Choose the paramagnetic species from below.
O2⁻
Zn
Ca
Cd2⁺
Nb3⁺
Nb3⁺
Each of the following sets of quantum numbers is supposed to specify an orbital. Which of the following sets of quantum numbers contains an error?
n = 1, l = 0, ml =0
n = 3, l = 0, ml =0
n = 3, l =3 , ml = -2
n = 2, l = 1 , ml = -1
n = 4, l = 2, ml =0
n = 3, l =3 , ml = -2
Each of the following sets of quantum numbers is supposed to specify an orbital. Choose the one set of quantum numbers that does not contain an error.
n = 4, l = 4, ml =0
n = 3, l = 1, ml = -2
n = 4, l = 0, ml =-1
n = 5, l = 3, ml =-3
n = 3, l = 2, ml =+3
n = 5, l = 3, ml =-3
Give the ground state electron configuration for Se.
[Ar]4s23d10
[Ar]4s24d104p4
[Ar]3d104p4
[Ar]4s23d104p6
[Ar]4s23d104p4
[Ar]4s23d104p4
Give the ground state electron configuration for Sr.
[Kr]5s25d105p2
[Kr]5s24d105p2
[Kr]5s24d2
[Kr]5s2
[Kr]5s24d10
[Kr]5s2
Give the set of four quantum numbers that could represent the last electron added (using the Aufbau principle) to the Sr atom.
n = 4, l = 1, ml = 1, ms = -1/2
n = 5, l = 1, ml =1 , ms = -1/2
n = 5, l = 1, ml = 0, ms = +1/2
n = 5, l = 0, ml = 0, ms = -1/2
n = 4, l = 1, ml = -1, ms = +1/2
n = 5, l = 0, ml = 0, ms = -1/2
How many different values of ml are possible in the 3d sublevel?
5
1
3
7
2
5
How many different values of ml are possible in the 4f sublevel?
5
2
3
1
7
7
How many of the following elements have 1 unpaired electron in the
ground state?
B Al S Cl
Answers:
2
1
4
3
3
How many sublevels are contained in the second shell (n=2) of a given atom?
3
1
4
9
2
2
How many unpaired electrons are present in the ground state Kr atom?
3
2
0
5
1
0
How many valence electrons does an atom of S have?
6
3
2
1
4
6
Identify the element that has a ground state electronic configuration of [Ar]4s23d104p1.
Ga
Al
In
B
Ga
Identify the location of the visible range of light.
between microwave and radio
between gamma ray and ultraviolet
between x-ray and gamma ray
between x-ray and ultraviolet
between ultraviolet and infrared
between ultraviolet and infrared
In which orbital below would an electron (on average) be farthest from the nucleus?:
2p
3s
3d
4f
1s
4f
Place the following elements in order of increasing atomic
radius.
P Ba Cl
Answers:
Ba < Cl < P
P < Cl < Ba
Ba < P < Cl
Cl < P < Ba
Cl < Ba < P
Cl < P < Ba
Place the following in order of increasing radius.
Br⁻ Na⁺ Rb⁺
Answers:
Rb⁺ < Br⁻ < Na⁺
Br⁻ < Rb⁺ < Na⁺
Rb⁺ < Na⁺ < Br⁻
Br⁻ < Na⁺ < Rb⁺
Na⁺ < Br⁻ < Rb+
Na⁺ < Br⁻ < Rb+
Place the following types of electromagnetic radiation in order of
increasing frequency.
visible light microwaves X-rays
Answers:
microwaves < visible light < X-rays
visible light < X-rays < microwaves
X-rays < microwaves < visible light
microwaves < X-rays < visible light
X-rays < visible light < microwaves
microwaves < visible light < X-rays
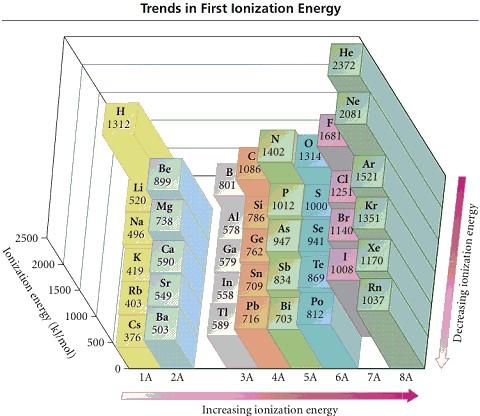
Refer to the figure. Place the following in order of increasing
IE1.
N F As
Answers:
As < N < F
N < As < F
F < N < As
F < N < As
As < F < N
As < N < F
What are the possible orbitals for n = 3?
s, p, d
s, p, d, f
s, p
s
s, p, d
When filling degenerate orbitals, electrons fill them singly at first, with parallel spins. This is know as
Aufbau principle
Hund's rule
Heisenberg uncertainty principle
Pauli's exclusion principle
Hund's rule
Which of the following occur as the wavelength of a photon increases?
the energy increases
the frequency decreases
Planck's constant decreases
the speed decreases
None of the above occur as the wavelength of a photon increases.
the frequency decreases
Which of the following quantum numbers describes the shape of an orbital?
magnetic quantum number
principal quantum number
Schrödinger quantum number
spin quantum number
angular momentum quantum number
angular momentum quantum number
Which of the following statements is TRUE?
An orbital that penetrates into the region occupied by core electrons is less shielded from nuclear charge than an orbital that does not penetrate and therefore has a lower energy.
An orbital that penetrates into the region occupied by core electrons is more shielded from nuclear charge than an orbital that does not penetrate and therefore has a lower energy.
Two electrons in the same orbital can have the same spin.
It is possible for two electrons in the same atom to have identical values for all four quantum numbers.
None of the above are true.
An orbital that penetrates into the region occupied by core electrons is less shielded from nuclear charge than an orbital that does not penetrate and therefore has a lower energy.
Which of the following types of electromagnetic radiation has the shortest wavelength?
green
yellow
violet
blue
orange
violet
Which of the following visible colors of light has the highest frequency?
orange
red
green
yellow
blue
blue
Which reaction below represents the second ionization of Sr?
Sr⁺(g) + e⁻ → Sr(g)
Sr(g) → Sr⁺(g) + e⁻
Sr2⁺(g) + e⁻ → Sr⁺(g)
Sr⁺(g) → Sr2⁺(g) + e⁻
Sr⁻(g) + e⁻ → Sr2⁻(g)
Sr⁺(g) → Sr2⁺(g) + e⁻
Which reaction below represents the second electron affinity of S?
S2⁻(g) → S⁻(g) + e⁻
S⁻(g) → S(g) + e⁻
S⁻(g) + e⁻ → S2⁻(g)
S(g) → S⁺(g) + e⁻
S(g) + e⁻ → S⁻(g)
S⁻(g) + e⁻ → S2⁻(g)
Write out the orbital diagram that represents the ground state of As. How many unpaired electrons are there?
0
3
2
1
4
3