Instructions for Side by Side Printing
- Print the notecards
- Fold each page in half along the solid vertical line
- Cut out the notecards by cutting along each horizontal dotted line
- Optional: Glue, tape or staple the ends of each notecard together
Results for Chemical and Physical Changes and Properties SD
front 1 What was observed during the following process? Mg (s) and H C l (a q) | back 1  |
front 2 What is the nature of the change in this process? : Mg (s) and H C l(a q) | back 2  |
front 3 What was observed during the following process? Zn (s) and C u S O4(a q) | back 3  |
front 4 What is the nature of the change in this process? : Zn (s) and C u S O4(a q) | back 4  |
front 5 What was observed during the following process? C u S O4(a q) and H C l(a q) | back 5  |
front 6 What is the nature of the change in this process? : C u S O4(a q) and H C l(a q) | back 6  |
front 7 What was observed during the following process? C u S O4(a q) and N a H C O3(a q) | back 7  |
front 8 What is the nature of the change in this process? : C u S O4(a q) and N a H C O3(a q) | back 8  |
front 9 What was observed during the following process? C u S O4(a q) and N H4 O H(a q) | back 9  |
front 10 What is the nature of the change in this process? : C u S O4(a q) and N H4 O H(a q) | back 10  |
front 11 What was observed during the following process? Mg (s) and C u S O4(a q) | back 11  |
front 12 What is the nature of the change in this process? : Mg (s) and C u S O4(a q) | back 12  |
front 13 What was observed during the following process? H C l(a q) and N a H C O3(a q) | back 13  |
front 14 What is the nature of the change in this process? : H C l(a q) and N a H C O3(a q) | back 14  |
front 15 What was observed during the following process? N H4 C l(a q) and H2O | back 15  |
front 16 What is the nature of the change in this process? : N H4 C l(a q) and H2O | back 16  |
front 17 What is the product of the following reaction equation, after balancing? If there was no reaction, choose "No Reaction" | back 17  |
front 18 What is the product of the following reaction equation? If there was no reaction, choose "No Reaction" | back 18 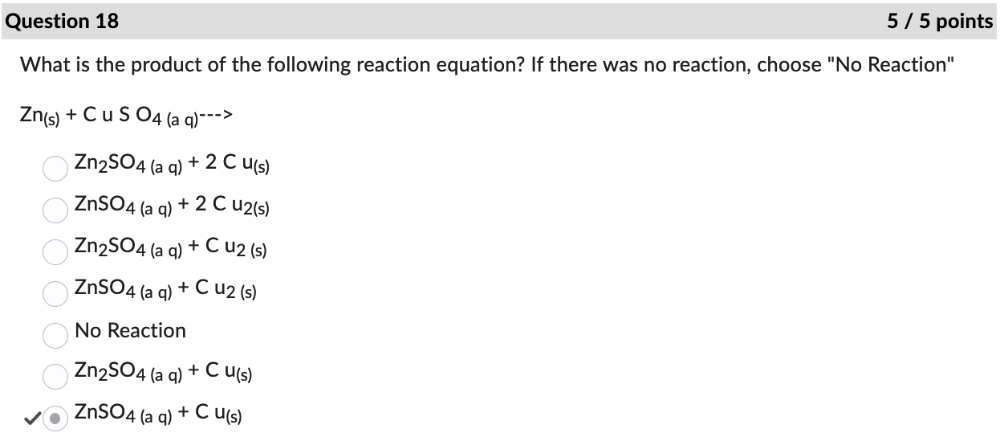 |
front 19 What is the product of the following reaction equation? If there was no reaction, choose "No Reaction" | back 19  |
front 20 What is the product of the following reaction equation? If there was no reaction, choose "No Reaction" | back 20  Ex. Net Ionic Equation for CuSO4 + NaHCO3 = CuCO3 + Na2SO4 + H2O + CO2 https://www.youtube.com/watch?v=cM4MdMhOXMY |
front 21 What is the product of the following reaction equation? If there was no reaction, choose "No Reaction" | back 21  |
front 22 What is the product of the following reaction equation? If there was no reaction, choose "No Reaction" | back 22 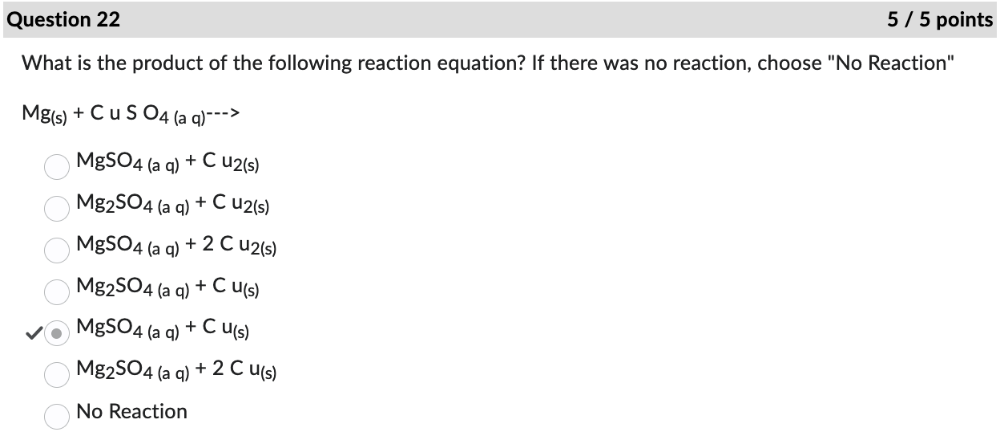 |
front 23 What is the product of the following reaction equation? If there was no reaction, choose "No Reaction" | back 23 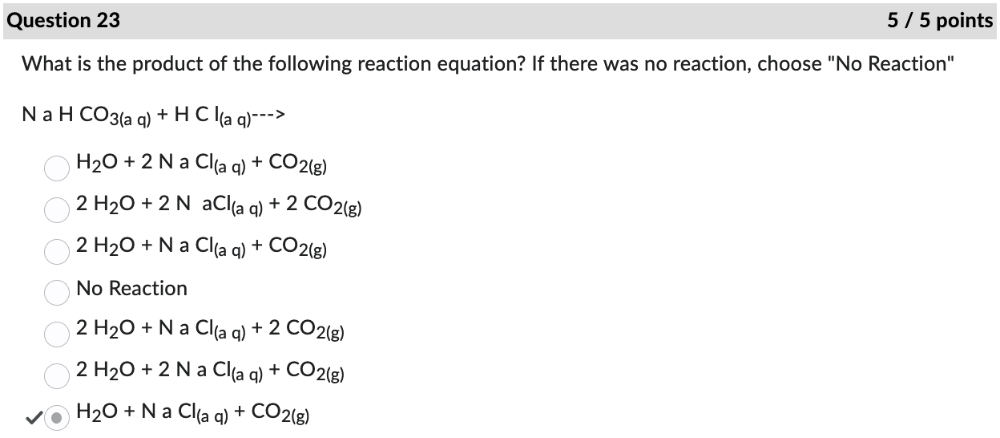 |
front 24 What is the product of the following reaction equation? If there was no reaction, choose "No Reaction" | back 24  |
front 25 Your instructor will give you instructions as what to do with this box. It may be used for bonus points, or for taking away points (for example late submissions) or for error analysis if something went wrong during the lab. If no instructions have been provided to you, please leave this box blank. | back 25 no data |