Instructions for Side by Side Printing
- Print the notecards
- Fold each page in half along the solid vertical line
- Cut out the notecards by cutting along each horizontal dotted line
- Optional: Glue, tape or staple the ends of each notecard together
Campbell Biology Chapter 1 to 5 test preparation
front 1 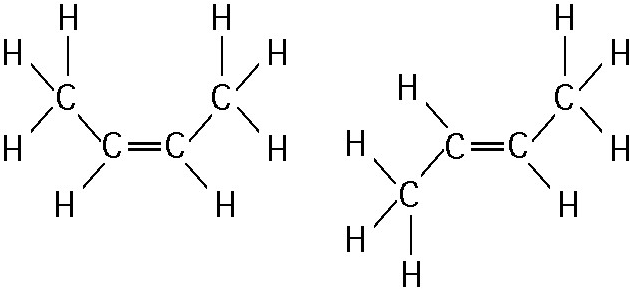 1) The two molecules shown in the figure above are best described
as | back 1 Answer: D |
front 2 2) Which of the following correctly describes any reaction that has
reached chemical equilibrium? | back 2 Answer: D |
front 3 3) Which of the following statements is true about buffer
solutions? | back 3 Answer: A |
front 4 4) In living systems molecules involved in hydrogen bonding almost
always contain either oxygen or nitrogen or both. How do you explain
this phenomenon | back 4 Answer: D |
front 5 5) About twenty-five of the ninety-two natural elements known to be
essential to life. Which four of these twenty-five elements make up
approximately 96 percent of living matter? | back 5 Answer: B |
front 6 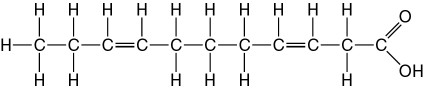 10) The molecule illustrated in the accompanying figure_____. | back 6 Answer: B |
front 7 11) Which of the following is the strongest evidence that protein
structure and function are correlated? | back 7 Answer: B |
front 8 12) What is the difference between covalent bonds and ionic
bonds? | back 8 Answer: D |
front 9 13) Which of the functional groups below acts most like an acid in
water? | back 9 Answer: C |
front 10 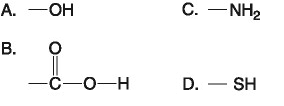 14) Which functional group shown above is a characteristic of
alcohols? | back 10 Answer: A |
front 11 15) Which of the following includes all of the pyrimidines found in
RNA and DNA? | back 11 Answer: C |
front 12 16) Fluorine has an atomic number of 9. Which of the following would
you do to a neutral fluorine atom to complete its valence
shell? | back 12 Answer: A |
front 13 17) The element present in all organic molecules is_____. | back 13 Answer: C |
front 14 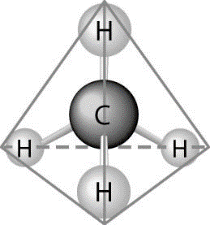 18) What causes the shape of the molecule shown above? | back 14 Answer: A |
front 15 19) Why is carbon so important in biology? | back 15 Answer: C |
front 16 20) Which of the following is NOT a polymer? | back 16 Answer: C |
front 17 21) Agrobacterium infects plants and causes them to form tumors. You
determine that tumor formation requires a large amount of the plant’s
energy for tissue formation. How might this change the number of
offspring a plant produces, and what is the most likely explanation
for this change? | back 17 Answer: D |
front 18 22) A salamander relies on hydrogen bonding to stick to various
surfaces. Therefore, a salamander would have the greatest difficulty
clinging to a______. | back 18 Answer: A |
front 19 23) Amino acids are acids because they always possess which
functional group? | back 19 Answer: A |
front 20 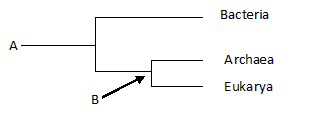 24) The phylogenetic tree below_____. A) Includes noncellular life-forms | back 20 Answer: B |
front 21 25) A carbon atom is most likely to form what kind of bond(s) with
other atoms? | back 21 Answer: C |
front 22 26) Which of the following types of cells utilize deoxyribonucleic
acid (DNA) as their genetic material but do not have their DNA encased
within a nuclear envelope? | back 22 Answer: A |
front 23 27) Why does ice float in liquid water? | back 23 Answer: C |
front 24 28) The molar mass of glucose is 180 grams per mole (g/mol). Which of
the following procedures should you carry out make a 1 M solution of
glucose? Into 0.8 liter (L) of water, dissolve____. | back 24 Answer: D |
front 25 29) How does a scientific theory differ from a scientific
hypothesis? | back 25 Answer: C |
front 26 30) If a DNA sample were composed of 10% thymine, what would be the
percentage of guanine? | back 26 Answer: B |
front 27 31) The atomic number of chlorine is 17. The atomic number of
magnesium is 12. What is the formula for magnesium chloride? | back 27 Answer: C |
front 28 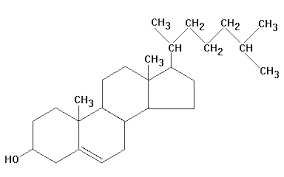 32) The molecule shown the accompanying figure is a _____. | back 28 Answer: A |
front 29 33) You find yourself standing next to a beautiful rose bush. Which
of the following do you and the rose have in common? | back 29 Answer: B |
front 30 34) The experimental approach taken in current biological
investigations presumes that____. | back 30 Answer: A |
front 31 35) Which level of protein structure do the a-helix and the B-pleated
sheet represent? | back 31 Answer: B |
front 32 36) Increased atmospheric CO2 concentrations might have what effect
on seawater? | back 32 Answer: B |
front 33 Use the following information when answering the corresponding
question(s). | back 33 Answer: C |
front 34 38) Why are hydrocarbons insoluble in water? | back 34 Answer: A |
front 35 39) Which of these provides evidence of the common ancestry of all
life? | back 35 Answer: D |
front 36 40) Which polysaccharide is an important component in the structure
of many animals and fungi? | back 36 Answer: D |
front 37 41) Which type of bond must be broken for water to vaporize? | back 37 Answer: A |
front 38 42) If the cytoplasm of a cell is at pH 7, and the mitochondrial
matrix is at pH 8, then the concentration of H+ ions_______. | back 38 Answer: B |
front 39 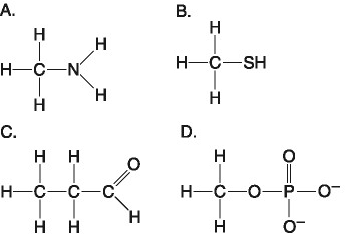 43) Which molecule shown above contains an amino acid functional
group, but is NOT an amino acid? | back 39 Answer: A |
front 40 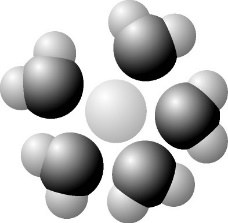 44) Based on your knowledge of the polarity of water molecules, the solute molecule depicted here is most likely_______. A) Nonpolar | back 40 Answer: D |
front 41 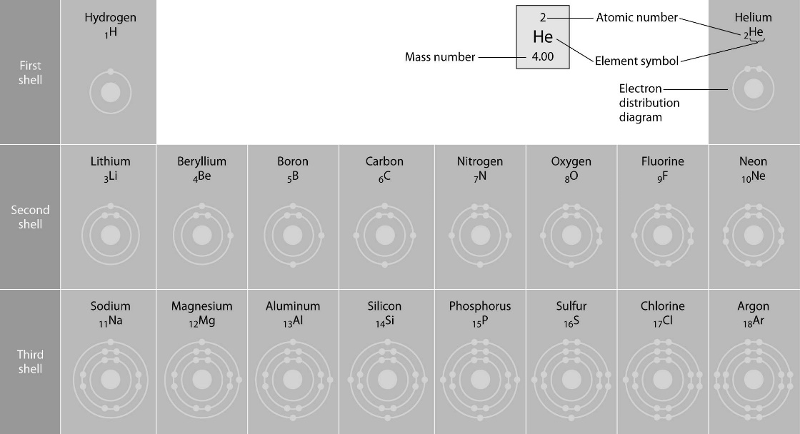 45) Refer to the figure above (first three rows of the periodic
table). What element has properties most similar to carbon? | back 41 Answer: A |
front 42 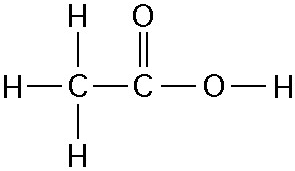 46) One mole of the compound above would weigh how many grams? (Note:
The atomic masses, in daltons, are approximately 12 for carbon, 1 for
hydrogen, and 16 for oxygen.) | back 42 Answer: C |
front 43 47) Normal hemoglobin is a tetramer, consisting of two molecules of B
hemoglobin and two molecules of a hemoglobin. In sickle-cell disease,
as a result of a single amino acid change, the mutant hemoglobin
tetramers associate with each other and assemble into large fibers.
Based on this information alone, we can conclude that sickle-cell
hemoglobin exhibits______. | back 43 Answer: D |
front 44 48) A controlled experiment_______. | back 44 Answer: D |
front 45 49) Which of these is an example of inductive reasoning? | back 45 Answer: C |
front 46 50) Hydrophobic substance such as vegetable oil are_____. | back 46 Answer: D |