.67L in a box of length L when it has (i) n = 1, (ii) n = 2. Take the wavefunction to be a constant in this range. | back 4  |
front 5  [8A.7(b)] | back 5 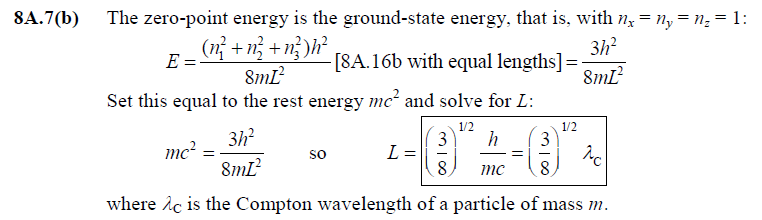 |
front 6  [8A.11(b)] | back 6  surprised to find degeneracy in a box with unequal lengths. Symmetry
is a matter of degree. |
front 7 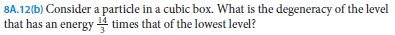 [8A.12(b)] | back 7 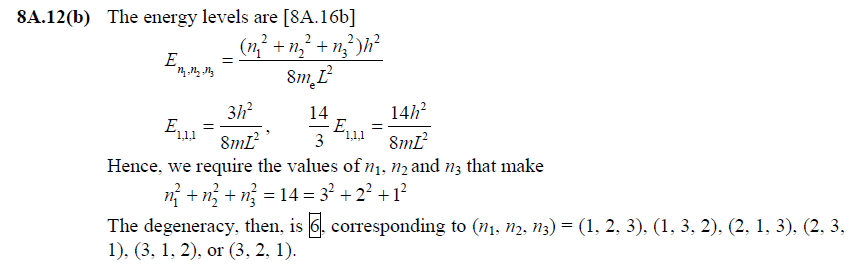 |