Instructions for Side by Side Printing
- Print the notecards
- Fold each page in half along the solid vertical line
- Cut out the notecards by cutting along each horizontal dotted line
- Optional: Glue, tape or staple the ends of each notecard together
Analytic techniques/week 6 reading
front 1 What are the four basic components in all mass spectrometers? | back 1 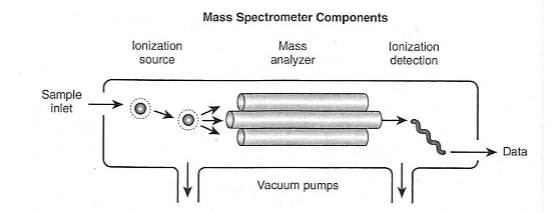
|
front 2 What is electron ionization(EI) in mass spec? | back 2 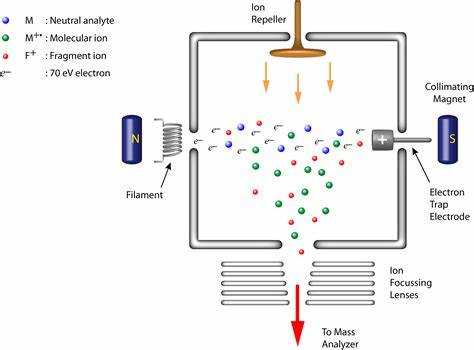 It requires a source of electrons in the form of a filament to which electric potential is applied, typically at 70 eV. |
front 3 What is electrospray ionization? | back 3 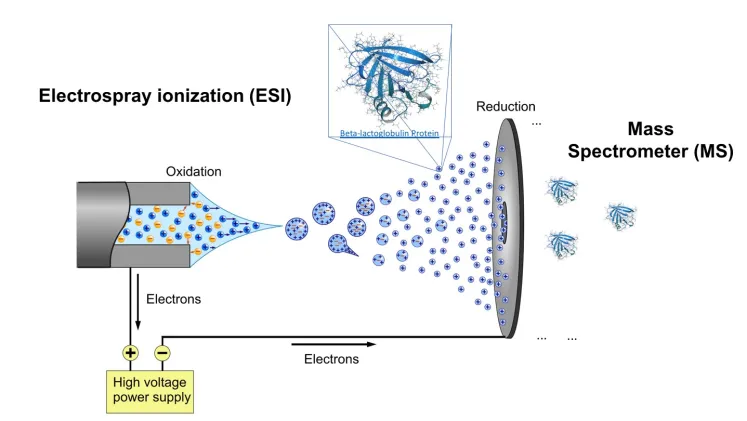 Passing the LC effluent through a capillary to which a voltage has been applied. The energy is transferred to the solvent droplets which become charged.
|
front 4 What is an atmospheric pressure chemical ionization? | back 4 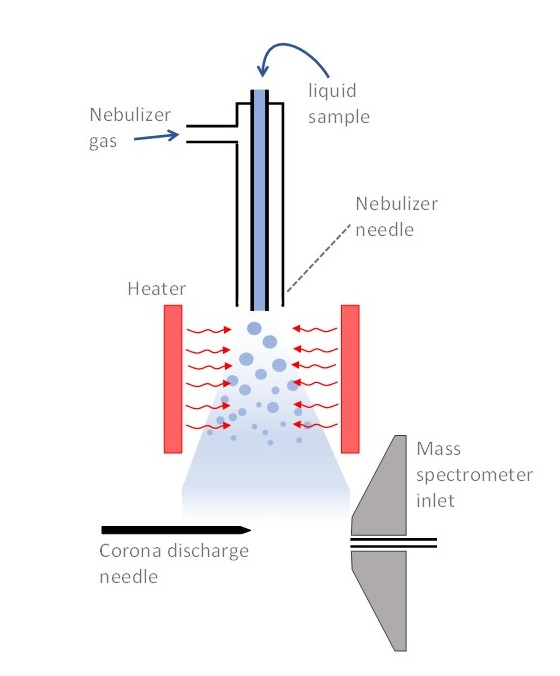 Liquid from LC is introduced directly into the ionization source. However the droplets are not charged and the source contain a heated vaporizer to allow rapid desolvation of the drops.
|
front 5 What is Matrix-Assisted Laser Desorption Ionization-Time of Flight(MALDI-TOF)? | back 5 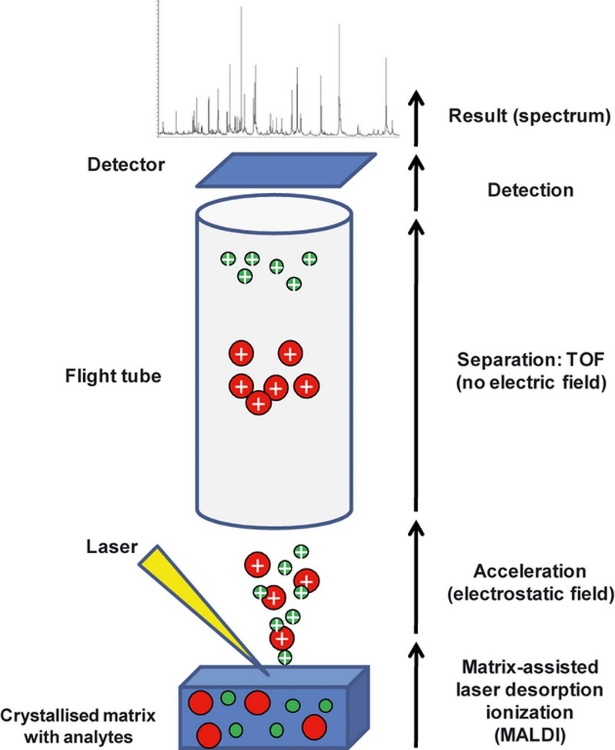 Ionization that is used for the analysis of biomolecules such as peptides and proteins. |
front 6 What is a quadrupole? | back 6 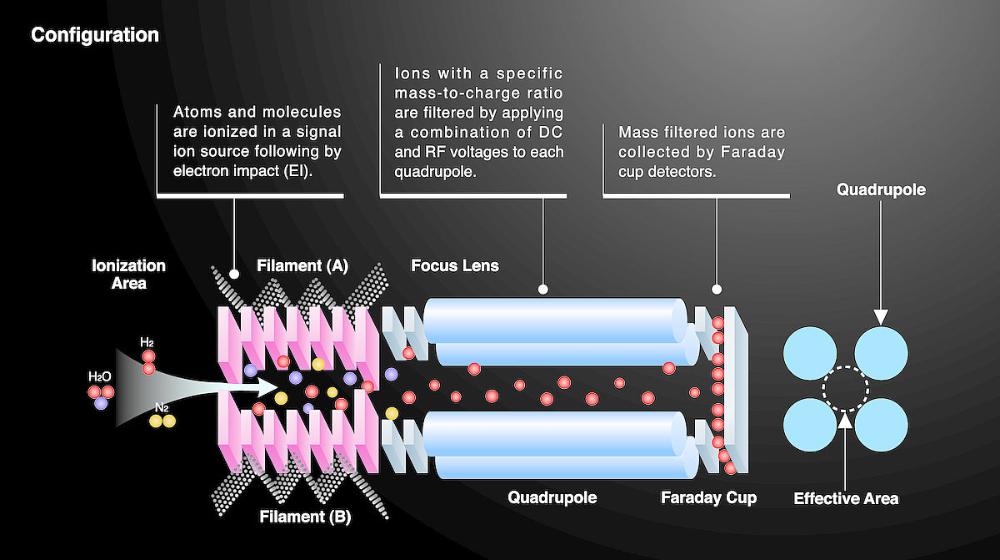 The analyzer consists of four rods or electrodes arranged across from each other. As the ions travel through the quadrupole they are filtered according to their m/z(mass to charge ratio) value so that only a single m/z value ion can strike the detector. |
front 7 What is selected ion monitoring(SIM)? | back 7 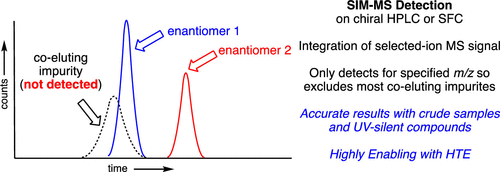
|
front 8 What is an ion trap? | back 8 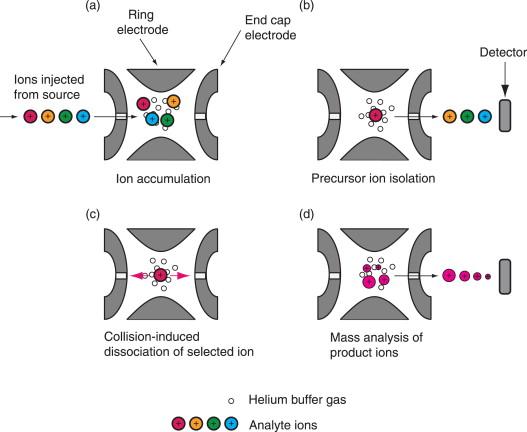
|
front 9 What is tandem mass spectrometry? | back 9 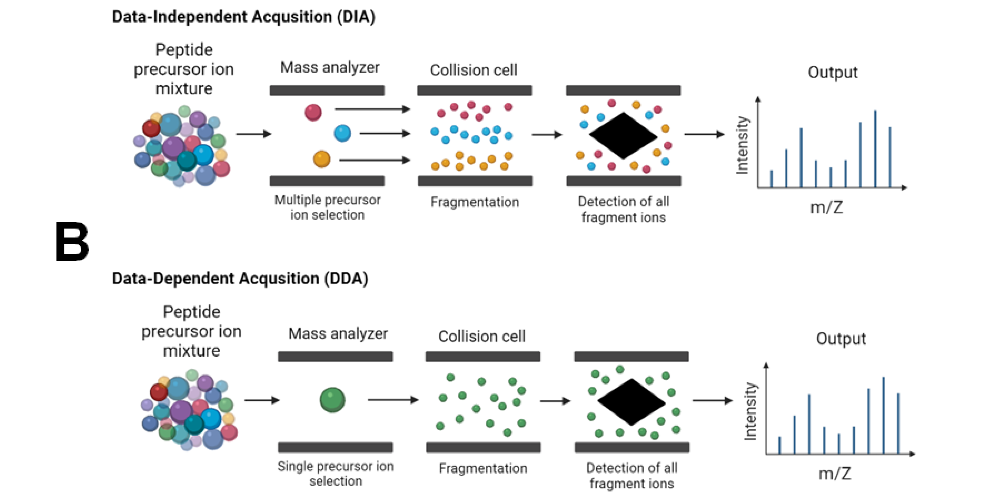
|
front 10 What is collision induced (activated) dissociation? | back 10 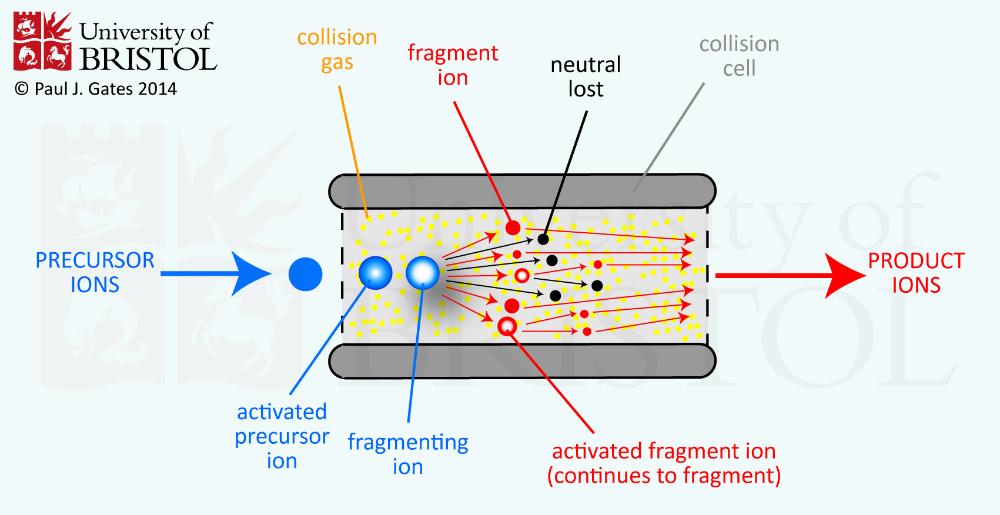 Ions are accelerated to high kinetic energy and allowed to collide with neutral gas molecules to fragment the ions |
front 11 What is small molecule analysis used for? | back 11 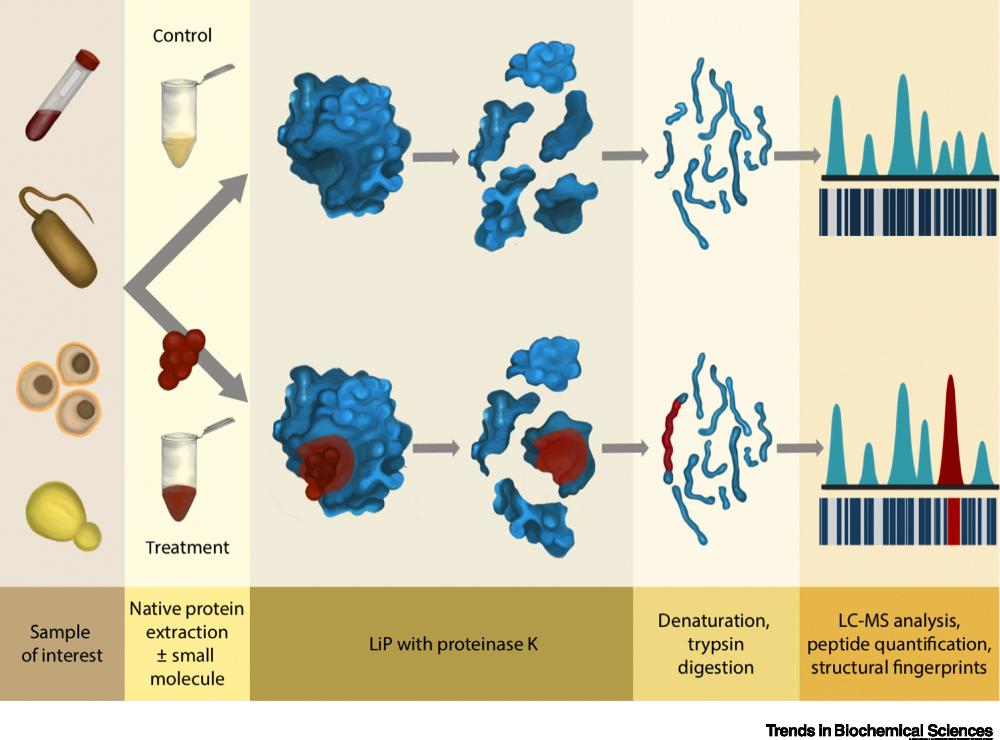
|
front 12 What is proteomics? | back 12 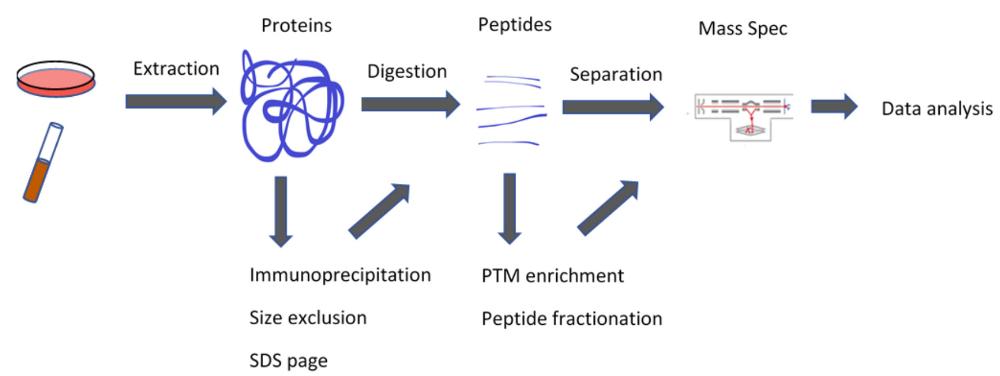 is the large-scale study of proteomes. A proteome is a set of proteins produced in an organism, system, or biological context. |
front 13 How does Matrix-Assisted Laser Desorption Ionization-Time of Flight (MALDI-TOF) detect pathogens? | back 13 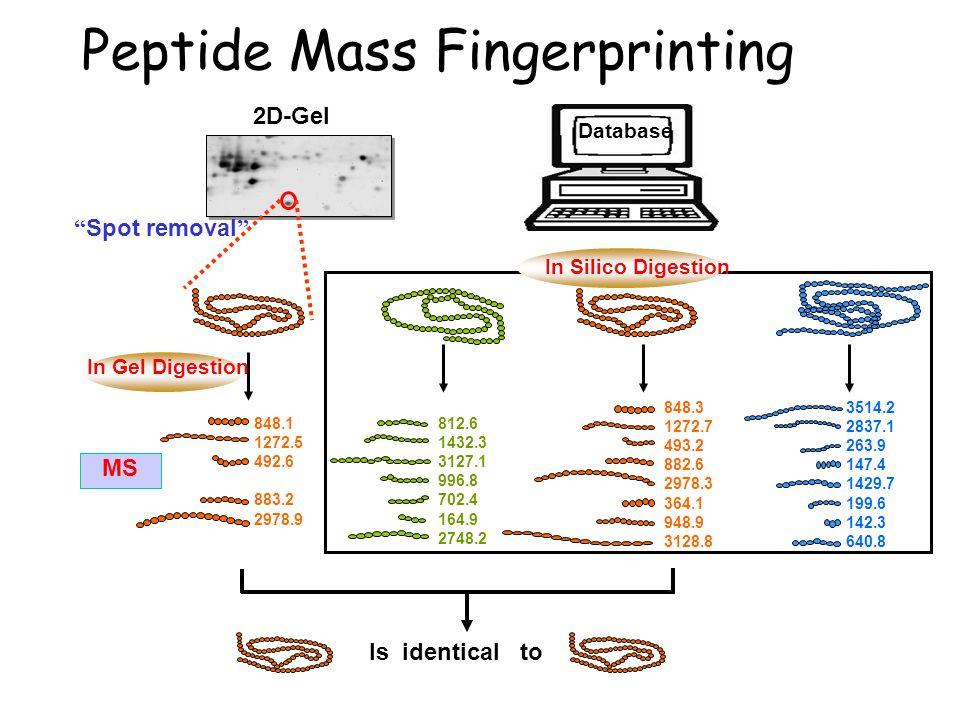 Isolated bacterial or fungal colonies can be directly spotted on the MALDI plate and ionized, which results in a "protein fingerprint" comprised mainly of ribosomal proteins. |
front 14 What is the most common mass separator in use today? | back 14 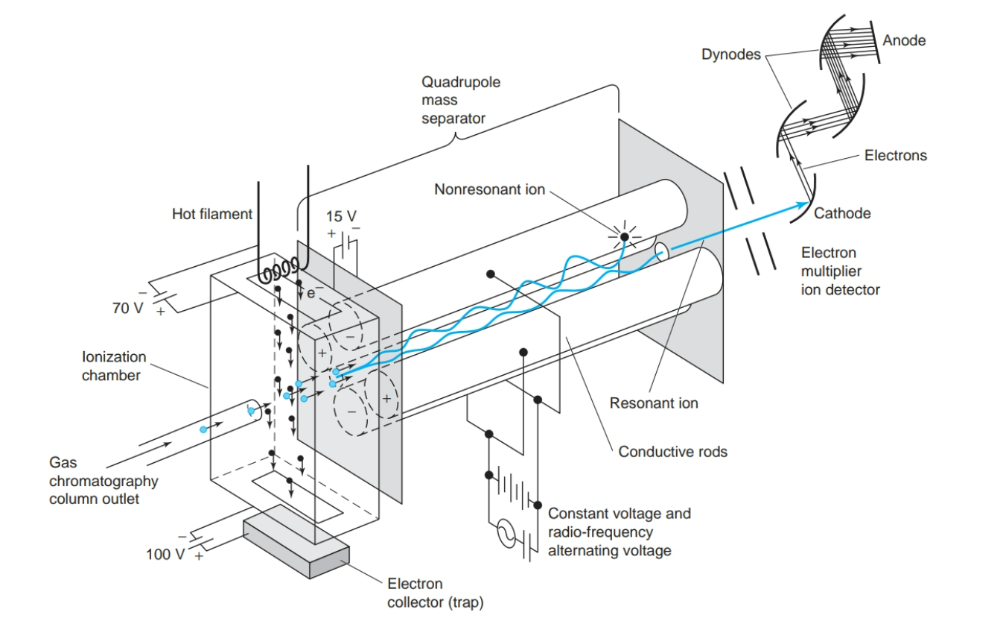 transmission quadrupole mass spectrometer |
front 15 What is electron ionization? | back 15 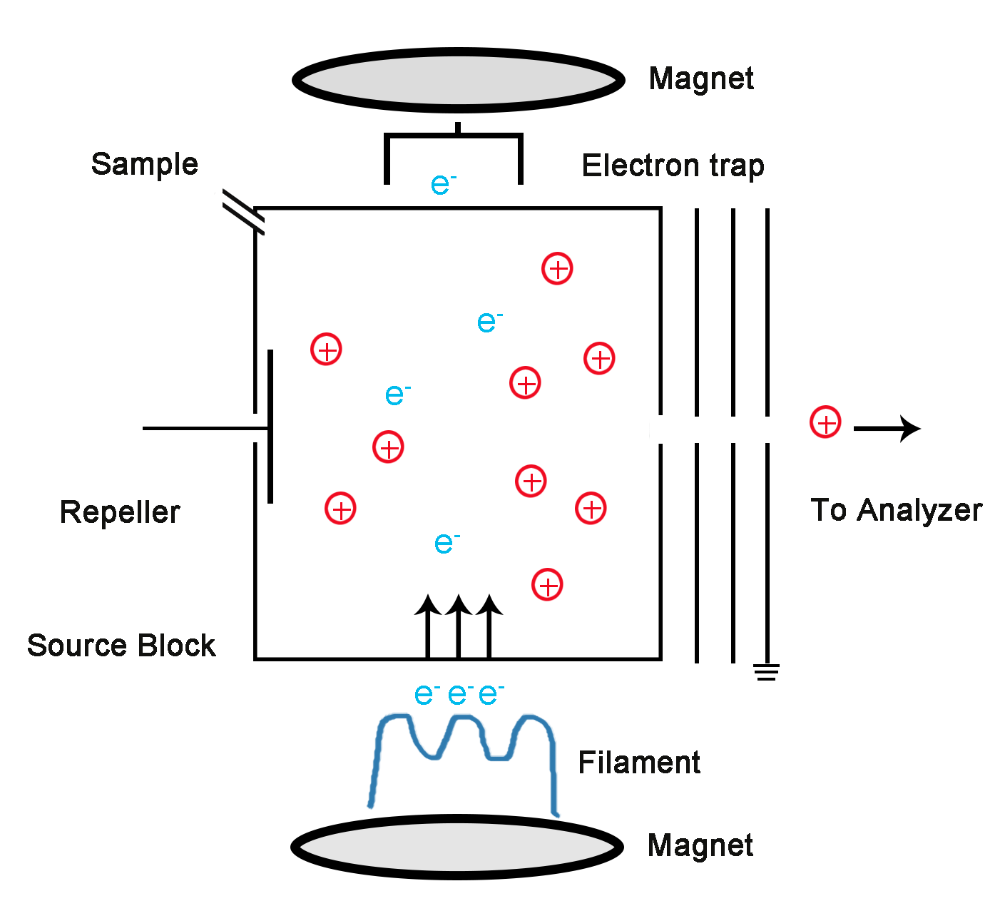 Molecules entering the ionization chamber are converted into ions |
front 16 What is a molecular ion? | back 16 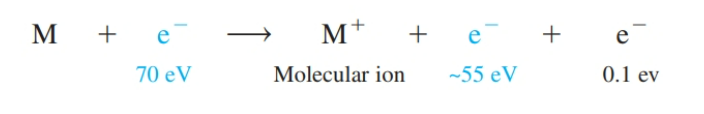 Some (,0.01%) analyte molecules (M) absorb enough energy (9–15 electron volts, eV) to ionize. The resulting cation, M+. |
front 17 What is a base peak? | back 17 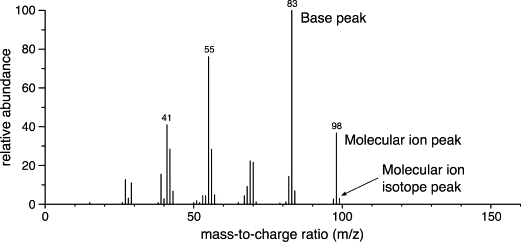 The most intense peak in a mass spectrum |
front 18 How is chemical ionization different from electron ionization? | back 18  it usually produces less fragmentation than electron ionization. For chemical ionization, the ionization chamber contains a reagent gas such as methane at a pressure of ,1 mbar. Energetic electrons (100–200 eV) convert CH4 into a variety of products: |
front 19 What is a reconstructed total ion chromatogram? | back 19 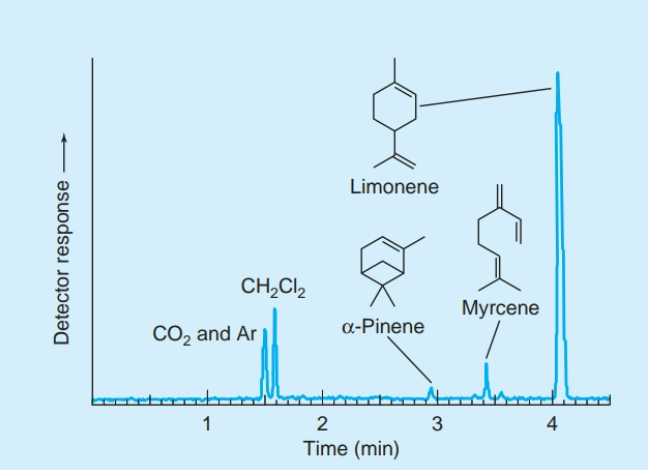 One way to use a mass spectrometer as a detector for chromatography is to record the total current from all ions produced by eluate. |
front 20 What are selected ion chromatograms? | back 20 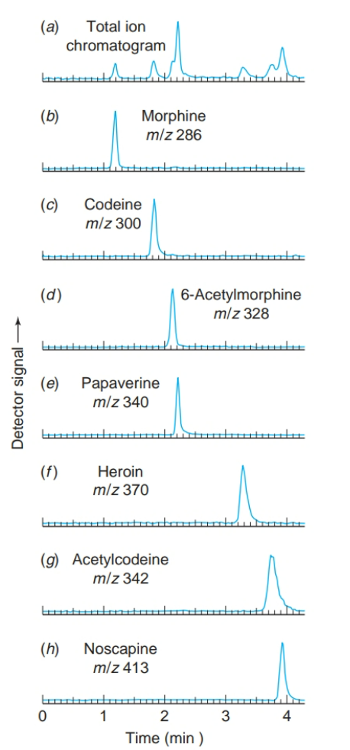 selected ion chromatograms in which the mass spectrometer is set to respond to just one mass in each trace. |
front 21 What is Atomic mass? | back 21 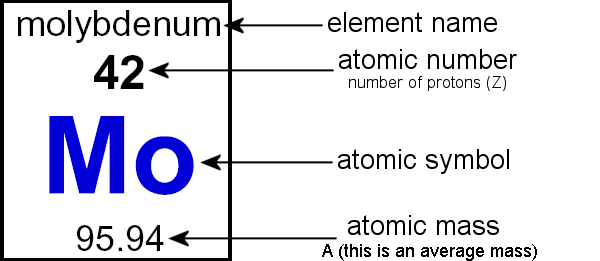 is the weighted average of the masses of the isotopes of an element |
front 22 What is molecular mass? | back 22 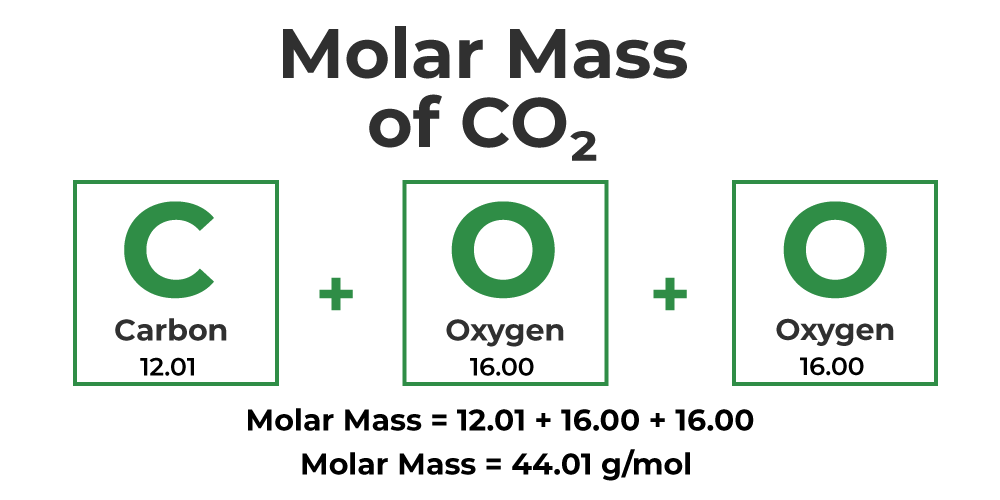 of a molecule or ion is the sum of atomic masses listed in the periodic table |
front 23 What is nominal mass? | back 23 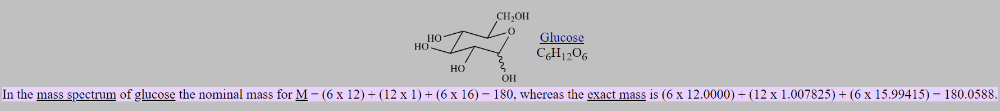 a molecule or ion is the integer mass of the species with the most abundant isotope of each of the constituent atoms |
front 24 What is the nitrogen rule? | back 24 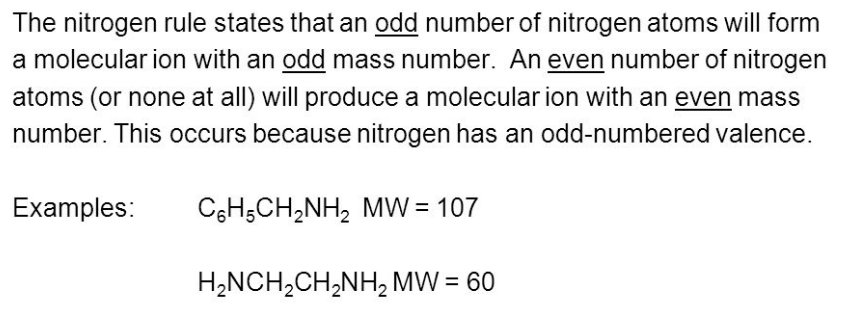 If the molecular mass of an unknown compound to the nearest integer value is an odd number, the compound contains an odd number of nitrogens in its molecular formula. Correspondingly, if the molecular mass is an even number, the compound contains zero or an even number of nitrogens in its molecular formula. |
front 25 What are each sample injection best at? | back 25 • split: routine method • splitless: best for quantitative analysis • on-column: best for thermally unstable solutes |
front 26 How does a flame ionization detector work? | back 26 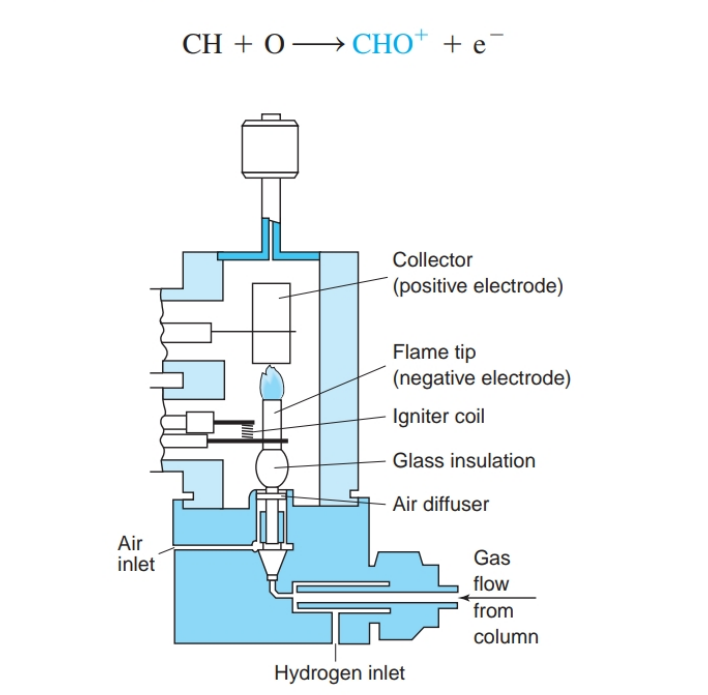 eluate is burned in a mixture of H2 and air. Carbon atoms (except carbonyl and carboxyl carbon atoms) produce CH radicals, which go on to produce CHO+ ions in the flame: |
front 27 What is Hydrophilic interaction chromatography (HILIC) is used for? | back 27 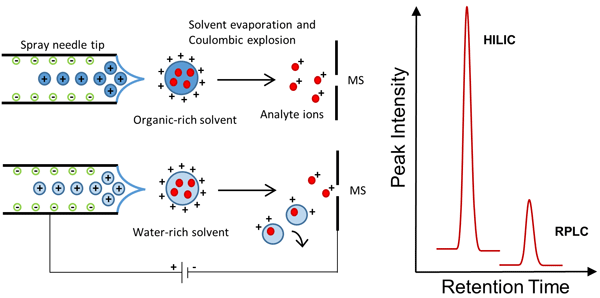 to separate molecules that are too polar to be retained by reversed-phase columns. In biochemistry, HILIC is useful for separating peptides and saccharides (sugars). |